-
Carotid artery stenosis (CAS) can lead to chronic cerebral hypoperfusion, recurrent ischemic stroke, and even disability or death, making it a serious threat to people's life and health. Studies have shown that approximately 50% of patients with severe carotid stenosis have associated cognitive decline[1, 2]. Approximately 1 in 6 patients with cognitive dysfunction may suffer an acute stoke, which leads to a huge financial burden on the family and society[3]. A previous study showed that cognitive dysfunction can be used as a predictor of mortality[4], and an investigation of carotid stenosis patients with cognitive dysfunction is necessary.
Coronary artery stenosis can lead to various forms of cognitive impairment. Although several published studies investigated potential risk factors of cognitive impairment in patients with CAS, inconsistent results were found[5-7]. The current study aimed to investigate potential risk factors of cognitive dysfunction in CAS patients. In addition to general cognitive function, we also considered memory function.
-
This was a cross-sectional, single-center study to evaluate potential risk factors of cognitive dysfunction in CAS patients. The study protocol was approved by the Institutional Review Board of Beijing Tiantan Hospital, Capital Medical University. Forty-seven CAS patients diagnosed by computed tomography angiography (CTA) in our institute from 2016 to 2018 were recruited. All CAS patients had stenosis ≥ 70%. The diagnostic criteria were based on the guidelines for CAS (criteria of the North American Symptomatic Carotid Endarterectomy Trial[8]), and the procedures followed were in accordance with the institution's guidelines. Patients were examined 1 week before the operative procedure. Written informed consent was obtained from all patients.
-
Age, gender, history of hypertension, diabetes, hyperlipidemia, smoking history, and other general characteristics were evaluated as potential risk factors. A history of infarction was defined as having once suffered a clinically diagnosed ischemic stroke. Abnormal blood perfusion, the degree of carotid stenosis, carotid artery plaque characteristics and potency of the Willis circle were also analyzed for their relationship with cognitive dysfunction. A potent Willis circle was defined as an anterior communicating artery, and at least one post communicating artery was sufficient.
-
All patients were examined by carotid CTA to evaluate the severity of carotid stenosis. A total of 40 patients agreed to undergo intracranial CTA to assess the Willis circle and intracranial arteries.
A total of 39 patients underwent computed tomography perfusion (CTP) examination to evaluate intracranial perfusion. Thirty-six patients underwent contrast-enhanced ultrasonography to determine whether plaques were vulnerable. In 19 patients, carotid artery high-resolution MRI was used to evaluate whether carotid artery plaques were ulcerated. The imaging findings were assessed and analyzed by experienced imaging specialists.
-
We used the Mini-Mental State Examination (MMSE) and Montreal Cognitive Assessment (MoCA) to assess overall cognitive function. The Wechsler Memory Scale (WMS) was used to evaluate memory function. An MMSE scale score of less than 27 points or an MoCA score of less than 26 points was defined as cognitive dysfunction[9-11]. A memory quotient score of less than 70 was defined as a memory disorder[12, 13]. Scale tests and analyses were performed by the same experienced clinical psychologist.
-
All statistical analyses were performed with SPSS 23.0 (Windows version 23.0, IBM). Normally and non-normally distributed continuous data were compared using the Student's t-test and the Wilcoxon rank-sum test test, respectively. Multiple factor analysis was performed by logistic regression. Pearson correlation analysis was used to assess the relation between risk factors and cognitive impairment. A probability value of P < 0.05 was considered to indicate significance.
-
A total of 47 patients were enrolled in the study, among which 10 (21.28%) were female and 36 (78.72%) were male. The average age of the enrolled patients was 62 ± 0.41 years (range: 44 to 73 years). Thirty-two (68.1%) patients were diagnosed with hypertension, 16 (34.0%) with diabetes, 12 (25.5%) with heart disease, and 8 (17.0%) with hyperlipidemia. Twenty-eight patients (59.6%) had a history of cerebral infarction, 22 (46.8%) had a history of alcohol drinking, and 27 (57.4%) had a history of smoking. Seventeen (36.2%) patients had completed education above high school.
-
To determine the impact of the potency of the Willis circle on cognition, a total of 40 patients underwent intracranial CTA examination, and 19 (40.4%) of them showed a potent Willis circle. Considering that the degree of stenosis may influence cognitive function, patients were categorized as having severe stenosis (carotid stenosis > 70%) or occlusion (carotid stenosis ≥ 99%). Thirty-nine (83.98%) patients were found to have severe stenosis and 8 (16.02%) had carotid occlusion. A total of 39 patients underwent CTP examination to assess intracranial perfusion. Results showed that 8 (20.51%) patients had normal CTP, and the other 31 (79.49%) patients showed abnormal intracranial perfusion. Thirty-six patients underwent contrast-enhanced ultrasonography to determine vulnerable plaques, and 25 (72.22%) patients were found to have vulnerable plaques. The characters of plaques were analyzed using high-resolution MRI, and 7 (36.84%) of 19 patients showed ulceration in their carotid plaques.
-
A total of 47 patients agreed to undergo cognitive function assessment. Results of the MMSE showed that 22 people (46.8%) had cognitive impairment. MoCA test results showed that 10 people (21.3%) had an impaired cognitive function (Figure 1). The WMS test showed that 1, 2, and 9 patients had severe, moderate, and mild memory impairment, respectively (Figure 2). We conducted an independent-sample t-test to evaluate the relationships between various risk factors and each test score. We found that the average memory quotient of females was 81.70, significantly higher than that of males (P = 0.024). Cognitive and memory performance was significantly better in patients with a higher vs. a lower educational background (P = 0.045). The MMSE scores of patients with abnormal intracranial perfusion were worse (P = 0.024) than those of patients with normal perfusion. Patients with abnormal blood perfusion scored an average of 76.97 on the WMS, significantly lower than those with normal perfusion, who scored an average of 92.25 (P = 0.007). Patients with a history of brain infarction also obtained a lower score on the MMSE (P = 0.047) and WMS (P = 0.018). As for other potential risk factors, no significant relation was found between any of the factors and the decreased score (Table 1). Moreover, no statistical significance was found when the above risk factors were analyzed via binary logistic regression analysis. In an analysis of the relationship between risk factors and cognitive impairment by Pearson correlation analysis, cerebral hypoperfusion was found to be a risk factor by MoCA (P = 0.015) and a history of infarction was a risk factor for memory impairment (P = 0.031) (Table 2).
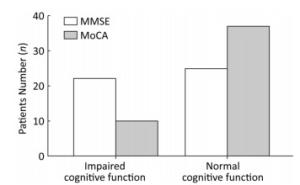
Figure 1. The general cognitive function of patients in this study. The MMSE results showed that 22 people (46.8%) had cognitive impairment. The MoCA test results showed that 10 people (21.3%) had impaired cognitive function.
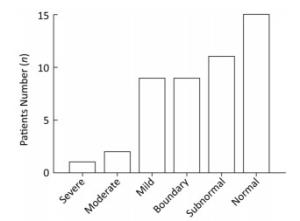
Figure 2. Results of WMS in 47 patients with CAS. One patient had severe memory impairment, 2 had moderate memory impairment, 9 had mild memory impairment, 8 were in a borderline condition, 12 had subnormal and 15 had normal memory.
Table 1. Analysis of Risk Factors and Different Cognitive Assessment Score by t-test
Item MoCA MMSE WMS n Score P n Score P n Score P Gender male 37 20.81 0.851 37 23.78 0.116 37 79.30 0.024 female 10 18.60 10 26.90 10 81.70 Age, years ≥ 55 37 20.19 0.538 37 24.86 0.930 37 78.81 0.178 < 55 10 22.80 10 24.70 10 82.40 Education ≥ high school 17 23.18 0.178 17 25.86 0.002 17 88.88 0.045 < high school 30 18.73 30 21.94 30 74.67 Side unilateral 25 20.52 0.765 25 24.44 0.586 25 78.32 0.575 bilateral 22 21.00 22 25.27 22 81.00 Unilateral side left 11 20.00 0.559 11 24.55 0.719 11 75.27 0.912 right 14 20.93 14 24.36 14 80.71 Symptom N 17 21.24 0.448 17 24.64 0.871 17 83.29 0.264 Y 30 19.83 30 24.33 30 77.83 Ulcerated plagues N 12 22.25 0.851 12 25.33 0.116 12 83.50 0.342 Y 7 24.43 7 26.49 7 84.29 Vulnerable plagues N 11 20.00 0.814 11 23.63 0.511 11 75.00 0.904 Y 25 20.64 25 24.04 25 80.88 Willis circle close 21 21.24 0.889 21 25.43 0.646 21 81.00 0.964 open 19 21.00 19 24.68 19 80.79 Cerebral perfusion normal 8 24.88 0.024 8 24.38 0.868 8 92.25 0.007 abnormal 31 20.13 31 24.74 31 76.97 Degree of stenosis severe 39 20.26 0.103 39 79.64 0.951 39 24.15 0.322 occlusion 8 20.75 8 80.63 8 25.86 Hypertension N 15 20.53 0.857 15 25.80 0.383 15 81.47 0.587 Y 32 20.84 32 24.38 32 78.69 Diabetes N 31 21.39 0.261 31 24.87 0.940 31 82.94 0.450 Y 16 19.50 16 24.75 16 73.06 Hyperlipemia N 39 21.21 0.200 39 25.13 0.386 39 79.54 0.973 Y 8 18.50 8 23.38 8 79.75 History of infarction N 19 22.63 0.047 19 25.32 0.600 19 86.21 0.018 Y 28 19.46 28 24.50 28 75.07 Alcohol N 25 21.64 0.230 25 24.12 0.319 25 82.20 0.238 Y 22 19.73 22 25.64 22 76.59 Smoking N 20 20.55 0.834 20 23.55 0.145 20 83.40 0.163 Y 27 20.89 27 25.78 27 76.74 Note.N, No; Y, Yes. Table 2. Relation between Risk Factors and Cognitive Impairment according to Pearson Correlation
Item MoCA MMSE WMS Coefficient P Coefficient P Coefficient P Female 0.208 0.160 -0.860 0.567 0.108 0.472 Age 0.172 0.246 -0.175 0.239 0.089 0.552 High education -0.169 0.257 0.262 0.075 0.267 0.069 Bilateral stenosis -0.135 0.365 -0.060 0.689 -0.008 0.957 Symptomatic 0.141 0.345 0.138 0.355 0.096 0.522 Ulcerated plagues -0.321 0.180 0.012 0.962 -0.031 0.898 Vulnerable plagues -0.072 0.678 0.069 0.688 0.007 0.965 Willis circle closing -0.087 0.889 -0.150 0.355 0.087 0.594 Cerebral hypoperfusion 0.387 0.015 -0.088 0.593 0.298 0.065 Carotid occlusion 0.006 0.971 -0.029 0.847 -0.027 0.857 Hypertension -0.192 0.197 0.090 0.550 0.015 0.919 Diabetes 0.112 0.455 -0.046 0.759 0.258 0.079 Hyperlipemia 0.135 0.364 -0.142 0.340 -0.027 0.857 History of infarction 0.214 0.149 0.270 0.067 0.316 0.031 Alcohol 0.060 0.687 -0.145 0.329 0.278 0.059 Smoking -0.100 0.944 -0.117 0.432 0.147 0.323 -
Considering that patients with a history of cerebral infarction and abnormal intracranial perfusion had a lower scores on the WMS, we analyzed the subgroup scoring of the WMS to determine the influence of abnormal perfusion and infarction on specific memory domains. We found that patients with a history of cerebral infarction had a significantly lower score than those with no history of cerebral infarction (P = 0.044). In subtests including associative learning, free recall of a picture, recognition of meaningless figures, and portrait character association retrieval, patients with no history of infarction performed better, but no statistical difference was found. Also, patients with intracranial perfusion abnormalities experienced a significant decrease in directed memory compared with those with normal perfusion (P = 0.003). Also, patients with normal cerebral perfusion had a significantly higher score on recognition of meaningless figures (P = 0.012). Patients with normal cerebral perfusion obtained higher scores on other subtests; however, no significant difference was found. The scoring distribution is shown in Table 3.
Table 3. WMS Subgroup Score Based on History of Infarction and Cerebral Perfusion
Item History of Infarction Cerebral Perfusion Yes
(n = 19)No
(n = 28)P Abnormal
(n = 31)Normal
(n = 8)P Directed memory 10.00 13.53 0.044 10.03 16.25 0.003 Associative learning 11.86 11.89 0.979 12.03 14.25 0.215 Free recall of picture 12.21 14.37 0.185 12.97 16.25 0.113 Recognition of meaningless figures 15.46 18.37 0.152 16.13 20.38 0.012 Portrait character association retrieval 12.75 15.74 0.082 13.29 16.38 0.163 -
Carotid stenosis, as an important factor in vascular dementia, has drawn a lot of attention and invoked intensive discussion among researchers. Studies focused on the degree of carotid stenosis, plaque characteristics, and cerebral perfusion have been conducted to determine the relationship between cognitive dysfunction and carotid stenosis. However, a more comprehensive study is needed to identify potential factors that may lead to cognitive impairment in patients with carotid stenosis.
Both the MMSE and the MoCA scale are currently used to assess the overall cognitive performance of adults, and both scoring systems have their merits and drawbacks. The MMSE is more suitable for patients with moderate or severe dementia, but the test is also appropriate for patients with impaired cognitive domains other than the main complaint of memory loss. In contrast, the MoCA scale is more suitable for patients with mild cognitive impairment, especially those with impairment of individual cognitive domains, such as memory impairment. A combined use of the two scales may improve diagnostic sensitivity, which will help to uncover more potential risk factors of cognitive impairment.
After assessing age, gender, education level, hypertension, diabetes, tobacco use, and alcohol use, we found that patients with an educational background above high school performed better, and females scored higher on the WMS test. Other factors showed no significant correlation with cognitive impairment. Although differences in cognitive performance between male and female patients were not found in several previous studies[14, 15], one study suggested that anti-estrogen endocrine therapy in patients with breast cancer is associated with aberrant verbal learning/memory[16]. Another study indicated that estrogen could improve spatial memory by vasodilation and increased cerebral blood flow[17]. We speculated that estrogen may act as a protective factor of patients' memory function. We found that those with a level of education above high school scored higher on the MMSE and WMS, in agreement with an earlier study[18]. People with higher education may have more active minds, which may slow down the decline of cognitive function. Although other studies have shown the possibility of a correlation between other factors and cognitive impairment, no significant association was found in our study[19-22].
Cognitive function is closely related to the degree of carotid stenosis. Previous studies have shown that patients with severe carotid stenosis scored significantly lower on the MMSE than those with moderate carotid stenosis[23]. A study found that attention, memory, visual spatial ability, and language fluency were significantly lower in patients with carotid stenosis than in healthy controls and negatively related to the degree of carotid artery stenosis[24]. We predicted that carotid artery occlusion may lead to a more severe decrease in the cerebral blood supply compared with severe stenosis, which is likely to be one of the factors responsible for aggravated cognitive dysfunction. No significant difference in cognitive function was found between patients with or without carotid occlusion. We speculated that a sufficient collateral blood supply has formed in the process of carotid occlusion, which results in the stabilization of cerebral blood perfusion in the process.
According to the Gray-Weale classification, vulnerable plaques are highly susceptible to rupture and eventually lead to clinical events, especially subtle cognition impairment[25]. Microthrombosis and quickly accentuated carotid stenosis caused by plaques were thought to be the reason for impaired cognition. Vulnerable plaques have a strong relationship with cognitive impairment among stroke-free patients[26, 27]. We identified vulnerable plaques using contrast-enhanced ultrasound, but no significant correlation between plaque vulnerability and impaired cognition was found. We expected that ulcerated plagues would be more likely to cause microthrombosis, so we used high-resolution MRI to assess the characters of carotid plaques. Interestingly, ulcerated plaques were not associated with the decline of cognition. Patients with a history of cerebral ischemia were not excluded, which may have led to the lack of correlation between vulnerable or ulcerated plagues and cognitive disfunction. Further research should be carried out to determine in more detail the role of plagues in the progression of cognitive impairment.
Studies have demonstrated that patients with ischemic stroke have a better prognosis when they have a collateral blood supply than when they do not. In addition, patients with a high collateral compensative capacity would have a better response to revascularization surgery[28, 29]. There are usually three types of collateral circulation, including the Willis circle, one of the most efficient ways[29]. We expected that a potent Willis circle could improve the blood supply and alleviate the decline of cognitive function. However, no obvious relationship between a potent Willis circle and cognitive function was observed in the current study. One study indicated that without considering the factor of age, atherosclerosis of the Willis circle is associated with dementia[30]. In another study, although the role of the Willis circle in the process of cognitive impairment was not explained, it was noted that occlusion of the Willis circle is one of the independent risk factors for early postoperative cognitive impairment[31], indicating that the Willis circle could influence cognitive function. Furthermore, other types of collateral arteries may impact the progress of cognitive impairment in CAS patients. Further studies should be conducted to determine the association between collateral circulation and cognitive dysfunction.
Cerebral atherosclerosis may lead to decreased cerebral vascular reserve capacity and decreased cerebral blood flow, which can result in abnormal brain function as well as cognitive dysfunction[32]. Currently, some scholars propose that long-term hypoperfusion leads to the white matter disorder, which may lead to cognitive impairment[33]. Other studies have shown that hypoperfusion may lead to decreased neuronal activity, abnormal brain connections, and hence, to cognitive dysfunction[34]. In this study, we also found that patients with normal intracranial perfusion had significantly higher MoCA scores and memory quotients than those with abnormal perfusion. It is clear that cerebral hypoperfusion in patients with carotid artery stenosis is one of the most important factors leading to cognitive dysfunction. But this study did not clarify the association between the specific area of low perfusion and decreased cognitive function.
-
As this was a cross-sectional study, the role of various potential factors in the development of cognitive impairment could not be clearly defined, and potential factors such as the role of ulcerated plagues in the progress of stenosis may have been omitted. In addition, we had a relatively small sample size of 47 patients, which may undermine the power of the correlation between these risk factors and cognitive impairment in CAS patients.
-
This study aims to identify factors associated with cognitive dysfunction in patients with carotid stenosis. We found that a history of cerebral infarction and abnormal cerebral perfusion were associated with a decline in overall cognitive function and memory. Being female and having an education level above high school may be protective factors in the progress of cognitive dysfunction.
doi: 10.3967/bes2018.107
An Integrated Analysis of Risk Factors of Cognitive Impairment in Patients with Severe Carotid Artery Stenosis
-
Abstract:
Objective To investigate cognitive dysfunction in patients with carotid artery stenosis (CAS) and potential risk factors related to cognitive-especially memory-dysfunction. Methods Forty-seven patients with carotid artery stenosis were recruited into our study cohort. The Mini-Mental State Examination (MMSE) and the Montreal Cognitive Assessment (MoCA) were adopted to assess cognitive function, the Wechsler Memory Scale (WMS) to assess memory function, high-resolution MRI and enhanced ultrasound to evaluate carotid plaques, and computed tomography perfusion (CTP) imaging to evaluate intracranial blood perfusion. Single-factor analysis and multiple-factor regression analysis were used to analyze potential risk factors of cognitive impairment. Results Mini-Mental State Examination test results showed that 22 patients had cognitive impairment, and MoCA test results showed that 10 patients had cognitive impairment. Analysis of various risk factors indicated that the average memory quotient of female patients was higher than that of males (P=0.024). The cognitive and memory performance of those with an educational background above high school were significantly better than those of participants with high school or lower (P=0.045). Patients with abnormal intracranial perfusion performed worse on the MMSE test (P=0.024), and their WMS scores were significantly lower (P=0.007). The MMSE scores and the memory quotients were significantly lower in patients with a history of cerebral infarction (MMSE, P=0.047, memory quotient score, P=0.018). Conclusion A history of cerebral infarction and abnormal cerebral perfusion are associated with decline in overall cognitive function and memory in patients with carotid stenosis. Being female and having an educational background above high school may be protective factors in the development of cognitive dysfunction. -
Key words:
- Carotid artery stenosis /
- Cognitive impairment /
- Memory
-
Table 1. Analysis of Risk Factors and Different Cognitive Assessment Score by t-test
Item MoCA MMSE WMS n Score P n Score P n Score P Gender male 37 20.81 0.851 37 23.78 0.116 37 79.30 0.024 female 10 18.60 10 26.90 10 81.70 Age, years ≥ 55 37 20.19 0.538 37 24.86 0.930 37 78.81 0.178 < 55 10 22.80 10 24.70 10 82.40 Education ≥ high school 17 23.18 0.178 17 25.86 0.002 17 88.88 0.045 < high school 30 18.73 30 21.94 30 74.67 Side unilateral 25 20.52 0.765 25 24.44 0.586 25 78.32 0.575 bilateral 22 21.00 22 25.27 22 81.00 Unilateral side left 11 20.00 0.559 11 24.55 0.719 11 75.27 0.912 right 14 20.93 14 24.36 14 80.71 Symptom N 17 21.24 0.448 17 24.64 0.871 17 83.29 0.264 Y 30 19.83 30 24.33 30 77.83 Ulcerated plagues N 12 22.25 0.851 12 25.33 0.116 12 83.50 0.342 Y 7 24.43 7 26.49 7 84.29 Vulnerable plagues N 11 20.00 0.814 11 23.63 0.511 11 75.00 0.904 Y 25 20.64 25 24.04 25 80.88 Willis circle close 21 21.24 0.889 21 25.43 0.646 21 81.00 0.964 open 19 21.00 19 24.68 19 80.79 Cerebral perfusion normal 8 24.88 0.024 8 24.38 0.868 8 92.25 0.007 abnormal 31 20.13 31 24.74 31 76.97 Degree of stenosis severe 39 20.26 0.103 39 79.64 0.951 39 24.15 0.322 occlusion 8 20.75 8 80.63 8 25.86 Hypertension N 15 20.53 0.857 15 25.80 0.383 15 81.47 0.587 Y 32 20.84 32 24.38 32 78.69 Diabetes N 31 21.39 0.261 31 24.87 0.940 31 82.94 0.450 Y 16 19.50 16 24.75 16 73.06 Hyperlipemia N 39 21.21 0.200 39 25.13 0.386 39 79.54 0.973 Y 8 18.50 8 23.38 8 79.75 History of infarction N 19 22.63 0.047 19 25.32 0.600 19 86.21 0.018 Y 28 19.46 28 24.50 28 75.07 Alcohol N 25 21.64 0.230 25 24.12 0.319 25 82.20 0.238 Y 22 19.73 22 25.64 22 76.59 Smoking N 20 20.55 0.834 20 23.55 0.145 20 83.40 0.163 Y 27 20.89 27 25.78 27 76.74 Note.N, No; Y, Yes. Table 2. Relation between Risk Factors and Cognitive Impairment according to Pearson Correlation
Item MoCA MMSE WMS Coefficient P Coefficient P Coefficient P Female 0.208 0.160 -0.860 0.567 0.108 0.472 Age 0.172 0.246 -0.175 0.239 0.089 0.552 High education -0.169 0.257 0.262 0.075 0.267 0.069 Bilateral stenosis -0.135 0.365 -0.060 0.689 -0.008 0.957 Symptomatic 0.141 0.345 0.138 0.355 0.096 0.522 Ulcerated plagues -0.321 0.180 0.012 0.962 -0.031 0.898 Vulnerable plagues -0.072 0.678 0.069 0.688 0.007 0.965 Willis circle closing -0.087 0.889 -0.150 0.355 0.087 0.594 Cerebral hypoperfusion 0.387 0.015 -0.088 0.593 0.298 0.065 Carotid occlusion 0.006 0.971 -0.029 0.847 -0.027 0.857 Hypertension -0.192 0.197 0.090 0.550 0.015 0.919 Diabetes 0.112 0.455 -0.046 0.759 0.258 0.079 Hyperlipemia 0.135 0.364 -0.142 0.340 -0.027 0.857 History of infarction 0.214 0.149 0.270 0.067 0.316 0.031 Alcohol 0.060 0.687 -0.145 0.329 0.278 0.059 Smoking -0.100 0.944 -0.117 0.432 0.147 0.323 Table 3. WMS Subgroup Score Based on History of Infarction and Cerebral Perfusion
Item History of Infarction Cerebral Perfusion Yes
(n = 19)No
(n = 28)P Abnormal
(n = 31)Normal
(n = 8)P Directed memory 10.00 13.53 0.044 10.03 16.25 0.003 Associative learning 11.86 11.89 0.979 12.03 14.25 0.215 Free recall of picture 12.21 14.37 0.185 12.97 16.25 0.113 Recognition of meaningless figures 15.46 18.37 0.152 16.13 20.38 0.012 Portrait character association retrieval 12.75 15.74 0.082 13.29 16.38 0.163 -
[1] Smith EE. Clinical presentations and epidemiology of vascular dementia. Clin Sci (Lond), 2017; 131, 1059-68. doi: 10.1042/CS20160607 [2] Pettigrew LC, Thomas N, Howard VJ, et al. Low mini-mental status predicts mortality in asymptomatic carotid arterial stenosis. Asymptomatic Carotid Atherosclerosis Study investigators. Neurology, 2000; 55, 30-4. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=3bce91b3c3d42dcfbad91e156cce849a [3] Romero JR, Beiser A, Seshadri S, et al. Carotid artery atherosclerosis, MRI indices of brain ischemia, aging, and cognitive impairment:the Framingham study. Stroke, 2009; 40, 1590-6. doi: 10.1161/STROKEAHA.108.535245 [4] Popovic IM, Lovrencic-Huzjan A, Simundic AM, et al. Cognitive performance in asymptomatic patients with advanced carotid disease. Cogn Behav Neurol, 2011; 24, 145-51. doi: 10.1097/WNN.0b013e3182313020 [5] Lal BK, Dux MC, Sikdar S, et al. Asymptomatic carotid stenosis is associated with cognitive impairment. J Vasc Surg, 2017; 66, 1083-92. doi: 10.1016/j.jvs.2017.04.038 [6] Stirland LE, O'Shea CI, Russ TC. Passive smoking as a risk factor for dementia and cognitive impairment:systematic review of observational studies. Int Psychogeriatr, 2017; 1-11. http://europepmc.org/abstract/MED/29249209 [7] Yan Z, Liang Y, Shi J, et al. Carotid stenosis and cognitive impairment amongst older Chinese adults living in a rural area:a population-based study. Eur J Neurol, 2016; 23, 201-4. doi: 10.1111/ene.12906 [8] Ferguson GG, Eliasziw M, Barr HW, et al. The North American Symptomatic Carotid Endarterectomy Trial:surgical results in 1415 patients. Stroke, 1999; 30, 1751-8. doi: 10.1161/01.STR.30.9.1751 [9] Creavin ST, Wisniewski S, Noel-Storr AH, et al. Mini-Mental State Examination (MMSE) for the detection of dementia in clinically unevaluated people aged 65 and over in community and primary care populations. Cochrane Database Syst Rev, 2016; CD011145. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=S1463423617000202 [10] Ciesielska N, Sokołowski R, Mazur E, et al. Is the Montreal Cognitive Assessment (MoCA) test better suited than the Mini-Mental State Examination (MMSE) in mild cognitive impairment (MCI) detection among people aged over 60? Meta-analysis. Psychiatr Pol, 2016; 50, 1039-52. doi: 10.12740/PP/45368 [11] Lu J, Li D, Li F, et al. Montreal cognitive assessment in detecting cognitive impairment in Chinese elderly individuals:a population-based study. J Geriatr Psychiatry Neurol, 2011; 24, 184-90. doi: 10.1177/0891988711422528 [12] Lal BK, Younes M, Cruz G, et al. Cognitive changes after surgery vs stenting for carotid artery stenosis. J Vasc Surg, 2011; 54, 691-8. doi: 10.1016/j.jvs.2011.03.253 [13] Nanba T, Ogasawara K, Nishimoto H, et al. Postoperative cerebral white matter damage associated with cerebral hyperperfusion and cognitive impairment after carotid endarterectomy:a diffusion tensor magnetic resonance imaging study. Cerebrovasc Dis, 2012; 34, 358-67. doi: 10.1159/000343505 [14] Wardlaw JM, Allerhand M, Eadie E, et al. Carotid disease at age 73 and cognitive change from age 70 to 76 years:A longitudinal cohort study. J Cereb Blood Flow Metab, 2017; 37, 3042-52. doi: 10.1177/0271678X16683693 [15] Yue W, Wang A, Zhu R, et al. Association between Carotid Artery Stenosis and Cognitive Impairment in Stroke Patients:A Cross-Sectional Study. PLoS One, 2016; 11, e0146890. doi: 10.1371/journal.pone.0146890 [16] Underwood EA, Rochon PA, Moineddin R, et al. Cognitive sequelae of endocrine therapy in women treated for breast cancer:a meta-analysis. Breast Cancer Res Treat, 2018; 168, 299-310. doi: 10.1007/s10549-017-4627-4 [17] Olver TD, Hiemstra JA, Edwards JC, et al. Loss of Female Sex Hormones Exacerbates Cerebrovascular and Cognitive Dysfunction in Aortic Banded Miniswine Through a Neuropeptide Y-Ca2+-Activated Potassium Channel-Nitric Oxide Mediated Mechanism. J Am Heart Assoc, 2017; 6. http://europepmc.org/articles/PMC5721796/ [18] Everts R, Wapp M, Burren Y, et al. Cognitive and emotional effects of carotid stenosis. Swiss Med Wkly, 2014; 144, w13970. http://europepmc.org/abstract/med/24984222 [19] Liu JT, Lee IH, Wang CH, et al. Cigarette smoking might impair memory and sleep quality. J Formos Med Assoc, 2013; 112, 287-90. doi: 10.1016/j.jfma.2011.12.006 [20] Yuan JQ, Lv YB, Chen HS, et al. Association Between Late-Life Blood Pressure and the Incidence of Cognitive Impairment:A Community-Based Prospective Cohort Study. J Am Med Dir Assoc, 2018. [21] Witt LS, Rotter J, Stearns SC, et al. Heart Failure and Cognitive Impairment in the Atherosclerosis Risk in Communities (ARIC) Study. J Gen Intern Med, 2018; 33, 1721-8. doi: 10.1007/s11606-018-4556-x [22] Ioime L, Guglielmo R, Affini GF, et al. Neuropsychological Performance in Alcohol Dependent Patients:A One-Year Longitudinal Study. Psychiatry Investig, 2018; 5, 505-13. http://europepmc.org/abstract/MED/29674602 [23] Martinić-Popović I, Lovrencić-Huzjan A, Demarin V. Assessment of subtle cognitive impairment in stroke-free patients with carotid disease. Acta Clin Croat, 2009; 48, 231-40. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=Open J-Gate000000302488 [24] Casas-Hernanz L, Garolera M, Badenes-Guia D, et al. The effect of carotid occlusion in cognition before endarterectomy. Arch Clin Neuropsychol, 2012; 27, 879-90. doi: 10.1093/arclin/acs075 [25] Nighoghossian N, Derex L, Douek P. The vulnerable carotid artery plaque:current imaging methods and new perspectives. Stroke, 2005; 36, 2764-72. doi: 10.1161/01.STR.0000190895.51934.43 [26] Rossetti HC, Weiner M, Hynan LS, et al. Subclinical atherosclerosis and subsequent cognitive function. Atherosclerosis, 2015; 241, 36-41. doi: 10.1016/j.atherosclerosis.2015.04.813 [27] Brickman AM, Siedlecki KL, Muraskin J, et al. White matter hyperintensities and cognition:testing the reserve hypothesis. Neurobiol Aging, 2011; 32, 1588-98. doi: 10.1016/j.neurobiolaging.2009.10.013 [28] Bang OY, Saver JL, Kim SJ, et al. Collateral flow averts hemorrhagic transformation after endovascular therapy for acute ischemic stroke. Stroke, 2011; 42, 2235-9. doi: 10.1161/STROKEAHA.110.604603 [29] Shuaib A, Butcher K, Mohammad AA, et al. Collateral blood vessels in acute ischaemic stroke:a potential therapeutic target. Lancet Neurol, 2011; 10, 909-21. doi: 10.1016/S1474-4422(11)70195-8 [30] Suemoto CK, Ferretti RE, Grinberg LT, et al. Association between cardiovascular disease and dementia. Dement Neuropsychol, 2009; 3, 308-14. doi: 10.1590/S1980-57642009DN30400008 [31] Sussman ES, Kellner CP, Mergeche JL, et al. Radiographic absence of the posterior communicating arteries and the prediction of cognitive dysfunction after carotid endarterectomy. J Neurosurg, 2014; 121, 593-8. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=e7cd5505c6ce0e13e853f55b1e69973e [32] Avirame K, Lesemann A, List J, et al. Cerebral autoregulation and brain networks in occlusive processes of the internal carotid artery. J Cereb Blood Flow Metab, 2015; 35, 240-7. doi: 10.1038/jcbfm.2014.190 [33] Turk M, Pretnar-Oblak J, Zupan M, et al. Ultrasound diagnosis of carotid artery stiffness in patients with ischemic leukoaraiosis. Ultrasound Med Biol, 2015; 41, 64-71. doi: 10.1016/j.ultrasmedbio.2014.08.002 [34] Nouri A, Martin AR, Mikulis D, et al. Magnetic resonance imaging assessment of degenerative cervical myelopathy:a review of structural changes and measurement techniques. Neurosurg Focus, 2016; 40, E5. http://www.ncbi.nlm.nih.gov/pubmed/27246488 -




 下载:
下载:



 Quick Links
Quick Links