-
Chronic obstructive pulmonary disease (COPD) is a global health concern, accounting for > 3 million deaths each year[1]. The Global Initiative for Chronic Obstructive Lung Disease (GOLD) defines COPD as follows: a common preventable and treatable disease that is characterized by airflow limitation, which is usually progressive and associated with an enhanced chronic inflammatory response—in the airways and the lung—to noxious particles or gases. Importantly, chronic airway inflammation plays a central role in the pathophysiologic development of COPD, demonstrated either by an increase in circulating cytokines, chemokines, and acute phase proteins, or by abnormalities in circulating cells[1]. Multiple factors are involved in the development of COPD, including genetics, environmental exposures, co-morbidities, and age-related degenerative changes. Thus far, numerous studies have investigated the association between air pollution exposure and hematological changes; however, their conclusions have not been consistent[2]. Therefore, the effects of improved air pollution on the hematological system in COPD patients are not clear.
The systemic inflammatory response is characterized by activation and mobilization of inflammatory cells into systemic circulation, and by production of acute-phase proteins along with circulating inflammatory mediators[3]. A primary component of the systemic inflammatory response is the stimulation of the hematopoietic system, including the bone marrow, which results in the release of leukocytes (e.g., neutrophils, lymphocytes, monocytes, eosinophils, and basophils) and platelets into circulating blood. In recent decades, large population-based studies have consistently demonstrated that circulating leukocyte levels are a good predictor of cardiovascular health effects; this predictive effect remains even after adjustment for other risk factors[4-5]. We and other researchers have performed controlled studies of ambient air pollutant exposure to understand the mechanisms underlying the effects of short-term air pollutant exposure on biological pathways in humans. Previously, we demonstrated that air pollutants do not affect coagulation markers (D-dimmer and blood gas) in COPD patients. However, it is conceivable that air pollutant exposure [especially to nitrogen dioxide (NO2)] could activate systemic inflammation in human subjects, without a requirement for activation of the coagulation cascade. Therefore, we hypothesized that air pollution improvement could reduce hematological parameters, as reflected by decreases in leukocytes, erythrocytes, hemoglobin, and platelet levels in COPD patients[6].
In this study, we used stable COPD as a clinical model to test the hypothesis that air pollution reduction is associated with relative inactivation of the hematological system. This would provide a link between air pollutants and hematological events in patients with stable COPD. We prospectively monitored blood levels of surrogate markers for hematological parameters (leukocytes, erythrocytes, hemoglobin, and platelet levels) in a 3-year study of a COPD patient cohort, comparing 51 intervention days in 2010 with the same calendar dates in two baseline years (2009 and 2011).
The geographical location of the study area has been described in detail previously[6]. The current study was conducted in Guangzhou, China. Guangzhou includes nine different districts and many counties. In 2000, the population of Guangzhou was approximately 13.2 million, representing 1% of China's total. The target population consisted of permanent inhabitants in Haizhu District (90.4 km2), which is within Guangzhou; the total number of inhabitants was estimated at approximately 1.55 million in 2015. Two continuous state-controlled air pollution-monitoring sites were situated in the area. Surrounding air pollution in Haizhu District is mainly caused by emissions from automobile exhaust. The predominant air pollutants detected were NO2 (nitrogen dioxide), PM10 (particulate matter, a diameter measuring less than 10 μm), and SO2 (sulfur dioxide).
All participants were asked to complete six clinical visits, with two in each of the pre-Asian Game periods (November 1 to December 21, 2009), during the Asian Game periods (November 1 to December 21, 2010), and in the post-Asian Game periods (November 1 to December 21, 2011). Initially, 41 patients were recruited. Only 36 patients participated in all data analyses; the remaining five completed fewer than six visits and were excluded from the analysis. Additional information was obtained by questionnaire at the first visit; this included health status, pulmonary symptoms, and smoking history. Inclusion criteria were: male patients with stable COPD; aged 60-70 years (65.6 ± 2.8); no history of smoking or occupational exposures; no cooking; no medication that might affect systemic inflammation within 24 h prior to a clinical visit; cancer-free and unaffected by other respiratory diseases; and permanent Haizhu District residents living within well-ventilated indoor conditions. The present work includes subjects from a continuing Guangzhou Institute of Respiratory Disease (GIRD) and State Key Laboratory of Respiratory Disease COPD Biobank Project, established by the Guangzhou Institute of Respiratory Disease and the State Key Laboratory of Respiratory Disease. Briefly, it recruited COPD patients during their first hospital admission because of an exacerbation of disease. Airway obstruction was diagnosed using the Global Lung Function Initiative (GLI) definition of FEV1/FVC (43.7 ± 8.6) < the lower limit of normal (LLN) and Z-score.
Blood was drawn using standardized procedures for venipuncture, collection, storage, and shipment at the First Affiliated Hospital of Guangzhou Medical University. Blood samples were immediately placed on ice and centrifuged within 2 h. Blood was collected using 21-gauge needles, then stored in K2-EDTA plasma tubes (Becton Dickinson, Plymouth, UK). Total and differential cell counts were performed using an automated hematology analyzer (CELL-DYN; Abbott Laboratories). Samples were analyzed within 24 h after collection. All blood tests were performed in the translational medicine laboratory of the First Affiliated Hospital of Guangzhou Medical University. The study protocol was approved by the institutional review board of Guangzhou Medical University (Ethics Committee of The First Affiliated Hospital: GZMC2009-08-1336). Each subject provided written informed consent before participating.
Meteorological data (daily mean temperature and relative humidity) were obtained from the Guangzhou Meteorological Bureau to allow adjustment for the effects within our assessment model; this monitoring is consistent with International World Meteorological Organization standards. In addition, outdoor air quality data were provided by the Guangzhou Municipal Environmental Protection Monitoring Center, a central governmental agency. Air concentrations of PM10, SO2, and NO2 were obtained as daily mean value measurements from the two state-controlled monitoring sites in Haizhu District. Both sites are consistent with International World Meteorological Organization standards.
We estimated the change in hematological parameters (leukocytes, erythrocytes, hemoglobin, and platelet levels) between 'before the Games' and 'during the Game period' and between 'during the Game period' to 'after the Games'; we used these hematological parameters as dependent variables, and the time period as the independent variable, in Mixed-Linear Models (MLM) with normally distributed errors. To control the family-wise type Ⅰ error rate at a 0.05 level, a Bonferroni correction was applied. With 32 between-period comparisons (16 biomarkers for each of two between-period changes), each individual 2-sided test was considered statistically significant at a 0.0016 significance level. We then used this model to estimate the change in both neutrophil counts and total leukocyte counts associated with each 10 μg/m3 increase in pollutant concentration during the 24 h prior to each clinic visit (lag day 0), as well as the six previous 24 h periods (lag days 1-6). We report the mean percentage change, along with its 95% CI, for the leukocyte and neutrophil counts associated with each 10 μg/m3 pollutant increase.
All analyses were conducted using the lme and gls packages in R (version 2.14.1; R Development Core Team, Vienna, Austria).
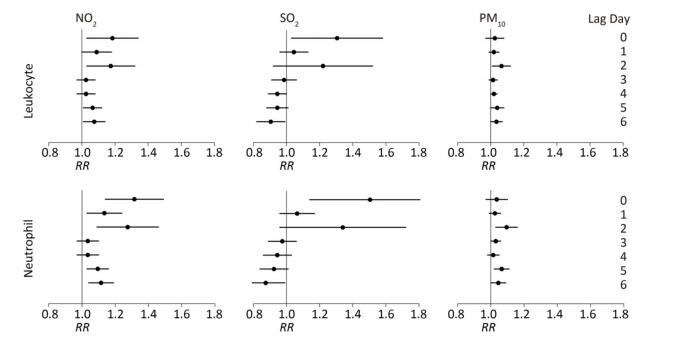
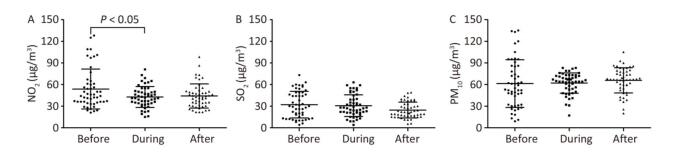
We observed statistically significant improvements in NO2 levels during the intervention days (Figure 1). There were no significant changes to SO2, PM10, or meteorological conditions. Period-specific means, 95% CI, and between-period changes for the assessed hematological parameters, as well as whether these changes were statistically significantly different from 0, are shown in Table 1. As hypothesized, using 2-sided tests conducted at the 0.0016 significance level, we observed statistically significant reductions in leukocyte and neutrophil counts by 15.51% and 23.01%, respectively (95% CI: -16.98% to -14.04% and 95% CI: -25.17% to -20.85%, respectively). Total leukocyte counts decreased from a pre-Asian Games mean of 10.54 × 109 cell/L to a during-Asian Games mean of 8.91 × 109 cell/L; neutrophil counts decreased from 8.48 × 109 cell/L to 6.53 × 109 cell/L. In the post-Asian Game period when pollutant concentrations increased, most pollutant measurements in our study approximated pre-Asian Game levels; thus, we expected that hematological parameters would be significantly worsened from the during-Asian Game period. However, we observed that significant changes were still present in both leukocyte and neutrophil counts. For both leukocyte and neutrophil counts, we detected statistically significant increases that were associated with an increase in NO2 concentration at lag days 0-2/5-6, an increase in SO2 concentration at lag day 0, and an increase in PM10 concentration at lag day 2 (Figure 2).

Figure 1. Comparison of air pollution conditions between the intervention period and the baseline period in Guangzhou.
Biomarker, Units Asian Games Perioda (No. of COPD = 36), Mean (95% CI) Between-Period Percentage Changeb, Δ Mean (95% CI) Before During After Before to During During to After Leukocyte (109/L) 10.54 (9.28, 11.81) 8.91 (7.59, 10.23) 9.66 (8.37, 10.96) -15.51 (-16.98, -14.04) 8.47 (7.84, 9.1) Neutrophil (109/L) 8.48 (7.13, 9.82) 6.53 (5.31, 7.74) 7.41 (6.18, 8.64) -23.01 (-25.17, -20.85) 13.58 (13.1, 14.06) Lymphocyte (109/L) 1.22 (0.93, 1.52) 1.31 (1.12, 1.49) 1.33 (1.15, 1.51) 6.82 (-0.51, 14.15) 1.91 (1.1, 2.72) Monocyte (109/L) 0.77 (0.62, 0.91) 0.75 (0.62, 0.88) 0.72 (0.59, 0.84) -1.81 (-4.97, 1.35) -4.8 (-5.5, -4.1) Eosinophil (109/L) 0.17 (0.14, 0.20) 0.24 (0.08, 0.39) 0.20 (0.14, 0.27) 38.82 (27.45, 50.19) -15.02 (-30.47, 0.43) Basophil (109/L) 0.03 (0.02, 0.03) 0.03 (0.02, 0.04) 0.03 (0.02, 0.04) 7.53 (2.66, 12.4) -1 (-5.23, 3.23) Erythrocyte (1012/L) 4.59 (4.41, 4.78) 4.48 (4.23, 4.72) 4.41 (4.19, 4.64) -2.58 (-6.81, 1.65) -1.37 (-4.02, 1.28) Hematocrit 0.42 (0.41, 0.44) 0.4 (0.38, 0.42) 0.39 (0.37, 0.4) -5.89 (-12.05, 0.27) -2.24 (-5.03, 0.55) MCV (fL) 91.92 (89.74, 94.09) 89.58 (85.96, 93.19) 88.50 (85.58, 91.42) -2.55 (-5.51, 0.41) -1.2 (-3.54, 1.14) MCH (pg) 29.73 (28.92, 30.54) 29.77 (28.44, 31.09) 29.69 (28.42, 30.97) 0.12 (-1.16, 1.4) -0.25 (-1.44, 0.94) MCHC (g/L) 323.4 (319.1, 327.7) 331.9 (327.9, 335.8) 334.6 (328.4, 340.9) 2.61 (-0.08, 5.3) 0.84 (-0.86, 2.54) DWVC (%) 13.99 (13.59, 14.40) 14.18 (13.70, 14.65) 14.54 (14.08, 15.00) 1.31 (-4.18, 6.8) 2.57 (1.81, 3.33) Hemoglobin (g/L) 135.9 (131.0, 140.9) 131.5 (125.4, 137.6) 129.4 (123.6, 135.3) -3.25 (-6.76, 0.26) -1.56 (-3.22, 0.1) Platelet (109/L) 210 (182, 239) 222 (188, 256) 212 (191, 233) 11.66 (8.21, 15.11) 10.39 (8.59, 12.18) MPV (fL) 9.21 (8.69, 9.72) 9.09 (8.66, 9.51) 9.06 (8.51, 9.60) -1.3 (-3.88, 1.28) -0.31 (-2.23, 1.61) PDW (fL) 14.18 (13.25, 15.11) 14.17 (13.2, 15.13) 14.94 (14.09, 15.79) -0.1 (-1.49, 1.29) 5.49 (1.5, 9.48) Plateletcrit (%) 0.19 (0.17, 0.21) 0.19 (0.17, 0.22) 0.19 (0.17, 0.21) 2.63 (-1.07, 6.33) -1.31 (-2.04, -0.58) Note. MCV, mean corpuscular volume; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration; DWVC, distribution width variation coefficient; MPV, mean platelet volume; PDW, platelet distribution width. aBefore the Asian Games represents November 1 to December 21, 2009; during, November 1 to December 21, 2010; and after November 1 to December 21, 2011. bSignificance is established if P value < 0.0016, the individual significance level needed to maintain a family-wise Type Ⅰ error rate of 0.05; P value < 0.0016 is presented in bold. Table 1. Biomarker Concentrations based on Period and Between-Period Changes in Participant-Specific Biomarker Concentrations in COPD Patients
Figure 2. RRs and 95% CIs estimates for changes in hematological indices associated with leukocyte and neutrophil counts, related to each 10 μg/m3 changes in pollutant levels, by 0-6 lag days.
Previously, we and others have concluded that there is an enhanced coagulative state in stable COPD patients; in particular, we reported evidence that mitigated air pollution affects blood coagulability[7]. In the same study, we also determined that blood coagulability was not the most severe effect of exposure to NO2, PM10, and SO2, in our patient sample. However, we suggested that exposure to air pollutants, particularly NO2, could activate systemic inflammation in human COPD subjects, independent from activation of the coagulation cascade. Therefore, we conducted the present study to further investigate the role of air pollutants in COPD exacerbation. In our current study, we observed that air pollution improvement could significantly reduce the levels of COPD-related hematological parameters (leukocytes, erythrocytes, hemoglobin, and platelet levels). This study is notable because it further elucidates the relationship between improved air pollution and hematological indices in COPD patients.
COPD is a chronic, inflammatory lung disease with variable development; some patients experience a relatively stable course of disease, while others suffer relentless progression that results in serious breathlessness, frequent acute exacerbations, respiratory failure, and possible death[8]. Since 25%-45% of patients with chronic airflow limitation are never-smokers[9], this strongly suggests that non-smoking-related factors play a pivotal role in the progression of COPD development[10]. Exposure to air pollution should therefore be highlighted as an important factor that influences COPD patient outcomes. There is thus a growing body of work that supports the notion that air pollution exposure influences COPD progression in susceptible individuals. In recent years, epidemiological studies have identified strong relationships between air pollution exposure and COPD outcomes, including exacerbations, hospital admissions and mortality[6, 11]. Vehicle emissions are a dominant contributor to ambient pollution in the urban environment, as exemplified by the geographical area included in this study. The main polluting components of vehicle emissions are SO2, NO2, and PM10. Thus, we selected PM10, NO2, and SO2 as our target pollutants for the current study. These pollutants could readily deposit within human alveoli and cause impacts on the endothelium, either directly through oxidant effects, or indirectly through increased airway susceptibility to other irritating factors, such as respiratory virus infection. Further, these pollutants could contribute to an increase in biological toxicity, thereby eliciting systemic inflammation and/or altered coagulability. For instance, a study[12] showed that exposure to air pollutants for 1 h at 300 μg/m3 caused increases in peripheral neutrophil levels. Exposure of COPD patients to air pollutants (a principal source of NO2) might induce bodily responses mediated by vascular endothelial cells, leukocytes, and/or platelets, thereby enhancing pulmonary inflammation and increasing levels of circulating platelets, neutrophils, and coagulation factors. Activation of these inflammatory pathways may potentially result in increased endothelial dysfunction, vasoconstriction, or vascular reactivity, thus causing adverse effects in COPD patients. Our current study is consistent with prior studies in finding that NO2 is the most problematic air pollutant for COPD patients; further, our study suggests beneficial public health effects may be achieved by a simple reduction in NO2. These discoveries support efforts to decrease air pollution and improve public health through anthropogenic interventions. Recently, we conducted a meta-analysis, using data from this study together with our previous findings, to determine whether there were consistent outcomes. We suspect that we now have consistent evidence of the positive relationship between NO2 and COPD exacerbation, irrespective of other confounding factors.
The exact mechanisms linking inhalation of air pollutants to COPD exacerbation have not been completely elucidated. Disturbance of the hematological balance is a potential mechanism. The current study evaluated changes in inflammatory markers, particularly hematological indices, which follows from a variety of prior studies. First, evidences showed that various inflammatory cells, including monocytes, lymphocytes, eosinophils, and neutrophils, are implicated in COPD[13-14]. Second, inflammation is an important feature of COPD and its related clinical manifestations. The total leukocyte count (and its associated differential cell counts) provides markers of inflammation that are widely available in clinical practice. Additionally, routine hematological examination is quite easy to interpret and is reliable, inexpensive, and routinely ordered, both in inpatient and outpatient settings. The increase in total leukocyte counts in association with increased air pollution is generally defined as indicative of an elevated inflammatory response. For example, in the Evaluation of COPD Longitudinally to Identify Predictive Surrogate Endpoints (ECLIPSE) study, it[15] demonstrated an association between leukocytes and mortality in stable COPD; after adjustment for potential confounding factors, leukocyte counts were independently associated with mortality rates in COPD patients. In another study, it concluded that simultaneous elevations in leukocytes, fibrinogen, and high-sensitivity CRP (hsCRP) were associated with high exacerbation risk in COPD patients[16]. A cross-sectional study of patients with moderate to severe COPD found that elevated levels of leukocytes were associated with high healthcare resource utilization. It has been identified that increased leukocyte count was a significant predictor of both mortality and hospital admission[17]. This link was present even in milder cases of COPD and in those with no history of frequent exacerbations[16]. Increased differential cell counts, including counts of neutrophils, eosinophils, and monocytes, also predict the future incidence of COPD. In a COPD cohort study, evidence found that clusters of plasma proteins involved in neutrophil function were associated with FEV1 parameters[18]. In the current study of patients with stable COPD, the prognostic values of hematological indices were examined. We observed a statistically significant reduction in leukocyte and neutrophil levels that was associated with mitigated air pollution during the 2010 Asian Games. This study implies that decreased air pollution during the Asian Games is associated with reduced activation of the hematological system in COPD patients. We further suggest that air pollution might serve as an initiating factor for the inflammatory response in COPD.
In conclusion, we suggest that diminished air pollution was associated with reductions in leukocyte and neutrophil counts during the 2010 Asian Games. This implies a possible mechanism to link air pollutants to hematological events in stable COPD patients. This study further elucidates the relationship between improved air pollution and hematological system. Additional studies involving other susceptible populations and endpoints are pending.
HTML
National Natural Science Foundation of China 81520108001
the 973 Key Scheme of China 2015CB553406
Guangdong Natural Science Foundation 2016A030313593
Guangzhou Department of Education for Innovative Team 13C08
National Natural Science Foundation of China 81700043







 Quick Links
Quick Links
 DownLoad:
DownLoad: