-
Reactive oxygen species (ROS) not only alters signaling pathways in the cells but also influences genetic and epigenetic changes, thereby modulating cellular proliferation and differentiation. It has been reported that ROS production plays a role in hydroquinone-induced inhibition of erythroid differentiation of K562 cells and HD3 chicken erythroblast cells[1-2]. Reducing the levels of ROS using antioxidants was found to abolish lactic acid-induced erythroid differentiation of hematopoietic stem cells (HSCs) and K562 cells, which indicates that ROS is also a critical regulator of this process[3]. N-acetylcysteine (NAC) and ascorbic acid (AA) are well-known antioxidants that can scavenge oxygen-derived free radicals in vitro and in vivo. They are also potential treatment options for diseases characterized by the generation of free oxygen radicals. In this study, we investigated the changes in ROS levels in NAC-, AA-, and hemin-treated K562 cells and the mechanism of regulation of hemin-induced ROS levels and erythroid differentiation by NAC and AA.
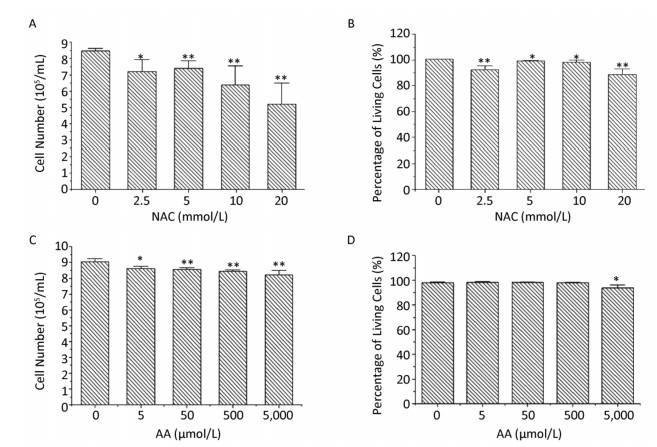
The proliferation and viability of K562 cells were investigated after direct exposure to various concentrations of NAC and AA for 24 h. As shown in Figure 1, the number of K562 cells decreased significantly in a concentration-dependent manner after exposure to 0, 2.5, 5, 10, and 20 mmol/L of NAC, and a similar trend was observed after exposure to 0, 5, 50, 500, and 5, 000 μmol/L of AA. After exposure to 20 mmol/L of NAC, there was 87.7% of viable cells, whereas > 90% of the total cells were viable after treatment with lower concentrations of NAC (0-10 mmol/L) and AA (0-5, 000 μmol/L). Therefore, NAC concentrations of 0, 2.5, 5, and 10 mmol/L and AA concentrations of 0, 50, 500, and 5, 000 μmol/L were used in subsequent experiments in this study.
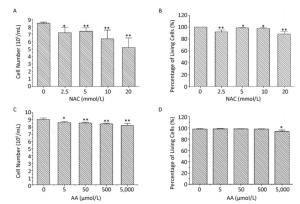
Figure 1. The proliferation and viability of NAC-and AA-treated K562 cells. K562 cells were treated with 0-20 mmol/L of NAC (A, B) or 0-5, 000 μmol/L of AA for 24 h (C, D), and then the total and viable cells were counted using trypan blue exclusion assay. Data are representative of three independent experiments and shown as mean ± SD. *P < 0.05 and **P < 0.01 (vs. corresponding control group).
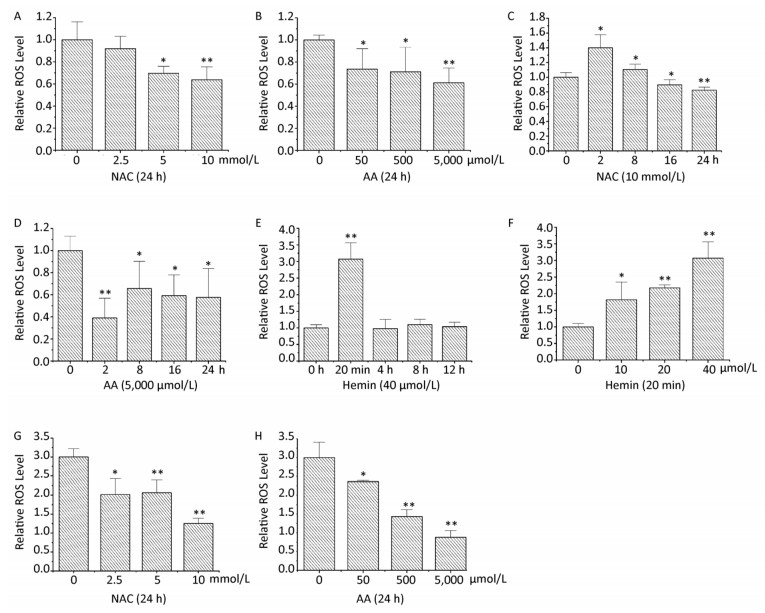
When K562 cells were treated with 0-10 mmol/L of NAC or 0-5, 000 μmol/L of AA for 24 h, the intracellular ROS levels were respectively decreased in a concentration-dependent manner (Figure 2A and 2B). Treatment with 10 mmol/L of NAC or 5, 000 μmol/L of AA for 24 h decreased the intracellular ROS levels to 64% and 61%, respectively. Intracellular ROS levels were increased when K562 cells were treated with 10 mmol/L of NAC for 2 h; however, there was a time-dependent decrease in ROS levels from 8 to 24 h, which were lower than the control levels after 16 h (Figure 2C). When K562 cells were treated with 5, 000 μmol/L of AA for 2-24 h, the intracellular ROS levels were significantly decreased and reached the lowest level when treated for 2 h (Figure 2D). It has been reported that acidosis induces ROS production in both MCF-7 and MDA-MB-231 cells, although the detailed mechanisms by which acidosis induces ROS production have not yet been determined[4]. Although the effect of NAC and AA on the pH of medium has not been determined, the acidity can be estimated by the color of culture medium after adding 10 mmol/L of NAC, whereas AA treatment did not change the medium pH obviously. The increase in ROS levels when K562 cells were treated with 10 mmol/L of NAC for 2 h may be due to the acidosis induced by NAC.
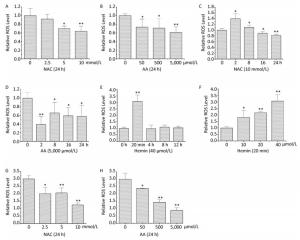
Figure 2. NAC and AA abolished hemin-induced ROS generation in K562 cells. After treating K562 cells with various concentrations of NAC, AA, and hemin for different periods of time, the intracellular ROS levels were analyzed by flow cytometry after staining with DCFH-DA. K562 cells were treated with 0-10 mmol/L of NAC for 24 h (A), 0-5, 000 μmol/L of AA for 24 h (B), 10 mmol/L of NAC for 0-24 h (C), 5, 000 μmol/L of AA for 0-24 h (D), 40 μmol/L of hemin for 0-12 h (E), and 0-40 μmol/L of hemin for 20 min (F). K562 cells were pretreated with 0-10 mmol/L of NAC (G) or 0-5, 000 μmol/L of AA (H) for 24 h and then treated with 40 μmol/L hemin for 20 min. Data are representative of three independent experiments and shown as mean ± SD. *P < 0.05 and **P < 0.01 (vs. corresponding control group).
When K562 cells were treated with 40 μmol/L of hemin, the intracellular ROS levels were transiently increased by 3.07-fold as early as 20 min. However, treatment with 40 μmol/L of hemin for 4, 8, and 12 h had no influence on the ROS levels (Figure 2E). Treatment with 0-40 μmol/L of hemin for 20 min resulted in a concentration-dependent increase in intracellular ROS levels in K562 cells, as expected (Figure 2F). This result is similar to that reported by studies showing that 20 μmol/L of hemin caused an approximately 12-fold increase over basal cellular ROS levels at 30 min in vascular smooth muscle cells, and that a significant increase in ROS levels was observed 1 h after the addition of 10 μmol/L of hemin in SH-SY5Y cells[5-6].
Furthermore, in this study, pretreatment with 0-10 mmol/L of NAC or 0-5, 000 μmol/L of AA for 24 h followed by exposure to hemin for 20 min resulted in a concentration-dependent decrease in the intracellular ROS levels, respectively (Figure 2G and 2H). Pretreatment with 10 mmol/L of NAC and 5, 000 μmol/L of AA for 24 h led to 42% and 29% decrease in the intracellular ROS levels, respectively, which were even lower than those observed with single NAC or AA treatment. These results demonstrated that NAC and AA could abolish hemin-induced ROS generation in K562 cells.
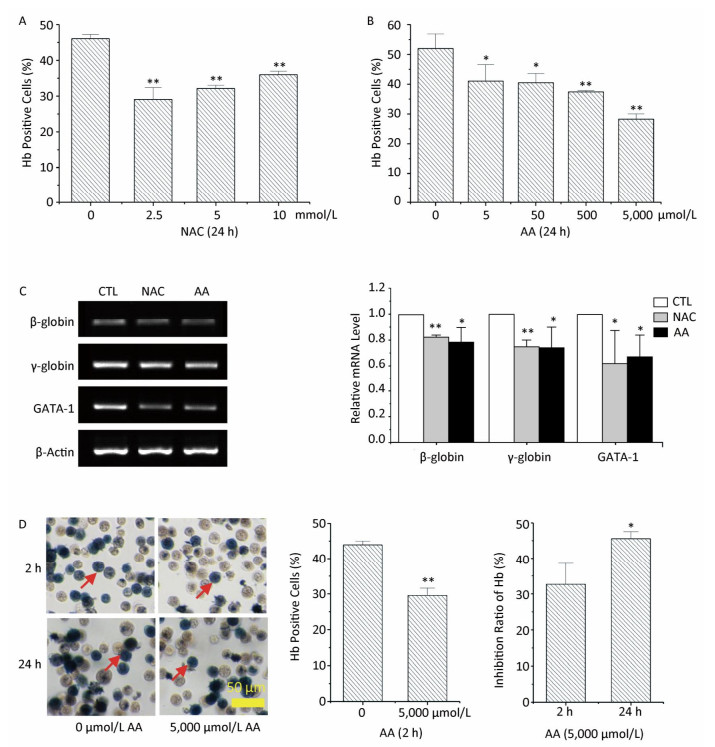
Hemoglobin (Hb) synthesis and mRNA levels of erythroid-specific genes were investigated in this study to determine the effects of NAC and AA on the erythroid differentiation potential of K562 cells. Pretreatment with NAC significantly decreased the percentages of Hb-positive cells, while pretreatment with AA resulted in a concentration-dependent decrease in the percentage of Hb-positive cells (Figure 3A and 3B). Pretreatment of K562 cells with 10 mmol/L of NAC or 5, 000 μmol/L of AA for 24 h resulted in a significant decrease in hemin-induced mRNA expression levels of β-globin, γ-globin, and GATA-1 genes (Figure 3C). These findings indicated that both NAC and AA inhibited the erythroid differentiation potential of K562 cells. However, the molecular mechanism involved in the downregulation of the expression of these erythroid-specific genes by NAC and AA remains largely unknown and requires further studies.
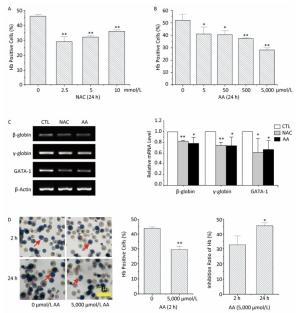
Figure 3. The effects of NAC and AA on the hemin-induced erythroid differentiation potential of K562 cells. After treating K562 cells with various concentrations of NAC or AA for the indicated time, NAC and AA were removed by replacing the culture medium, and hemin was added to induce erythroid differentiation for 48 h. Then, the percentage of Hb-positive cells was estimated by staining with benzidine. K562 cells were treated with 0-10 mmol/L of NAC for 24 h (A), 0-5, 000 μmol/L of AA for 24 h (B), and 0 or 5, 000 μmol/L of AA for 2 h (D). (C) Total RNA was extracted, and the mRNA levels of the indicated genes were analyzed using RT-PCR. CTL: control group. (D) Left panels show representative images of morphological analysis of benzidine staining captured under 20× magnification. The scale bar indicates 50 μm. Red arrows indicate Hb-positive cells. Right panels show the inhibition ratio of Hb synthesis in K562 cells treated with 5, 000 μmol/L of AA for 2 or 24 h. Data are representative of three independent experiments and shown as mean ± SD. *P < 0.05 and **P < 0.01 (vs. corresponding control group).
To sum up, treatment with 5, 000 μmol/L of AA for 2 h resulted in the lowest intracellular ROS levels. To determine the relationship between ROS levels and erythroid differentiation, K562 cells were treated with 5, 000 μmol/L of AA for 2 h, followed by the addition of hemin to induce erythroid differentiation for 48 h. This resulted in 43.93% and 29.54% of Hb-positive cells upon pretreatment with 0 and 5, 000 μmol/L of AA for 2 h, respectively (Figure 3D). The inhibition ratio of Hb synthesis was 33% when K562 cells were pretreated with 5, 000 μmol/L of AA for 2 h, which was considerably lower than 46% when the cells were pretreated with 5, 000 μmol/L of AA for 24 h. This result could suggest that ROS production was involved in the inhibition of erythroid differentiation. An earlier study has shown that thalidomide induces the generation of ROS in erythroid progenitor cells, which may activate p38 MAPK and acetylate histone H4, thereby leading to subsequent γ-globin gene expression[7]. Other studies have shown that increased ROS levels during the early stage of erythroblast development sensitize them to differentiate into mature blood cells[8]. However, we could not detect additional antioxidants to make the results more convincing.
As an antioxidant possessing a free radical scavenger property, NAC has been believed to be a safe and well-tolerated supplementary drug. Numerous dietary components such as vitamin C are believed to be involved in the antioxidant defense system and in the prevention of chronic diseases, particularly cancer and cardiovascular diseases. However, we found that NAC and AA reversed hemin-induced erythroid differentiation. Though the differentiation pathway of erythroid cells is still a matter of debate, erythroid cells at the terminal stages of differentiation shed their nucleus, endoplasmic reticulum, and mitochondria and as a result lose their ability to proliferate[9]. Terminal differentiation involves three to five additional cell divisions and accumulates important proteins required for Hb synthesis, chromatin condensation, and enucleation[10]. Whether the widely used NAC and AA exert toxic effects on the hematological system and the detailed molecular mechanism underlying this process remain largely unknown and require further investigation in our future studies.
In conclusion, we have demonstrated a novel role for NAC and AA in hemin-induced erythroid differentiation, which may be mediated by ROS. Therefore, modulating the intracellular ROS levels through antioxidants might be able to regulate the erythroid differentiation status; however, further studies are still required.
HTML
Natural Science Foundation of China 81072325
Natural Science Foundation of China 81573192







 Quick Links
Quick Links
 DownLoad:
DownLoad: