-
Lead, a heavy metal, which is nonessential but may be harmful to the human body, has been widely used to manufacture many products for use in the modern world. Lead-acid batteries have the advantages of low price, high safety, and advanced technology. Presently, our new energy policy is that electricity should replace fuels as an energy source. The use of lead-acid batteries continues to grow because of the development of electric vehicles and advancements in the automotive industry, meaning that lead-acid battery-producing companies still have a prominent position in China. However, the extensive use of lead usually leads to a series of health problems, such as damage to the hematopoietic system, nervous system, digestive system, urinary system, and reproductive system. Besides, lead emission by lead-acid battery factories into the air is considered a health hazard to residents of nearby communities[1]. In China, the Forum of National Occupational Disease reported that lead and its compounds were the major chemicals that were responsible for chronic occupational poisoning in 2015 and 2016. Thus, lead exposure is a very serious health hazard.
However, the toxic effects of lead and the clinical manifestations of lead-poisoning vary in different individuals. There are some differences in the biological markers and clinical symptoms among individuals who have been exposed to similar levels of lead. A previous study reported[2] that there was a significant association between the ALAD rs818708 polymorphism and the risk of lead poisoning. Tekin D et al.[3] reported that pregnant women with metallothionein 2A heterozygous AG genotype had higher BLLs than those with the AA homozygous genotype. These results indicated that, among lead-exposed workers, heterozygous genotype individuals are more susceptible to elevated BLLs than homozygous genotype individuals.
Long non-coding RNA (lncRNAs) are non-coding RNA (ncRNAs) with a length of more than 200 nucleotides. MALAT1, a lncRNA consisting of more than 8700 nt and located on chromosome 11q13, is localized to nuclear speckles, which contain a large number of nuclear proteins involved in mRNA splicing and transport. It has been shown that MALAT1 plays an important role in cancer and acts as a transcriptional regulator for numerous genes, including those involved in cell proliferation, migration, and metastasis[4]. The relationship between MALAT1 BLLs is unclear. Some studies have reported that lead could activate the MAPK signaling pathway to promote NGF-induced neurite outgrowth[5], indicating that lead can damage the nervous system through the MAPK signaling pathway. Moreover, Chen L found that the MAPK signaling pathway was inhibited in MALAT1-deficient N2a cells[6], suggesting a possible relationship between MALAT1 and BLLs. Thus, our study intended to further explore the relationship between lncRNA MALAT1 polymorphisms (rs3200401, rs619586, and rs11227209) and BLLs in lead-exposed workers.
Study Subjects All the 1, 213 participants were lead-exposed workers mainly engaged in scrubber board, slice post, and tablet post. They were from five different battery factories, which were located in the Northern part of the Jiangsu Province, far from the cities and towns (at least 10 kilometers away), and no other factory has been sited within a 5-kilometer radius from the cities since January 2004. The employees were enrolled from the relatively nearby designated towns, with similar lifestyles and not living nearby the factories. These participants experienced a similar external lead exposure dose (CTWA = 0.025 ± 0.009 mg/m3) during work. During the clinical evaluation, we excluded participants with a history of a hematological disorder, liver dysfunction, or kidney dysfunction, and participants who were on a lead-containing medical therapy were also excluded. All of them were informed about the details of this research and provided written informed consent before blood collection. Eighty-three participants were excluded from the study for not providing blood samples, or for unavoidable reasons. Eventually, 1, 130 participants met our inclusion criteria.
Blood Lead Determination Approximately 2 mL of blood was sufficient for the measurement of the BLLs. BLLs were determined using graphite furnace atomic absorption spectrometry.
DNA Extraction The blood samples to be used for DNA extraction and genotyping were collected in tubes containing ethylenediaminetetraacetic acid (EDTA). Generally, 200 μL of blood was sufficient for the DNA extraction. DNA was extracted using a TIANamp Blood DNA kit (Tiangen, Nanjing, China). The enzyme-linked immunoassay was used to measure the DNA concentration. The extracted DNA and the remaining blood samples were stored at -80 ℃ for later use.
SNP Selection MALAT1 SNPs were selected based on literature review and gene analysis using the Haploview 4.2 software. The following criteria were used to select the potential SNPs: 1) candidate SNPs should be located within the functional region of MALAT1, 2) minor allele frequency (MAF) for the selected SNPs should be greater than 0.05 in the Chinese Han Beijing (CHB) population, and 3) linkage disequilibrium value of r2 should be less than 0.8 for candidate SNPs. Ultimately, three candidate SNPs of MALAT1 met these criteria: rs11227209, rs3200401, and rs619586.
Polymorphism Genotyping Analysis Genotyping of the rs3200401, rs619586, and rs11227209 polymorphisms of MALAT1 was performed using ABI TaqMan SNP genotyping assays (Applied Biosystems, Foster City, CA, USA). Each reaction system for the genotyping contained 1.25 μL of distilled water, 2.625 μL PCR master mix (Roche, Branchburg, N.J., USA), 0.25 μL forward primer, 0.25 μL reverse primer, 0.125 μL FAM, 0.125 μL HEX (Applied Ji-ao Biosystems, Nanjing City, Jiangsu, China) and 0.5 μL of DNA. The reaction mixture was mixed and sequentially transferred onto 384 plates according to the manufacturer's instructions. The data for the genotyping were analyzed using the ABI 7900 system sequence detection software version 1.2.3 (Applied Biosystems).
Expression Quantitative Trait Loci We used the eQTL Browser of the University of Chicago for the expression quantitative trait loci (eQTLs) analysis.
Statistical Analysis x±s was used to describe the normal distribution of BLLs. We performed multiple linear regression to assess the correlation between MALAT1 polymorphism genotypes and BLLs.
The general characteristics, including sex, age, marriage, education, smoking status, drinking status, and BLLs of the 1, 130 participants are shown in Table 1.
Characteristics of Participants n(%) Sex Male 599 (53.0) Female 531 (47.0) Age (years) (20, 30) 83 (7.4) (30, 40) 275 (24.3) (40, 50) 619 (54.8) (50, 60) 136 (12.0) (60, 70) 17 (1.5) Marriage Single 3 (0.2) Married 1, 113 (98.5) Divorced 14 (1.3) Education Illiterate 67 (5.9) Literate, up to lower secondary level 158 (14.0) High, up to middle secondary level 676 (59.8) Higher, secondary level and above 229 (20.3) Smoking status No 829 (73.4) Yes 301 (26.6) Drinking status No 817 (72.3) Yes 313 (27.7) BLL (μg/L) Mean ± SD 386.73 ± 177.93 (17-1, 060) Note. BLL = blood lead level. Table 1. Demographic Characteristics and BLLs of Participants (N = 1, 130)
The results for the association between MALAT1 polymorphism genotypes and BLLs in the participants are summarized in Table 2. The MAFs of the MALAT1 SNPs, rs11227209, rs3200401, and rs619586, were 0.10, 0.17, and 0.11, respectively. The pooled regression coefficient (β) between the MALAT1 SNPs (rs11227209, rs3200401, and rs619586) and the BLLs were 0.000 for rs11227209, 0.084 for rs3200401, and 0.031 for rs619586. rs3200401 was significantly associated with BLLs (P = 0.004), whereas the other two SNPs did not show any significant association with BLLs (P > 0.05).
SNPs Genotypes Numbers MAF βa Plineara rs11227209 CC/CG/GG 1, 045/142/2 0.10 0.000 0.988 rs3200401 CC/CT/TT 1, 107/65/17 0.17 0.084 0.004 rs619586 AA/AG/GG 780/399/10 0.11 0.031 0.286 Note. The data were analyzed under a dominant genetic model and adjusted for age, sex, marriage, education status, smoking status, drinking status, and eating habits. Table 2. The genotypes of MALAT1 polymorphisms and BLLs in participants
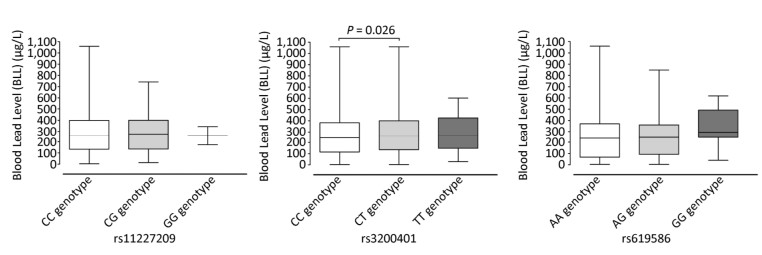
The results of the association between the genotypes and the BLLs are shown in Figure 1. When the BLLs were compared in the different genotypes of MALAT1 SNPs (rs11227209, rs3200401, and rs619586), there was no difference in the mean BLL for the rs11227209 and rs619586 polymorphism genotypes. However, for the rs3200401 polymorphism, there was a difference in the BLLs of subjects with homozygous CC genotype and heterozygous CT genotype (P = 0.026). The mean BLL for the heterozygous CT genotype was relatively higher than that of the homozygous CC genotype. There were no significances between the BLLs and the other two genotypes.

Figure 1. Joint effect of the SNPs on BLLs in each of the three cohorts. Lines indicate the means of the BLLs. Each box represents different amounts of alleles. P values were calculated by trend test.
In this study, we performed a genetic association analysis between the three MALAT1 SNPs (rs11227209, rs3200401, and rs619586) and BLLs in lead-exposed workers. The results suggested that the rs3200401 SNP may critically influence BLLs. In particular, the mean BLL of subjects with heterozygous CT genotype may be higher than that of subjects with homozygous CC genotype. There was a significant correlation between the rs3200401 SNP and BLLs (β = 0.084, P < 0.05). Further analysis revealed that the mean BLL of subjects with heterozygous CT genotype was higher than that of subjects with homozygous CC genotype (P < 0.05). This indicates that MALAT1 polymorphisms may be potential biomarkers for the risk of susceptibility to lead exposure. So far, this is the first study to report the association between BLLs and MALAT1 polymorphism genotypes.
Several researches have been conducted on the association between gene polymorphism and disease. Zhuo Y et al.[7] found that the rs619586 SNP of lncRNA MALAT1 was significantly associated with PAH risk. However, the mechanism underlying the effect of MALAT1 rs3200401 polymorphism on BLLs has not yet been fully elucidated. Chen L et al.[6]found that inhibition MALAT1 expression led to a significant suppression of the mitogen-activated protein kinase (MAPK) signaling pathway and activation of the P53 signaling pathway in N2a cells. MALAT1 played a critical role in the initial stage of neuronal differentiation through the aforementioned signaling pathways. Regarding researches on lead and the two signaling pathways, MAPK and p53, Williams TM proved that lead could promote NGF-induced neurite outgrowth by activating the MAPK signaling pathway[5]. Moreover, Xu J suggested that lead could induce p53 expression in mice PC 12 cells[8]. p53 plays an important role in lead-induced apoptosis in PC 12 cells[9]. Our study suggested that the mean BLL of subjects with heterozygous CT genotype may be higher. Therefore, we speculated that the heterozygous CT genotype of MALAT1 rs3200401 polymorphism may have an effect on BLLs through the MAPK signaling pathway or the P53 signaling pathway. Further experiments are needed to prove this.
Some studies found that gene polymorphisms may contribute to susceptibility to lead poisoning[10]. However, these studies were biased case-control studies, given that case-control studies employ few data points. Consequently, the findings of these studies cannot offer any useful information regarding individuals whose BLLs do not meet the diagnostic criteria. Conversely, our study mainly explored the relationship between BLL and gene polymorphisms by multiple linear regression analysis. BLL (value) is still a quantitative variable, which can reflect the differences between different subjects. Therefore, it is useful to a large extent.
In conclusion, our study provides a new evidence that there may be a significant association between MALAT1 and BLLs in Chinese populations. Subjects with MALAT1 heterozygous CT genotype were susceptible to lead exposure. Further studies need to be conducted to elucidate the mechanism underlying the effect of MALAT1 polymorphisms on BLLs.
HTML
the Jiangsu Province Special Project of Clinical Science and Technology BL2014082
the Jiangsu Provincial Medical Innovation Team CXTDA2017029
the Jiangsu Provincial Medical Youth Talent QNRC2016548







 Quick Links
Quick Links
 DownLoad:
DownLoad: