-
Truttmann MC et al.[1] recently reported that AMPylation of heat shock protein 70 (HSP70) family of chaperones participates in altering the aggregation properties and maintaining protein homeostasis (proteostasis), thereby playing a vital role in the development of neurodegenerative diseases (NDs). NDs are commonly manifested by protein aggregates, which exert harmful effects on proteostasis. Interestingly, it has been observed that AMPylation of heat shock proteins (HSPs) can maintain proteostasis by inhibiting the formation of protein aggregates. As previous studies only indicate that HSPs could regulate proteostasis, such a novel discovery further demonstrates the involvement of HSP70 AMPylation in the regulation of protein aggregation and the maintenance of proteostasis. Therefore, AMPylation can be considered to possess a therapeutic potential to target certain physiological processes related to proteostasis, such as age-related diseases.
AMPylation (also known as protein adenylylation), is a novel post-translational modification in prokaryotes and eukaryotes. AMPylation involves the covalent attachment of an adenosine monophosphate (AMP) group to a hydroxyl side chain of a protein. As the name indicates, a single ATP molecule is required for the process[2]. AMPylation frequently occurs reversibly in the hydroxyl group of threonine, serine, or tyrosine through a phosphodiester bond. Recent studies have demonstrated that AMPylation-modified proteins are involved in several important cellular functions such as proteostasis and innate immunity. This indicates the role of AMPylation in diverse processes with pleiotropic actions. Consequently, perturbations in AMPylation play a fundamental role in some pathological conditions.
In recent years, accumulating evidence has indicated the role of AMPylation in different pathophysiological processes. In a previous study by the authors, they demonstrated that Fic domain-containing proteins (Fic proteins) Fic-1, which can improve resistance toward pathogens in Caenorhabditis elegans, could be a vital AMPylator of heat shock 70 family proteins, histones H2 and H3, and translation elongation factor activity. Furthermore, innate immunity in C. elegans is relevant to FIC-1-mediated target AMPylation. Pseudomonas aeruginosa infection can trigger an innate immune response, thereby contributing to the release of reactive oxygen species (ROS), which subsequently lead to DNA damage and protein unfolding. Particularly, heat shock factor 1 (HSF-1) has been suggested to participate in modulating innate immunity in C. elegans. Moreover, HSF-1 can modulate the expression of HSPs to promote the refolding of unfolded proteins induced by the release of ROS and thus fight against bacterial infection. Similarly, AMPylation of eukaryotic elongation factor 1-alpha 2 (eEF-1A2) triggers the unfolded protein response (UPR), thus decreasing the translation level and enhancing the specific expression of activating transcription factor-4 (ATF-4), a molecule associated with the regulation of the transcription of UPR-related genes. AMPylation of histones may inhibit the transcription of infection-related genes and assist the repair of DNA damage triggered by ROS[3] (Figure 1). Therefore, modulation of HSPs, histones, and elongation factors by FIC-1 provides a crucial insight into the role of AMPylation in the regulation of innate immunity and cellular homeostasis in C. elegans.
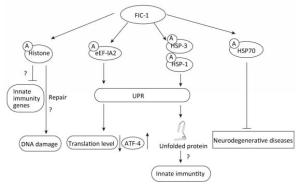
Figure 1. FIC-1-mediated target proteins AMPylation exerts different effects in Caenorhabditis elegans.
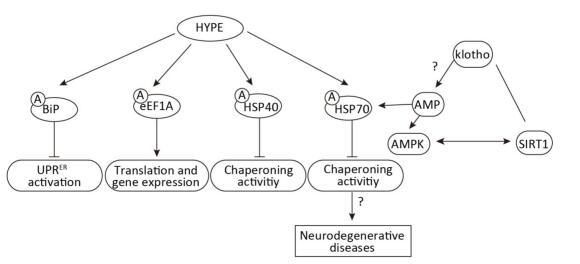
Then, what is the role of AMPylase in humans? Until 2009, huntingtin yeast-interacting protein E (HYPE) was confirmed as an AMPylase. Of note, similar to the bacterial AMPylase, GS-ATase possesses the ability of AMPylation and deAMPylation. HYPE, as a bifunctional enzyme, can also AMPylate and deAMPylate the major endoplasmic reticulum (ER) chaperone-binding immunoglobulin protein (BiP)[4]. Furthermore, Glu234 in HYPE is essential for the process of deAMPylation: interplays between the side chains of Glu234 and Arg371 sterically hinder proper alignment of ATP in the active site, thereby impeding HYPE from target modifications. While the variability of Glu234 side chain allows HYPE to contain ADP in its catalytic cleft, hence supporting target deAMPylation. Therefore, HYPE is primarily considered to be a deAMPylase. Moreover, it has been clearly confirmed that a variety of target proteins are AMPylated by HYPE, such as BiP, HSP70, eEF1A, and HSP40. Meanwhile, the AMPylation sites of some proteins have been discovered. The ER-resident HSP70 family protein Grp78/BiP is AMPylated on Ser365/Thr366 or Thr518, and unlike BiP, the AMPylation site of eEF1A is Thr261. Hence, the results of AMPylation of these proteins are different. BiP, as the major ER chaperone, can restrain UPR activation in the ER by AMPylating Thr518, whereas AMPylation of HSP40 and HSP70 can negatively regulate chaperoning activities[5]. Moreover, HYPE-mediated HSP70 AMPylation can disrupt proteostasis and induce HSF-1-dependent heat shock response in humans[6]. AMPylation of eEF1A at Thr261 modulates translation and gene expression[7]. Interestingly, age-related genes, klotho and sirtuin 1 (SIRT-1), have been demonstrated to be associated with AMP-activated protein kinase (AMPK). Loss of klotho inhibits not only SIRT-1 expression but also AMPKα activity[8]. However, the correlation between AMPK and SIRT-1 remains controversial[9, 10]. Then, based on the abovementioned facts, how does klotho regulate AMPKα expression? Thus, it has been hypothesized that klotho may regulate AMPK by affecting the AMP level, and because AMPylation is a process of adding AMP to proteins, klotho might modulate HSP70 AMPylation, eventually leading to the development of age-related diseases (Figure 2). Altogether, these findings indicate that AMPylation of diverse HYPE target proteins can affect proteostasis and cellular homeostasis and inhibit ER stress. Remarkably, klotho partially correlates with AMPylation, thereby participating in the development of age-related diseases.
Till date, although various modifications have been reported to participate in the regulation of neurodegenerative diseases, a study on HSP70-targeted AMPylation has not yet been conducted. In detail, the expression of hyper-AMPylation [FIC-1(E274G)] favors the formation of amyloid-β (Aβ) and α-synuclein (α-syn) aggregates and attenuates the toxicity of Aβ and α-syn. However, overexpression of FIC-1(E274G) reduces worm survival and influences larval development. In addition, AMPylase-deficient fic-1(n5823) increases the sensitivity to aggregation-prone polyglutamine (polyQ) proteins, promotes the generation of small aggregates, and inhibits the formation of larger aggregates.
More specifically, although protein aggregates have been proposed to be the major culprit in NDs, there is a wide and different discrepancy between different types and sizes of aggregates. These oligomers, soluble aggregates, have been reported to be the primary toxic aggregates. Conversely, the toxicity of larger, insoluble aggregates is lower[11]. Moreover, these aggregation-prone proteins that can cause disease are initially in the form of monomers and then gradually convert into intermediate, soluble aggregates. In fact, it has been suggested that HSPs possess the ability to prevent the formation of protein oligomers. Furthermore, HSPs inhibit the oligomerization of mutant huntingtin (mHtt), Aβ, and α-syn and attenuate the cytotoxicity of these proteins by avoiding the conversion of monomers into oligomers, promoting the degradation of oligomers through the ubiquitin–proteasome and autophagy-lysosomal pathways, or accelerating oligomer deposition into large, insoluble aggregates[12]. In particular, under pathological conditions, HSPs are excessively required, which results in an inadequate supply to cope with protein aggregates. Under such situation, limitation of HSP70 activity by AMPylation is beneficial to survival, as such activity-limited HSP70 can support the formation of large aggregates from another level. Therefore, this study has revealed a novel perspective to mitigate aggregation-related toxicity. Both HSP activators and inhibitors (AMPylation) enable the protection of cells from toxic aggregates, which is due to the fact that activators can prevent the formation of aggregates, whereas inhibitors can promote the formation of large, cytoprotective aggregates.
In general, although AMPylation is a post- translational mechanism of protein modification that has been proposed decades earlier, it has become increasingly important as its functions are being gradually recognized. As mentioned earlier, AMPylation participates in different pathophysiological processes such as cellular homeostasis and innate immunity. More remarkably, interactions between HSP expression level and AMPylation promote the development of NDs, such as Alzheimer's disease (AD), Huntington's disease (HD), and Parkinson's disease (PD).
Therefore, a further study is required to understand the role of HSP70 AMPylation site in the development of NDs or to assess whether there is another protein that can be AMPylated and has an effect on NDs. Furthermore, drugs targeting the human AMPylase HYPE and enhancing the activity of HYPE may provide an attractive therapeutic strategy to suppress the adverse effects observed in NDs.
HTML
the grants from the National Natural Science Foundation of China 81470434







 Quick Links
Quick Links
 DownLoad:
DownLoad: