HTML
-
Liver is not only the main organ of glycogen synthesis and storage, gluconeogenesis, and glycogenolysis[1, 2] but also the main target organ of insulin action, maintaining the production and output of endogenous glucose[3]. Liver is also the largest organ in early embryonic development[4]; it requires abundant blood supply and large amounts of oxygen supply, additionally exhibits a high speed of growth and metabolism, and is extremely sensitive to hypoxia. In fetus, the 'saving' growth pattern is triggered, which shown as firstly maintain adequate oxygen in the heart, brain, kidney, and other important organs, and decreasing the oxygen supply to the liver, which is also a consequence resulting from hypoxia[5]. As a consequence, the oxygen saturation of fetal liver will decline, leading to significant liver inflammation associated with functional retardation[6]. Therefore, fetal liver growth and development are retarded owing to blood supply restriction, which causes a unique metabolic response mode that results in abnormal metabolism from birth[7-9].
Primary hepatic culture models are increasingly needed to study liver metabolism, secretion, and regeneration medicine. Studies have successfully isolated rat hepatocytes for primary cultures, allowing for a variety of metabolic studies[10, 11]. However, in rats and other small animals, a small size may limit attempts to correlate in vitro data with in vivo studies of the fetus, particularly during early gestation. The chronically catheterized fetal lamb is a well-described in vivo model that has been proven useful in the study of fetal metabolism over a wide range of gestational ages[12, 13]. Our previous study with a fetal lamb indicated that this animal is an ideal model to conduct in vivo and in vitro experiments with satisfying results[14]. Therefore, modifying the current methods of isolating hepatic cells from fetal lamb is crucial to the fields of liver metabolism, pathophysiology, and regeneration medicine.
Hypoxia exerts multiple adverse effects on different organs, such as lung, heart, pancreas, and kidney. This condition reduces fatty acid synthesis and glucose metabolism and increases systemic inflammation[9]. Hypoxia during pregnancy shows a strong impact on the intrauterine environment for fetal growth, causing delayed proliferation and cell apoptosis in fetal hepatocytes[15, 16]. Our previous studies have shown that prenatal hypoxia, which is a kind of intrauterine growth restriction, could significantly affect the development of fetal liver, and the effect can be extended to postnatal and later life[9]. Numerous research results have indicated alterations of the liver functions, such as nonalcoholic fatty liver disease, under hypoxia in animal experiments[9, 17]. However, a variety of environmental factors, such as diet, different developmental stages, and treatment conditions, may affect the outcomes. The primary culture of hepatocytes can be used to avoid these confounding factors to reveal relatively definite factors, such as hypoxia degree, drug dosage, and duration of each intervening factor. Existing primary hepatocytes isolation methods[13, 18] had been demonstrated to harvest embryonic hepatocytes with similar yield and purity, but they require more intense surgical training and more procedural steps compared with our protocol. Therefore, we sought to develop a technique for the isolation and primary culture of fetal ovine hepatocytes to compare in vitro data with the main body of available in vivo sheep studies. Furthermore, the effect of hypoxia on the primary cultured hepatocytes, which were isolated from fetal liver with our modified protocol, was examined.
-
The pregnant ewes (47 ± 3 kg) were housed indoors under controlled conditions at 22 ± 2 ℃, 55% ± 5% humidity, and acclimated to 12 h light and dark cycles. All experimental animals were given free access to water and food. All the experimental procedures followed the guidelines of the National Institutes of Health Guide for the Care and Use of Laboratory Animals in China and approved by the Animal Care Service at Soochow University.
-
At about (128 ± 3) gestation days, the pregnant ewes were anesthetized, and the fetus was delivered by cesarean section. The time interval from the fetus delivery to hepatocyte isolation was controlled within 2 h. According to our recent studies[13], a midline abdominal incision was made to expose the liver. The common bile duct was ligated, and heparin (100 U/mL, MCE, 9041-08-1) was injected into the exposed abdominal aorta. Then, heparin was rapidly injected into the umbilical vein to perfuse the open heart. The liver was carefully dissected following hepatic pedicle ligation and then transferred to a sterile plate. The Leffert's buffer containing heparin (100 U/mL) was injected from the umbilical vein catheter to rinse residual blood in the liver. After the liquid clarified, EGTA solution (1 mmol/L, pre-warmed at 37 ℃) (LMAI Bio, LM25-01780) was used for circulating perfusion through the above umbilical vein catheter. The fetal liver gradually turned pale, and we switched to a perfusion solution containing collagenase Ⅳ (Yeasen, 40510ES60; 1 g/L) with calcium ion pre-warmed at 37 ℃ for another 15 min. Then, the liver full of collagenase was placed in vibration water bath at 37 ℃ for vibration and digestion for 20-30 min. The digested liver was mechanically dissected to remove the fiber block, connective tissue, and liver capsule. Then, 30 mL Dulbecco's Modified Eagle Medium (DMEM) (HyClone, sh30022.02b) containing 10% fetal bovine serum (FBS) (Gibco, 10091148) was added to terminate digestion. The liquid was filtered by a 100-mesh cell strainer (BD, 352340) and centrifuged at the rate of 800 rpm/min for 5 min. The supernatant was discarded, and the pellets were washed thrice with Leffer buffer. The cell pellets were re-suspended in 10 mL Leffer buffer.
-
Exactly 2 mL of the above cell suspension was acquired and then mixed with 5 mL Percoll (sigma, p7828) separating medium Ⅱ and centrifuged at the rate of 8, 000 rpm/min for 30 min at 4 ℃. Then, the pellet was washed thrice with Leffer buffer by centrifuging at the rate of 8, 000 rpm for 10 min at 4 ℃. Finally, the cell pellet was re-suspended in 5 mL culture DMEM-F12 containing 10% FBS. The cell viability was measured by Trypan blue (Solarbio, T8070) exclusion. The viability of hepatocytes over 90% was determined for experimental use. The cell density was adjusted to 2.0 × 105/mL when seeded into the culture flask. The flask was placed in an incubator at 37 ℃ with humidified atmosphere of 95% air and 5% CO2. After 24 h incubation, the flask was rinsed once with cold serum-free media, and fresh DMEM-F12 with 10% FBS was added.
-
The cells were seeded into 24-well culture plate at 1.0 × 105 per well and incubated for 24 h. Afterward, the wells were rinsed with cold serum-free medium to remove the floating cells, and the medium was replaced with fresh DMEM-F12 containing 10% FBS. The culture medium was changed every 3 days, and the cells were counted under an inverted microscope (Nikon, Ts2R). The cell growth curve was plotted with cell count every 24 h for a period of 12 days.
-
The glass coverslips were preset in the 24-well plates. The cells were seeded into each well following the above protocol. After 48 h incubation, the coverslips with cells were collected for experiments.
-
The cultured cells were fixed, oxidized, and rinsed. Then, Schiff agent (Solarbio, G1286-50) and drops of 2% methyl green (Solarbio, M8020-1) were added. The coverslips were washed and air-dried, dehydrated with gradient ethanol (Sigma-Aldrich, 32221-2.5L-GL), treated with xylene (TRC, X749400), and finally mounted with optical resin. The cell morphology was examined under optical microscope[19].
-
CK8 (Boster, BA3152), CK18 (Boster, BM0032), CK19 (Proteintech, Ag1085), and alpha-fetoprotein (AFP; Proteintech, Ag6089) were detected with immunocytochemistry to determine the purity degree of the harvested hepatocytes according to manufacturer's instructions. The results were examined under optical microscope[20].
-
The primary cultured hepatocytes were harvested after 72 h incubation and rinsed with phosphate-buffered saline (PBS), followed by 24 h fixation in 70% ethanol. The hepatocytes were rinsed with PBS and centrifuged. The cell pellet was re-suspended in 1 mL propidium iodide solution (50 µg/mL) and filtered with 300-mesh screen after 30 min incubation in the dark at room temperature. Then, the cell cycle and proliferative index (PI) was determined by FCM (BD FACSCalibur). PI = cells in (S+G2M) stage/cells in (G0/G1+S+G2M) stage × 100%.
-
The isolated hepatocytes were seeded into culture flasks and then placed in airtight 5% CO2 chambers containing either 2% or 21% O2 at 37 ℃ followed by 72 h culture. After exposure for 24, 48, and 72 h, the cells were harvested. To mimic hypoxia for the cultured cells, the oxygen depletion can range from 0% to less than 20%. The present study selected 2% oxygen as hypoxia treatment to simulate extreme hypoxic condition.
-
The treated cells were trypsinized, rinsed thrice with warm PBS (37 ℃), and then fixed with 2.5% glutaraldehyde in 0.1 mol/L PBS at 4 ℃ overnight. After washing with PBS, the cells were postfixed in 1% osmic acid, dehydrated in graded alcohols, embedded in Epon 812, sectioned, and stained with uranyl acetate/lead citrate and were observed with a TEM (Tokyo, Japan).
-
SPSS 13.0 software was used for statistical analysis using t-test or one-way analysis of variance. Each experiment was repeated three or more times. Data were represented with mean ± sx, and P < 0.05 indicated statistical significance.
Experimental Animals
Isolation of Fetal Ovine Hepatocytes
Purification and Cultivation of Parenchymal Hepatic Cells
Growth Curve Plotting of the Primary Cultured Hepatocytes
Preparation of Cell Attachment and Growth on Coverslip
Identification of Hepatocytes with Glycogen Staining
Identification of Hepatocytes by Immunocytochemistry
Identification of Cell Purity and Proliferation Activity by Flow Cytometry (FCM)
Establishment of Hypoxia Model for Hepatocytes in Primary Culture
Sample Preparation under Transmission Electron Microscope
Statistical Analysis
-
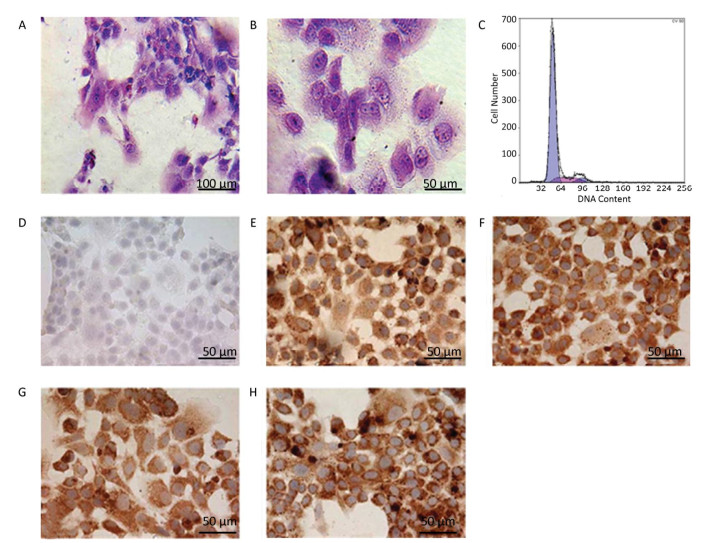
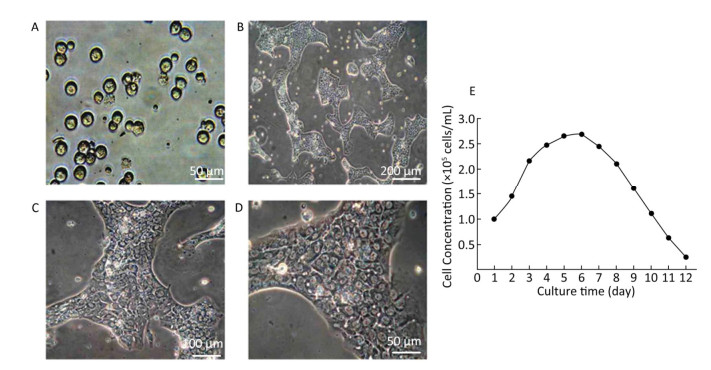
Growth of Hepatocytes in Primary Culture Observed under Inverted Microscope The freshly isolated hepatocytes appeared round and transparent with similar size and evenly spread without cell cluster (Figure 1A). After 24 h cultivation, 80%-90% of hepatocytes attached to the wall and displayed a polygonal form that is typical of epithelial cells. All the cells showed clear edges, rich cytoplasm, distinct karyon, and large nucleus. Some cells displayed a visible dikaryon with a high nucleo-cytoplasmic ratio, flat and thin soma, and significantly large volume. The cells connected to each other, forming an island-like shape. After 48 h cultivation, the hepatocytes became flattened and stretched, closely attaching to the wall of culture bottle with about 90% confluence. The cells were mixed together to connect and form an island-like piece. No other forms of adherent cells were observed under microscopic observation (Figure 1B-1D).
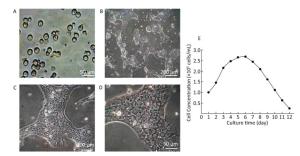
Figure 1. Parenchymal hepatic cell of fetal ovine in primary culture and its growth curve expression. A: Parenchymal hepatic cells for primary culture after fresh separation (400×), scale bar = 50 μm; B: Parenchymal hepatic cells after 48 h of culture (100×), scale bar = 200 μm; C: Parenchymal hepatic cells after 48 h of cultivation (200×), scale bar = 100 μm; D: Parenchymal hepatic cells after 48 h of cultivation (400×) scale bar = 50 μm. E: The growth curve was plotted according to daily cell counted in continuous record until day 12. Day 1: entered exponential phase, Day 5-6: reached the stationary phase.
Growth Curve of Hepatocytes in Primary Culture The fetal lamb hepatocytes entered the exponential phase after 24 h cultivation under standard culture condition, with cell doubling at about 60 h (2.5 days), and reached the stationary phase at 5-6 days. Gradually, the cultured cells entered the death phase with visible cell debris in the medium. The growth curve was plotted according to daily cell count in continuous record until day 12 (Figure 1E).
-
Identification of Hepatocytes in Primary Culture with Glycogen Staining In human or other animals, the hepatocytes play a vital and pivotal role in storage and production of glycogen for energy resource. Thus, the significant characteristic should be a rich supply of glycogen in the cells, as demonstrated in the figure below (Figure 2A-B). With periodic acid-Schiff method, the positive purple-red staining of glycogenosome was observed under microscope.
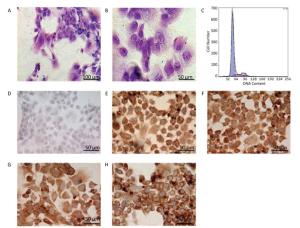
Figure 2. Identification of the parenchymal hepatic cells of fetal ovine in primary culture. (A)-(B): Glycogen staining of hepatocytes. (A) 200×, scale bar = 100 μm; (B) 400×, scale bar = 50 μm. The positively stained glycogenosome appeared purple-red, and the nucleus was blue under the microscope. (C) Results of detection of parenchymal hepatic cells of fetal ovine in primary culture after 72 h by FCM. (D)-(H) Immunocytochemistry identification of hepatocytes (400×), scale bar = 50 μm. (D) Negative control staining; (E) staining with first antibody of CK8; (F) staining with first antibody of CK18; (G) staining with first antibody of CK19; (H) staining with first antibody of AFP.
Identification of Hepatocytes by Immunocytochemistry Immunocytochemistry demonstrated that the isolated hepatocytes showed a positive reaction toward the primary antibodies against CK8, CK18, CK19, and AFP. Figure 2E-H show positive staining as brown particles in the cytoplasm compared with Figure 2D, which displays the negative control group with PBS administration. As AFP is an essential biomarker of hepatocytes, the chemical reaction toward this molecule was more intense. The positive cells were stained purple-red, whereas the cells showing a negative reaction, which were stained blue, were evaluated to determine whether the cell population purity was larger than 95%.
Characters and Purity of Hepatocytes in Primary Culture Identified by FCM The DNA contents were evaluated with FCM in primary cultured hepatocytes. At 72 h primary culture, as demonstrated in Figure 2C, the G1/G0 stage showed a sharp peak without any confounding peaks. The S phase fraction and PI were 12.316% and 17.394%, respectively, whereas the apoptosis ratio was 0.338% (Figure 2C).
-
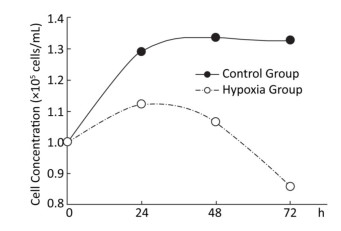
Effects of Hypoxia on the Growth of Hepatocytes in Primary Culture After molding, cells in the normal control group entered the platform stage after 24 h of proliferation stage. From 24 h to 72 h, the cell concentration showed a negligible change. However, during the first 24 h, cell concentration in the hypoxia group exhibited an increasing concentration, but the value was still less than that in the normal control group. From 24 h to 72 h, the cell concentration decreased, instead of increasing. At 72 h, a large amount of floating dead cells had been observed in the medium (Figure 3).
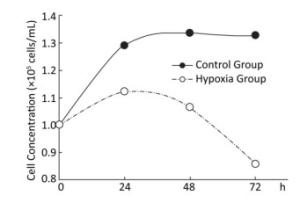
Figure 3. Effects of hypoxia on the growth of parenchymal hepatic cells of fetal ovine in primary culture.
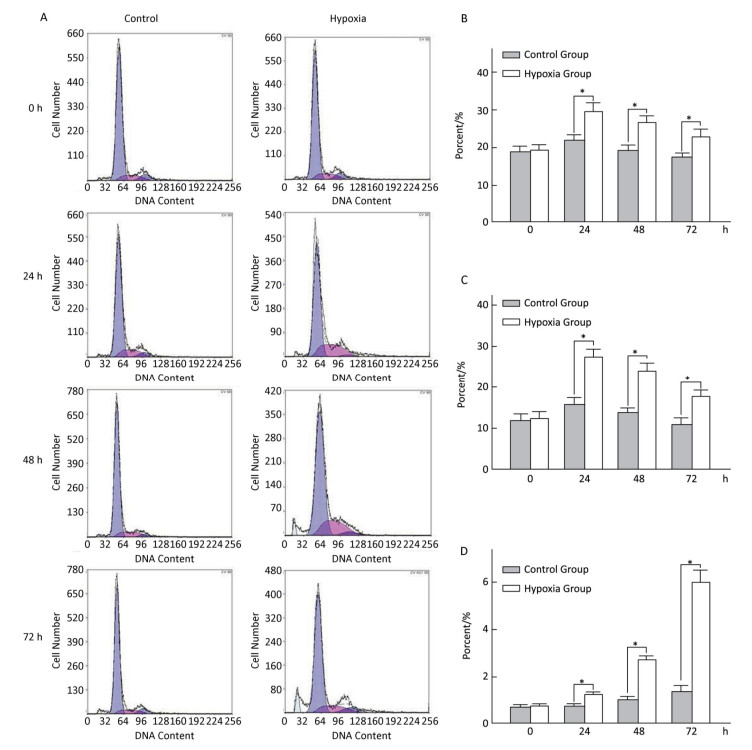
Effects of Hypoxia on the Cell-cycle of Hepatocytes in Primary Culture The cell ratio and PI in the S stage were the highest at 24 h, and their values gradually declined at 48 and 72 h under both hypoxia and normoxia. After 24, 48, and 72 h hypoxia exposure, the cell ratio and PI in the S stage in the hypoxia group were significantly higher than those in the normal control group at the same time point (P < 0.05). The apoptosis ratio gradually increased with hypoxia exposure in a time-dependent manner (Figure 4).
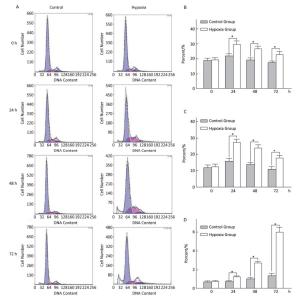
Figure 4. Effects of hypoxia on the period of parenchymal hepatic cells of fetal ovine in primary culture (n = 6). From 24 to 72 h after cell molding for cultivation, the cell ratio, proliferation index, and apoptosis ratio in stage S were detected both in hypoxia group and in control group. A-Control: Normal control group; A-Hypoxia: hypoxia group; B: Cell rate in stage S; C: Proliferation index; D: Apoptosis rate. *P < 0.05.
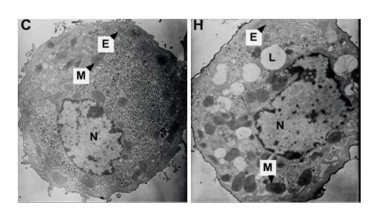
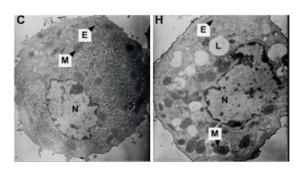
Effects of Hypoxia on the Ultrastructure of Hepatocytes in Primary Culture Under the standard normoxic condition, the hepatocytes showed normal-looking organelles, such as oval nucleus with evident nucleolus, mitochondria with rich cristae, lamellar shape of endoplasmic reticulum (ER), and glycogenosome particles, which were evenly distributed among the cytoplasm (Figure 5). However, the cells exposed to hypoxia demonstrated different swollen mitochondria with chaotic cristae arrangement, swelling ER, and multiple lipid droplets. An elevated amount of condensed electron matter was also observed within the cytoplasm (Figure 5).
Growth Characteristics of Hepatocytes in Primary Culture
Identification of Hepatocytes in Primary Culture
Effects of Hypoxia on Hepatocytes in Primary Culture
-
Primary culture has been successfully used in in vitro studies in cancer research, virology, pharmaceutical studies and genetic research and regeneration medicine as the primary cultured cells still represent the physiological characteristics of the original organ. An increasing number of studies focused on liver function and regeneration with primary cultured hepatic cells, and many researchers have developed methods to isolate the fresh hepatocytes for primary culture with good results[21-23]. However, developing an isolation method that can achieve a high cell yield with decent purity is still a huge challenge. As the liver cells differentiate faster than other cells, the hepatocytes may change their original characteristics after prolonged culture. Our present study results in number of hat our proposed isolation method is a relatively simple approach to harvest fresh fetal hepatocytes with a high yield reaching more than 1 × 109 cells per fetal liver; the purity is larger than 95%, with the majority of cells being parenchymal hepatic cells. One method to isolate the fetal hepatocytes[13] is by in situ perfusion, and collagenase digestion using occlusion of the ductus venosus obtains good cell yield and purity; Liu's laboratory[18] applied two-step collagenase perfusion and also achieved good results. These isolation methods for hepatocytes require well-trained surgical techniques and consume considerable time. Our modified cell isolation protocol is relatively straightforward and less time consuming but achieves similar yield and cell purity. Under microscopic observation, the freshly isolated cells maintained their original morphological characteristics, such as clear edge polygonal form, rich cytoplasm, and large nucleus. The hepatocytes are the main storage location of glycogen in the body[1-3]; thus, we applied periodic acid-Schiff to confirm this cell feature (Figure 2). The microscopic image showed that almost all the cells exhibited purple-red staining in the cytoplasm, indicating the presence of glycogen. To further test the purity of isolated hepatocytes, immunocytochemistry was conducted to reveal cytokeratin CK8, CK18, CK19, and AFP. The results indicated that the cell population purity is larger than 95%. AFP staining was more intense among these markers, thus fitting the concept of high AFP expression[24] at the fetal stage of hepatocytes. The FCM (Figure 2) result also further supports that the isolated hepatocytes exhibited high purity, as demonstrated by the sharp peak (G1/G0).
Cell culture is a good research means to correlate the in vitro studies with in vivo research, which has been used broadly. Thus, the ideal condition is using cultured cells with the original characteristics. As the hepatocytes grow and differentiate faster than any other cells, for any research purpose, the original cell characteristics of the freshly isolated cells must be retained, and the best time period for treatment must be revealed. The cell growth curve of the present study showed the primary hepatocytes started exponential growth phase from 24 h culture, and the cell number doubled at about 2-3 days period and then reached the platform at about 6 days (Figure 1). The current data recommend that the reasonable experiment treatment time is around 72 h culture time. Therefore, evaluation of the hypoxia effect on primary hepatocytes was conducted at 72 h.
A literature search revealed that the adverse intrauterine growth restriction (IUGR) during maternal pregnancy may increase the susceptibility of offspring to develop insulin resistance, DM, nonalcoholic fatty liver disease, and hypertension during adulthood[25, 26]. The main risk factor causing IUGR is prenatal hypoxia[9, 12], which is caused by placental insufficiency and preeclampsia. Consequently, the offspring obtained through IUGR features higher risks of suffering from adult obesity and other metabolic syndromes. Animal studies indicated that prenatal hypoxia increases an offspring's risk to develop metabolic syndromes and insulin resistance and hypertension in adulthood[15, 16, 27, 28]. Oxygen and nutrition are supplied through the circulation from placenta to fetus; thus, any blood flow interruption to the fetus will cause fetal development delay, lower birth weight, and retardation of organ formation[29]. An in vitro study demonstrated that hypoxia–reoxygenation injury can cause human liver endothelial cell death through apoptotic mechanisms[30]. Our current study also demonstrated the effect of hypoxia on cell growth. The experimental fetal hepatocytes showed a tendency to proliferate in the first 24 h with 2% oxygen incubation, which is significantly lower than that of the control group. Then, the cell death rate of the experimental group increased with time, whereas the control group continually grew up to 72 h. This study further provided evidence supporting that hypoxia causes hepatocyte death with apoptotic mechanism (Figure 4). At each time point, a significant difference (P < 0.05) was observed in the cellular cycle phase percentage of G1/G0, proliferation rate, and apoptosis percentage between the hypoxia and normoxia groups. Furthermore, the ultrastructure change under hypoxia showed the mitochondria and ER swelling and accumulation of lipid droplets. This cellular structure change may further provide evidence that prenatal hypoxia predisposes the offspring under prenatal hypoxia to develop nonalcoholic fatty liver.
Cellular injury may be caused by genotoxic damage that is either extrinsic, such as radiation, drugs, and hypoxia, or intrinsic, including DNA replication and repair errors. In most circumstances, the damage can be repaired by a cell's own protective mechanism. However, if the damaged cells cannot be repaired after prolonged exposure of the adverse insult, the cells may initiate apoptosis or senescence[31]. Cellular senescence is a fundamental complex mechanism with an important protective role from embryogenesis to late life across all species. The level at which cellular damage or injury occurs when cells go to different directions of apoptosis or senescence remains unclear[32]. Several studies demonstrated that cellular senescence plays a crucial role in aging of both individual organs and entire organism[33]; thus, senescence is a very important protective mechanism during embryonic development under IUGR including hypoxia. Our present study mainly focused on the effect of hypoxia on cellular growth, apoptosis, and cellular ultrastructural changes. Our future interest will examine senescence in the primary hepatic culture models under hypoxia and their effect on the offspring under gestational hypoxia exposure. In summary, we have modified the protocol in our previous study[9, 15] for fetal hepatocyte isolation for primary culture; this protocol is relatively convenient with s high cell yield along with a high purity of more than 95%. Meanwhile, the current study also evaluated the hypoxia effect on the cell growth and death of fetal hepatocytes. Our results indicated that under hypoxia treatment, the growth rate of the primary cultured hepatocytes decreased along with exposure time, although it showed a minimal increase in the first 24 h exposure, and cellular organelles, such as mitochondria and ER, became swollen with chaotic arrangement. The hepatocyte apoptosis rate under hypoxia gradually increased in a time-dependent manner, and at each time point, the result observed in the experimental group was significantly higher than in the control group. According to previous animal studies, these data further support the effect of prenatal hypoxia on the susceptibility of fetus later in life to metabolic syndromes. We will further use the primary culture system of fetal hepatocytes to test the mechanism of cell apoptosis under hypoxia and other pathway for biological changes, including autophagy and senescence.
-
All authors are acknowledged for their contribution to the study.
National Natural Science Foundation 81671535
Jiangsu Key Discipline of Human Assisted Reproduction Medicine Foundation FXK201749
Chinese Medical Association Clinical Medicine Research Reproductive Medicine 17020270696
Jiangsu Provincial Medical Talent of the Project of Invigorating Healthcare through Science, Technology and Education ZDRCA2016044
Jiangsu Science Foundation BE2015642
National Natural Science Foundation 81370719







 Quick Links
Quick Links
 DownLoad:
DownLoad: