-
Bisphenol A (BPA) is believed to affect a variety of physiological functions associated with human diseases[1]. Bisphenol F (BPF) is an alternative to bisphenol A. Ninety percent of BPF is used as a monomer in the synthesis of polyvinyl chloride (PVC) and other compounds[2]. Bisphenol S (BPS) is a structural analog of bisphenol A, which is widely used as a substitute in a variety of daily-use consumer goods and products. This compound is often detected in indoor dust, urine, and human blood.
Environmental pollutants are continuously released in various ecosystems around the world and different organisms are exposed to a large number of diverse and complex combinations of contaminants. Therefore, for this present study, Caenorhabditis elegans (C. elegans) was chosen to assess the effects of bisphenols on the mortality of a terrestrial species. The main objective was to evaluate the acute toxicity of a combination of three bisphenols (BPA, BPS, and BPF) in terms of the mortality rate, movement behavior, and physiological and biochemical indices of C. elegans.
Bisphenols were obtained from Sigma Chemical Reagent Company Chemical of China Pharmaceutical Group (Sigma-Aldrich Co., Shanghai, China) and superoxide dismutase (SOD) Kit was obtained from Nanjing Institute of Biological Research Center (China). Nematodes (wild-type N2) were obtained from Caenorhabditis Genetics Centre (Nanjing Southeast University, China). Nematodes were cultured on nematode growth medium (NGM) plates and fed Escherichia coli OP50; they were incubated in the dark at 20 °C before usage.
The 24 h-LC50 for each bisphenol was determined based on a single acute toxicity test. The concentration gradients of bisphenols were set to 1/5 LC50, 1/2 LC50, and LC50. The number of nematode deaths was recorded under a stereoscopic microscope (BX51 Olympus Cooperation, Tokyo, Japan) at 20× magnification using cellSens Entry 1.16 software (Olympus Cooperation, Tokyo, Japan) after exposure for 24 h.
To assess the head trashing frequency, the head thrashing index of the nematode was determined after 24 h of incubation at 20 °C. The thrashing frequency was observed under a stereo microscope (BX51 Olympus Cooperation, Tokyo, Japan) at 20× magnification and evaluated by determining the change in the direction of the mid body and the number of head thrashings in 1 min, following the protocol described previously[3]. The experiment was conducted in triplicate.
The body bending was assessed as a change in the direction of the part of nematodes corresponding to the posterior bulb of the pharynx along the y-axis while the nematode was moving along the X-axis[3].
To assess the effect on the body length, the nematodes were exposed to different concentrations (0, 0.001, 0.01, 0.1, 1, 10, and 100 μmol/L) of the bisphenol combinations. The physiological and biochemical indices for nematodes were determined after 24 h of incubation at 20 °C. The experiment was conducted in triplicate.
The SOD activity in nematodes (L4 stage of C. elegans was determined using the kit. The nematodes were transferred to NGM containing different concentrations (0, 0.001, 0.01, 0.1, 1, 10, and 100 μmol/L) of bisphenols. The organisms were later subjected to ultrasonication (SA53A, Guangdong, China) and the lysate was collected after centrifugation (Centrifuge 5810, Vaudaux-Eppendorf AG, Schnenbuch, Switzerland) at 3,000 rpm for 10 min at 4 °C. After the completion of reaction for determination of SOD activity, the absorbance was measured at a wavelength of 550 nm using a spectrophotometer (Shimadzu Co., Kyoto, Japan). The experiment was performed in duplicate.
Data were analyzed using SPSS 23.0 software (SPSS Inc., USA), and the value for 50% lethal concentration (LC50) was obtained by regression analysis. The graphs were processed using Origin 8.5 software (Origin Lab Corporation, Northampton, MA, USA) and statistical significance was defined as a P-value < 0.05 and < 0.01.
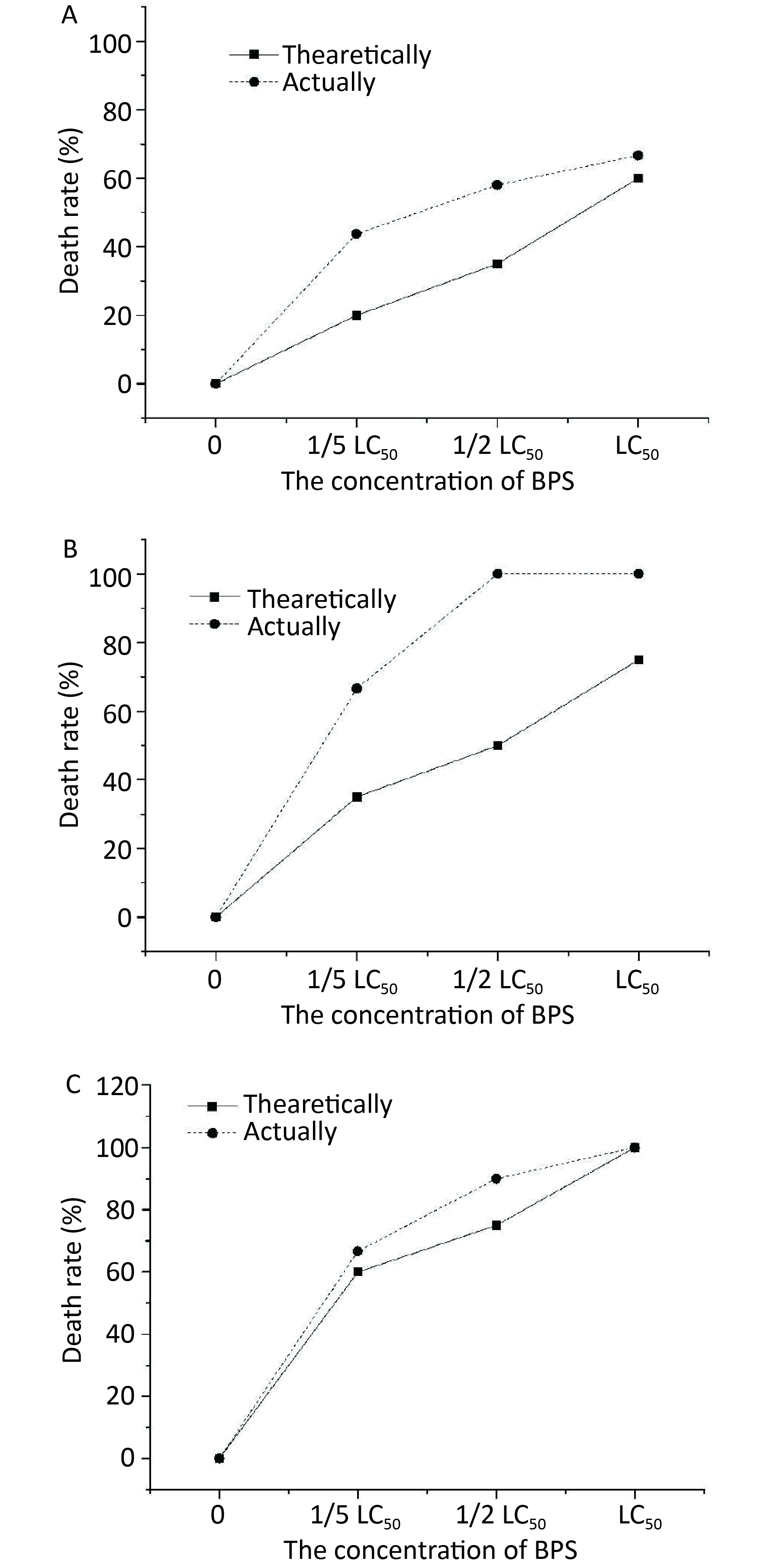
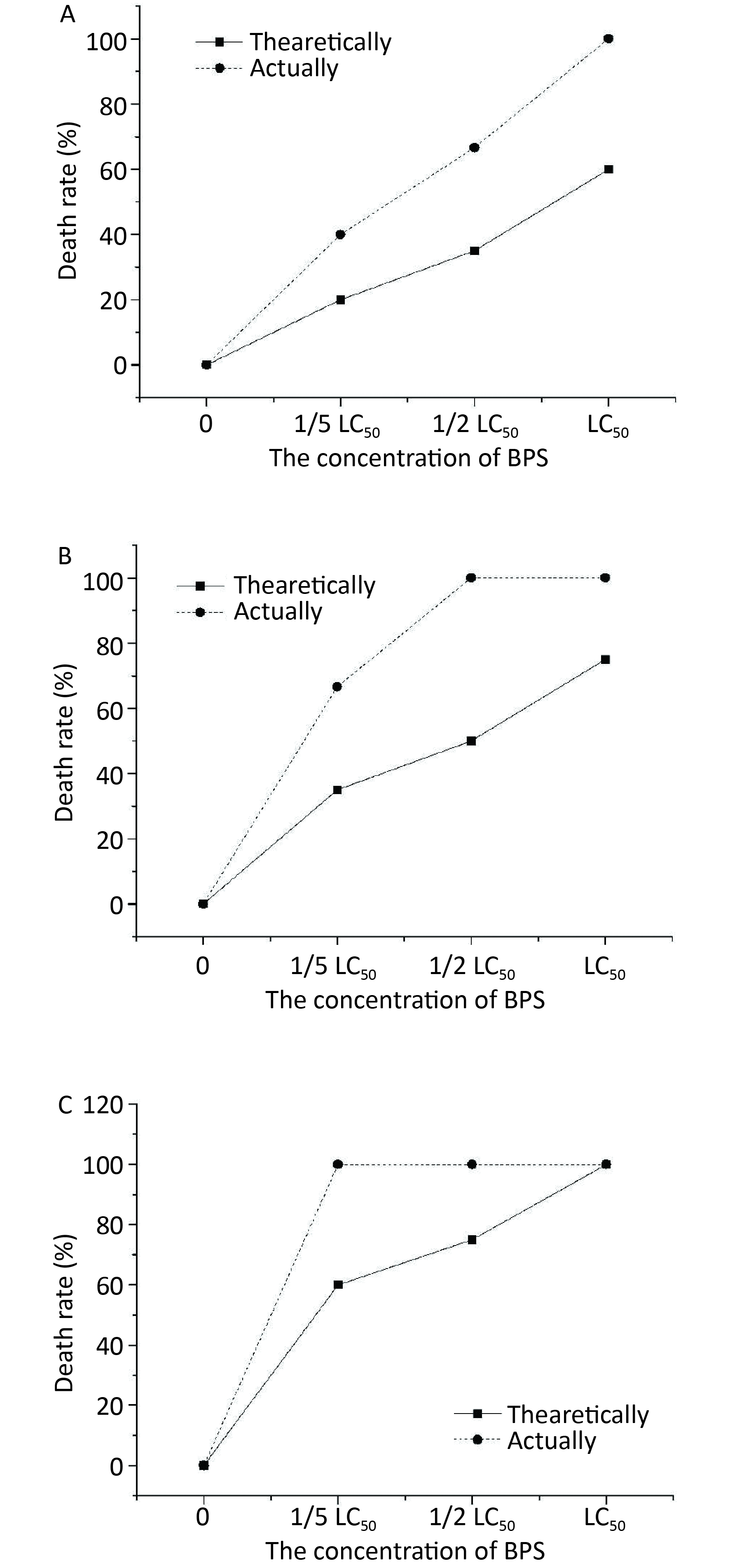
The toxicities of treatment with a combination of BPS and 1/5 LC50 BPA on C. elegans are shown in Figure 1A. The results showed that the toxicity determined in terms of the actual mortality rate (42%) of nematodes was higher than the theoretical mortality rate (18%), signifying that the combined toxicity of BPS and 1/5 LC50 of BPA was synergistic in nature. A more pronounced synergy in the effect of BPS in killing C. elegans was observed when 1/5 LC50 BPA concentration was added to BPS). The synergy between medium and high concentrations of BPS and low concentration (1/5 LC50) of BPA was significantly (P < 0.05) higher than that between the low concentrations of both BPS and BPA. As shown in Figure 1B, a similar observation was also made when combined 1/2 LC50 BPS and 1/2 LC50 BPA concentration, the actual and theoretical mortality rate of C. elegans was 58% and 32%, respectively. In this case, the synergistic effect of BPS and 1/2 LC50 BPA became significantly (P < 0.05) with the increase concentration of BPS and at medium and high concentration as compared with their lower concentrations. Noticeably, the actural mortality rate of C. elegans was 100% with the treatment of LC50 BPA and differernt concentrations of BPS as depicted in Figure 1C. The actual mortality (100%) of nematodes was higher than the theoretical mortality (58%) when BPS concentration was at LC50; thus, the combined toxicity of BPS and LC50 BPA was synergistic.
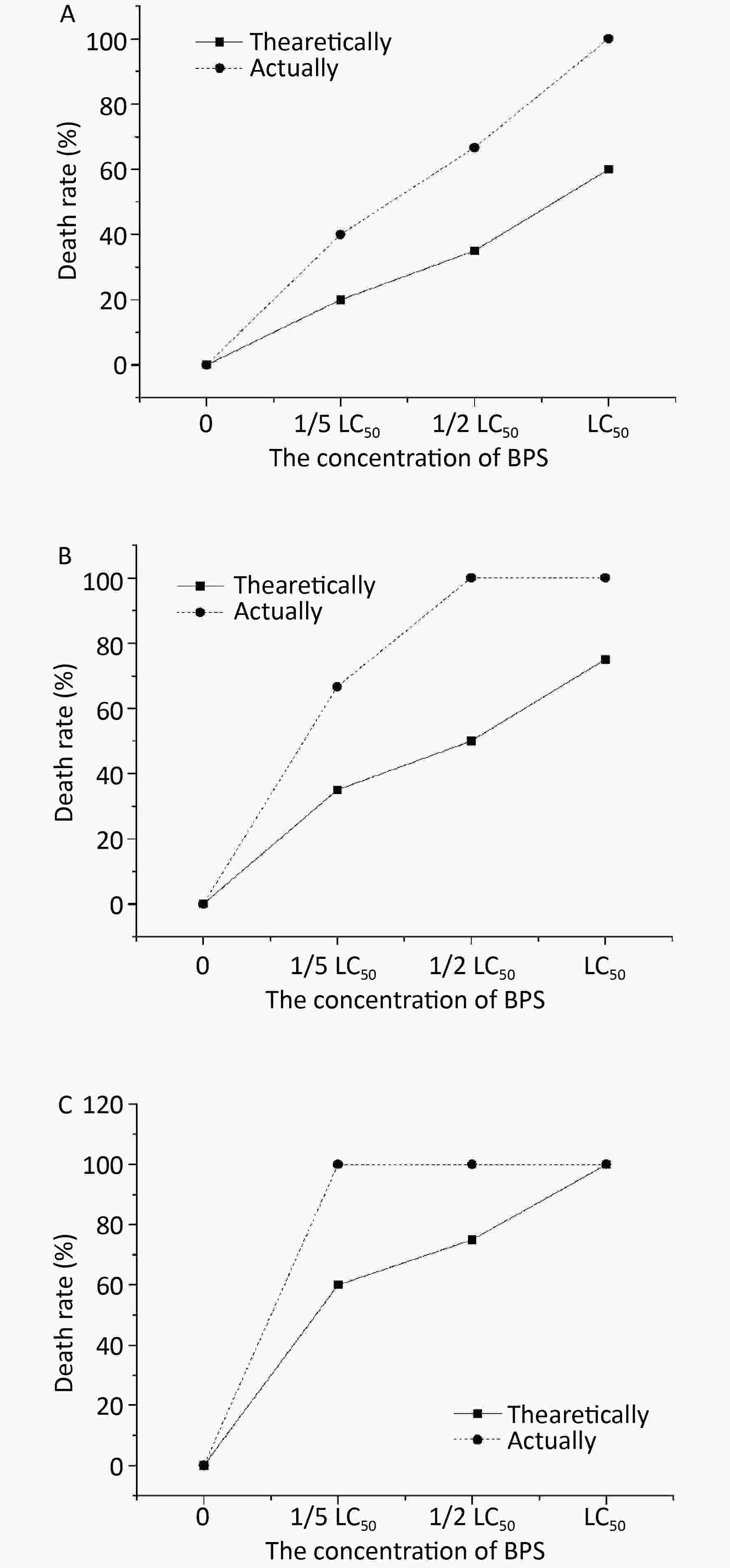
Figure 1. Joint toxicity of different BPA and BPS exposed to C. elagans. (A) Joint toxicity of 1/5 LC50 BPA and BPS; (B) Joint toxicity of 1/2 LC50 BPA and BPS; (C) Joint toxicity of LC50 BPA and BPS. Data (mean ± SEM) are expressed as the percentage value compared to the control group. The asterisks indicate significant differences between the exposure group and control group. BPA, Bisphenol A; BPS, Bisphenol S.
The actual and theoretical mortality rates of nematodes shown in Figure 2, also followed a similar synergistic trend for toxicity in the organism at their combined levels as observed for BPS and BPA. The weaker synergistic effect on toxicity was observed at combined 1/2 LC50 BPS and 1/5 LC50 BPF concentration (Figure 2A). The effects of combined 1/5 BPS and 1/2 BPF was synergistic as evidenced by higher actual mortality rate (68%) when compared with the theoretical mortality rate (38%) (Figure 2B). The synergistic effect was determined at LC50 of BPS and BPF, and higher actual mortality rate compared to the theoretical mortality rate was recorded (Figure 2C).
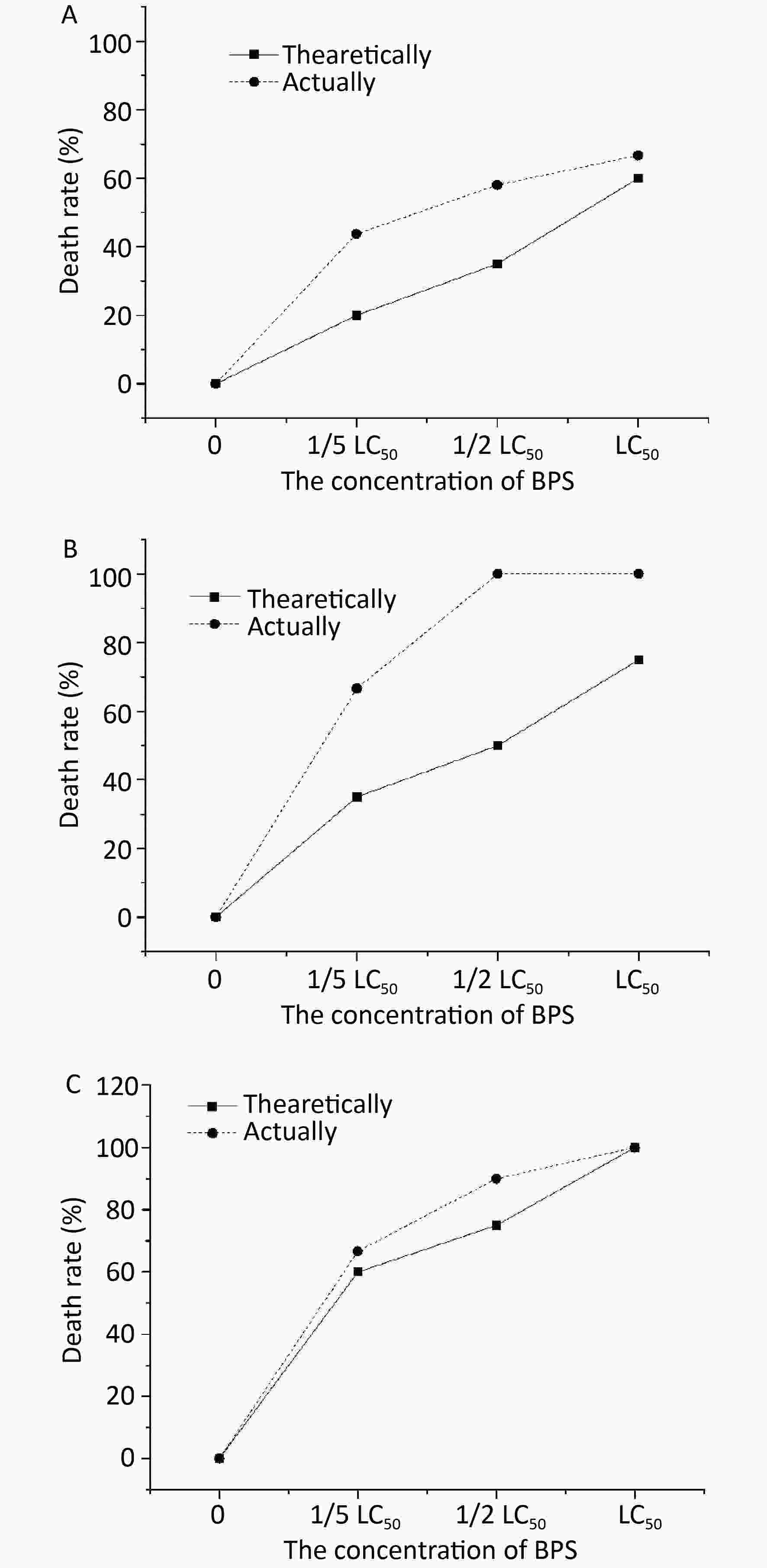
Figure 2. Joint toxicity of different BPF and BPS exposed to C. elagans. (A) Joint toxicity of 1/5 LC50 BPF and BPS; (B) Joint toxicity of 1/2 LC50 BPF and BPS; (C) Joint toxicity of LC50 BPF and BPS. Data (mean ± SEM) are expressed as the percentage value compared to the control group. The asterisks indicate significant differences between the exposure group and control group. BPF, Bisphenol F; BPS, Bisphenol S.
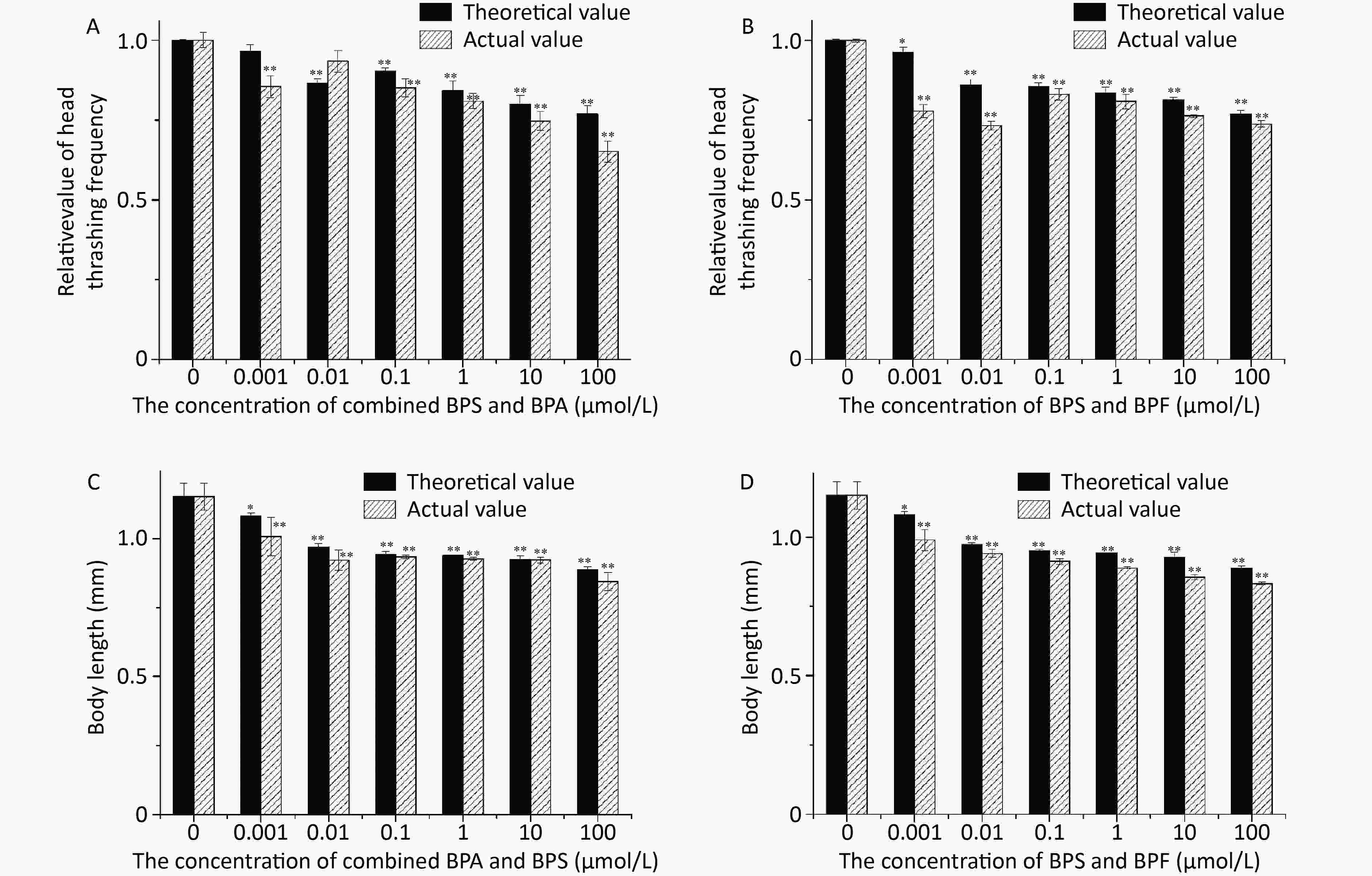
A comparison between the theoretical and actual relative effects of BPS and BPA on head thrashing frequency is presented in Figure 3A. It can be observed that higher was the concentration of bisphenols, greater was the inhibitory effect on the head swing frequency of nematodes. When the concentration was 0.01 μmol/L, the actual value was higher than the theoretical value.
A comparison between the theoretical and actual relative effects of BPS and BPF exposure on the toxicity is presented in Figure 3B. The results showed that the combined toxicity of BPS and BPF was synergistic effects on head thrashing of C. elegans under the series of concentrations studied. The synergistic effect of BPF and BPS was significant when the concentrations were 0.001 and 0.01 μmol/L, respectively.
The theoretical and actual relative effects of BPS and BPA exposure on C. elegans are exhibited in Figure 3C. A similar trend was observed for the body length of nematodes upon treatment with the BPS and BPA combinations. It was noted that an increase in the concentrations of bisphenols led to an increase in the inhibition of body length in terms of growth and development of C. elegans. Figure 3D which revealed also an increase in toxicity with increasing concentrations of combined BPS and BPF. Despite the synergy of toxicity in all the concentration only 0.001 μmol/L had a significant effect.
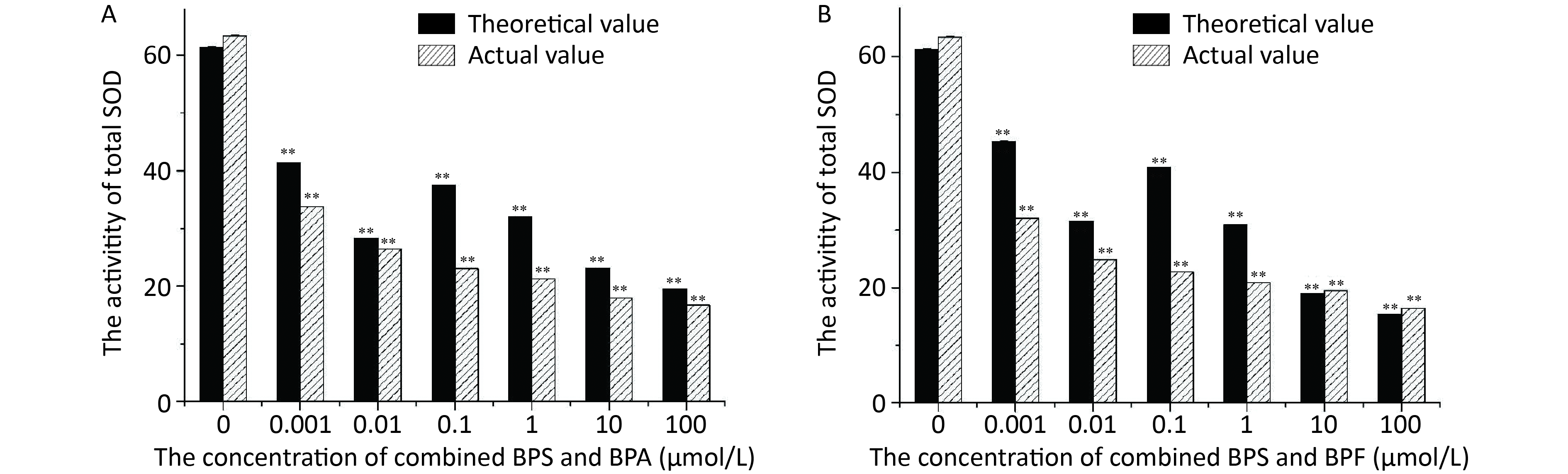
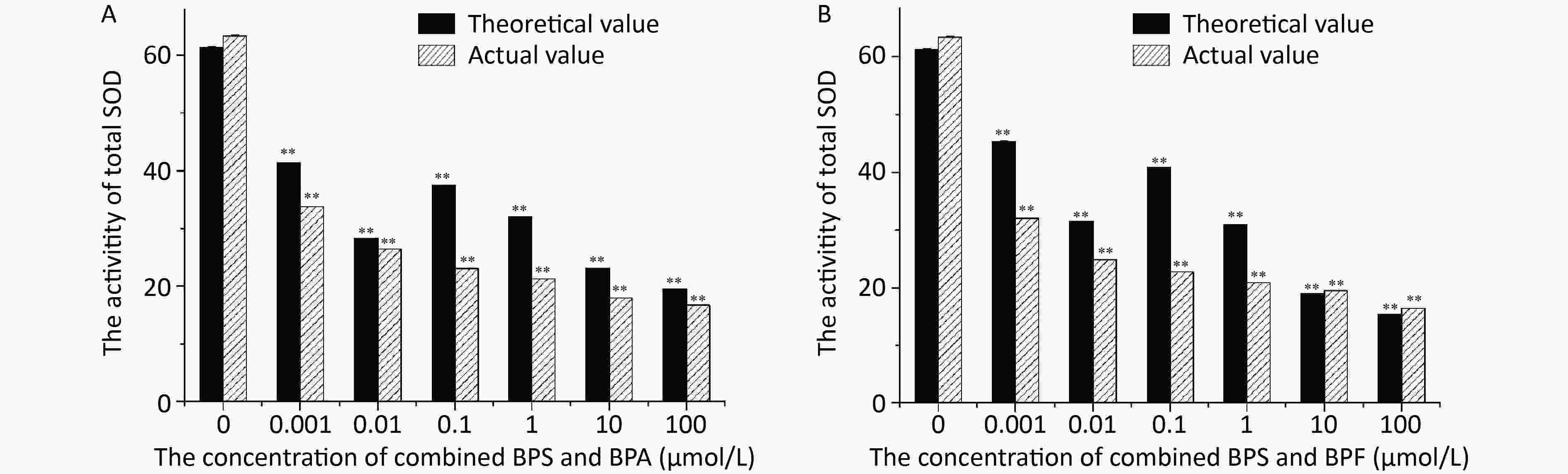
The SOD activities determined in L4 stage C. elegans exposed to BPA and BPS for 24 h are shown in Figure 4A. The increase in BPS and BPA concentrations led to a decrease in the SOD activity. The actual value of inhibition of SOD activity was slightly higher than the theoretical value, indicating that the combined toxicity of BPS and BPA with regard to the SOD activity in nematodes was synergistic. The SOD activity in C. elegans after 24 h of exposure to BPS and BPF is shown in Figure 4B. It can be observed that increase in BPS and BPF concentrations led to a decrease in the SOD activity.
For this research, the toxicity of three different concentrations was combined at various concentrations (1/5 LC50, 1/2 LC50, and LC50) and the mortality rate was measured. Similarly, García-Espieira, et al[4]. observed a synergistic effect for atrazine and glyphosate compounds used at various concentrations. Vingskes and Spann[5] also observed a synergetic effect of the antiseptics, triclosan and triclocarban, on the growth of C. elegans. Recent studies have shown that the combined toxicity increases in ternary mixtures with respect to the toxicity of binary combinations[6]. This finding is in accordance with the results of the present study, in which the percentages of synergistic mixtures at three representative levels (1/5 LC50, 1/2 LC50, LC50) and of seven different concentrations (0, 0.001, 0.01, 0.1, 1, 10, and 100 μmol/L) were used. These results showed that the nature of the toxicological interaction was dependent on the concentrations in the mixture, but was less influenced by the toxicological mode of action of each component. For the L4 stage, the BPA individually did not have any significant effect on the head thrashes at 0.01 μmol/L, which was also confirmed by Zhou, et al.[7]. According to Zhou, et al.[8], the head thrash is the most sensitive parameter for assessing the toxicity of BPA compounds. In the present study, locomotion behavior was used to evaluate the neurotoxicity in C. elegans at the L4 larval stage following exposure to the compound for 24 h. The results indicated that the joint toxicity of 1/5 LC50 BPA and BPS had a negative effect on the locomotion behavior of C. elegans, especially on the head thrashes. BPA is a typical endocrine disruptor associated with a variety of health symptoms, including changes in neurological disorders, dysplasia, and reproductive disorders[9]. When the concentrations of BPS in the combinations were LC50, 100% mortality was observed. Thus, higher the concentration of bisphenols, greater is the inhibitory effect on the head swing frequency of nematodes, and thus, the head swing frequency of the nematodes reduced significantly, as reported previously[10].
Also, there is evidence that combined toxicity of bisphenols has a certain effect on the endoplasmic reticulum, mitochondria, and cell membrane, and can regulate caspase activation, leading to apoptosis[11]. Moreover, oxidative stress has been shown to be involved in aging and in the development of various diseases[12]. We found that the combined exposure could significantly induce an increase in ROS levels and a decrease in SOD levels in C. elegans.
The study demonstrates that treatment of C. elegans with combinations of bisphenols has an effect on the mortality/death rate, locomotion behavior, and physiological and biochemical indices.
Effects of Treatment with Different Combinations of Bisphenol Compounds on the Mortality of Caenorhabditis Elegans
doi: 10.3967/bes2020.020
- Received Date: 2019-05-20
- Accepted Date: 2019-12-03
| Citation: | Naomi HAMAMBA, LI Jie, ZHANG Xiao Wei, ZHANG Cai Qin, Frederick SARPONG, ZHOU Xing Hua. Effects of Treatment with Different Combinations of Bisphenol Compounds on the Mortality of Caenorhabditis Elegans[J]. Biomedical and Environmental Sciences, 2020, 33(2): 145-149. doi: 10.3967/bes2020.020 |







 Quick Links
Quick Links
 DownLoad:
DownLoad: