-
Periodontitis belongs to periodontal diseases, which is a chronic inflammatory disease mainly involving periodontal supporting tissue[1]. Chronic periodontitis is the most common type of periodontitis, accounting for approximately 95% of patients with periodontitis[2]. The disease progression of chronic periodontitis is relatively slow. In most cases, microorganisms and their products first accumulate in gingiva for a long time, causing gingival inflammation. Once gingivitis is not treated properly, periodontal tissues including the periodontal ligament, cementum and alveolar bone could be damaged, resulting in the occurrence of periodontitis[3]. Periodontitis occurs primarily in adults but the incidence rates among young people increases, seriously affecting people’s life quality[4,5]. It is noteworthy that periodontitis affects not only oral tissues, but also many systemic diseases. Besides, periodontitis has been considered as a strong risk factor for diabetes and cardiovascular disease[6].
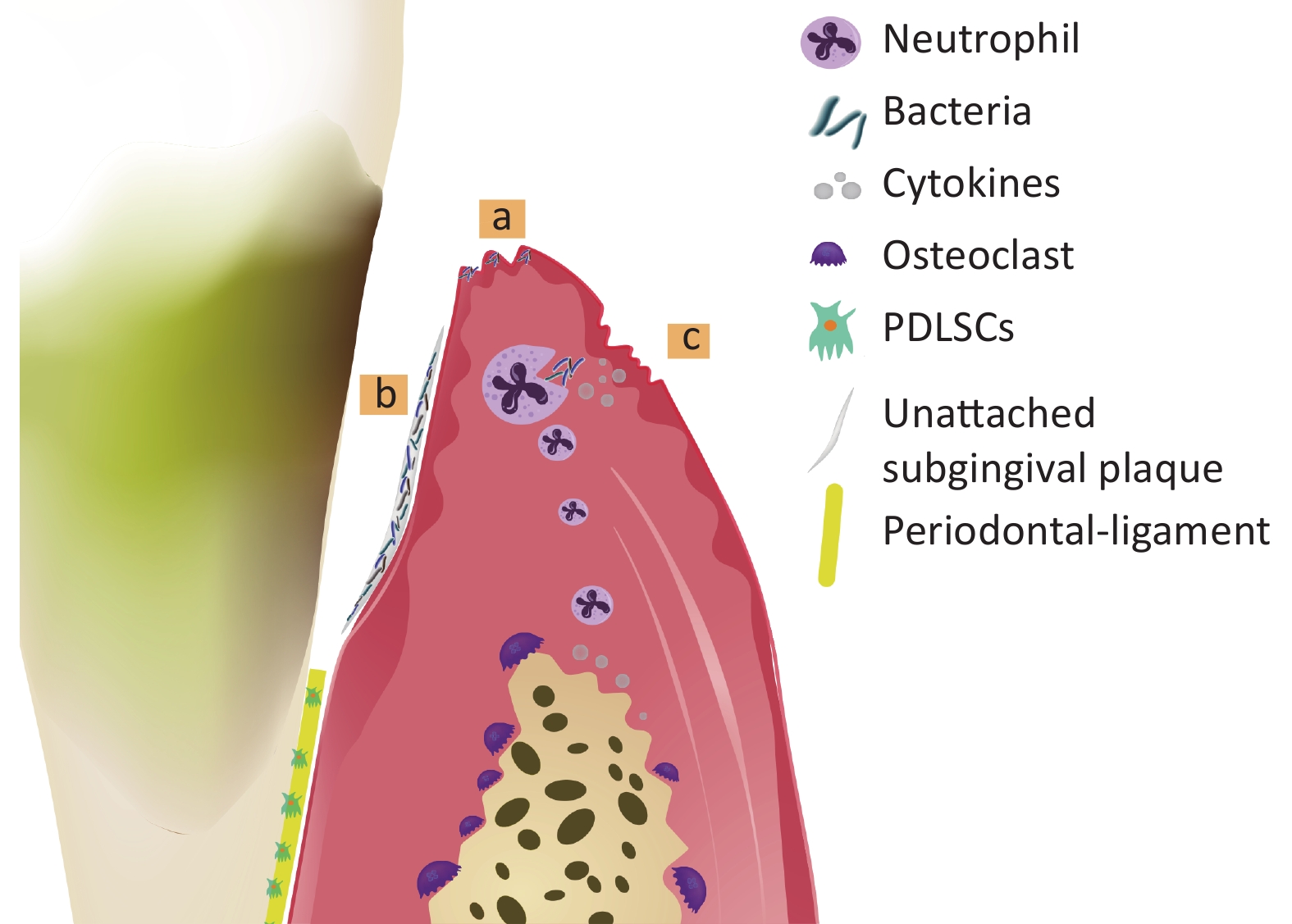
At present, periodontitis is acknowledged to be a multifactorial disease with complicated pathogenesis. Bacteria, host and environment are all involved in the occurrence and development of periodontitis[7]. Firstly, subgingival plaque formed by bacteria and their products is the initiation factor of periodontitis, which could potentially accelerate the destruction of alveolar bone[8,9]. Secondly, the host immune responses prevent microorganisms from entering periodontal tissue in the early stage of infection, but excessive cytokines, prostaglandins and matrix metalloproteinases production lead to periodontal supporting tissue damage and bone loss[10]. Periodontitis often occurs when the balance between microorganism and host defense is broken[11,12]. Finally, the host defensive responses are markedly influenced by environmental factors such as genetic background and smoking, which could also contribute to the development of periodontitis[13]. The pathogenesis of periodontitis is shown in Figure 1.

Figure 1. Pathogenesis of periodontitis. Bacteria invade the gingival tissues and cause gingival inflammation (a). Bacteria are not controlled timely and gather to form the unattached subgingival plaque, causing alveolar bone absorption and attachment loss (AL) (b). Although neutrophils engulf bacteria via phagocytosis at the early phase of inflammation, excessive pro-inflammatory cytokines produced during defense response accelerate the progress of periodontitis (c).
Non-coding RNAs (ncRNAs) are the RNAs that cannot be able to encode proteins. In the early stage, mRNAs are acted as a protein translation template while ncRNAs are considered as "transcription garbage"[14]. Heretofore, researchers found that around 98% of the human genome are transcribed into ncRNAs, extensively regulated the gene expression during the progress of diseases[15]. In inflammatory-related diseases like periodontitis, ncRNAs play the important role in regulating the homeostatic of cellular functions[16]. Studies about ncRNAs in the development of periodontitis mainly focus on microRNAs (miRNAs), long non-coding RNAs (lncRNAs) and circular RNAs (circRNAs)[17]. However, how these ncRNAs interact and which specific mechanism involves in periodontitis remains to be explored[18]. Therefore, our review focuses on the dual role of miRNAs, lncRNAs and circRNAs related to inflammation, cell proliferation, apoptosis and osteogenic differentiation in periodontitis, aiming to explore more diagnostic markers and therapeutic targets of periodontitis.
-
MiRNAs have about 22 nucleotides in length and are first transcribed into primary RNAs (pri-miRNAs), then undergo sequential biogenesis, from pre-miRNAs and finally into mature miRNAs[19]. Later, mature miRNAs mostly silence the target genes by degrading or preventing translation[20]. Besides, the ceRNA network formed with miRNA- lncRNA/circRNA-mRNA was closely associate with the development of many diseases[21-24]. More importantly, miRNAs provide a powerful pathway for the diagnosis and treatments of disease[25,26].
-
LncRNAs, a class of ncRNAs with length greater than 200 nucleotides, were processed by RNA polymerase II (Pol II)[27]. Unlike mRNA, lncRNAs prefer to enrich in nucleus with the following possible mechanisms: 1) degradation or tethering on chromatin; 2) longer distance between the 3′ splice site and the branch point; 3) through nuclear retention elements[28,29]. In the nucleus, lncRNAs are closely related to chromatin, and directly or indirectly regulated chromatin organization by chromatin regulatory proteins (SWI/SNF or PRC subunits), thereby affecting the chromatin activity[30-32]. In addition, nuclear lncRNAs are also widely involved in transcriptional process through cis/trans regulation or transcription factors, in post-transcriptional process through alternative splicing or A-to-I editing, finally regulated the genes expression[33-36]. In the cytoplasm, diverse RNA binding proteins (RBPs) or miRNAs were interacted with lncRNAs to implement functions[37]. So far, many studies suggested the functional interactions among lncRNAs and miRNAs: 1) lncRNAs are degraded by miRNAs[38]; 2) lncRNAs serve as sponges/decoys for miRNAs to form ceRNAs, thereby derepressing miRNA target mRNAs[39]; 3) lncRNAs compete with miRNAs for degradation of target mRNAs[40]; 4) lncRNAs produce miRNAs[41]. In general, lncRNAs are widely involved in many cell processes and thus to regulate the progress of disease[42].
-
CircRNAs belong to a special class of ncRNAs with a covalent closed loop structure formed by trans-splicing. Up to now, the biological functions of circRNAs are summarized as follows: 1) as a ceRNA or miRNA sponge; 2) binding to proteins; 3) as a potential translation template for proteins; 4) regulating the pre-RNA splicing and parental gene expression[43]. As more molecular biology techniques were addressed to RNA studies, circRNAs with biological functions related to diseases has gradually been discovered, especially in various cancers[44,45]. Importantly, numerous circRNAs have been demonstrated to be potential biomarkers, such as circCHST15, circ-PTPDC1 and hsa_circRNA_0039480[46-48].
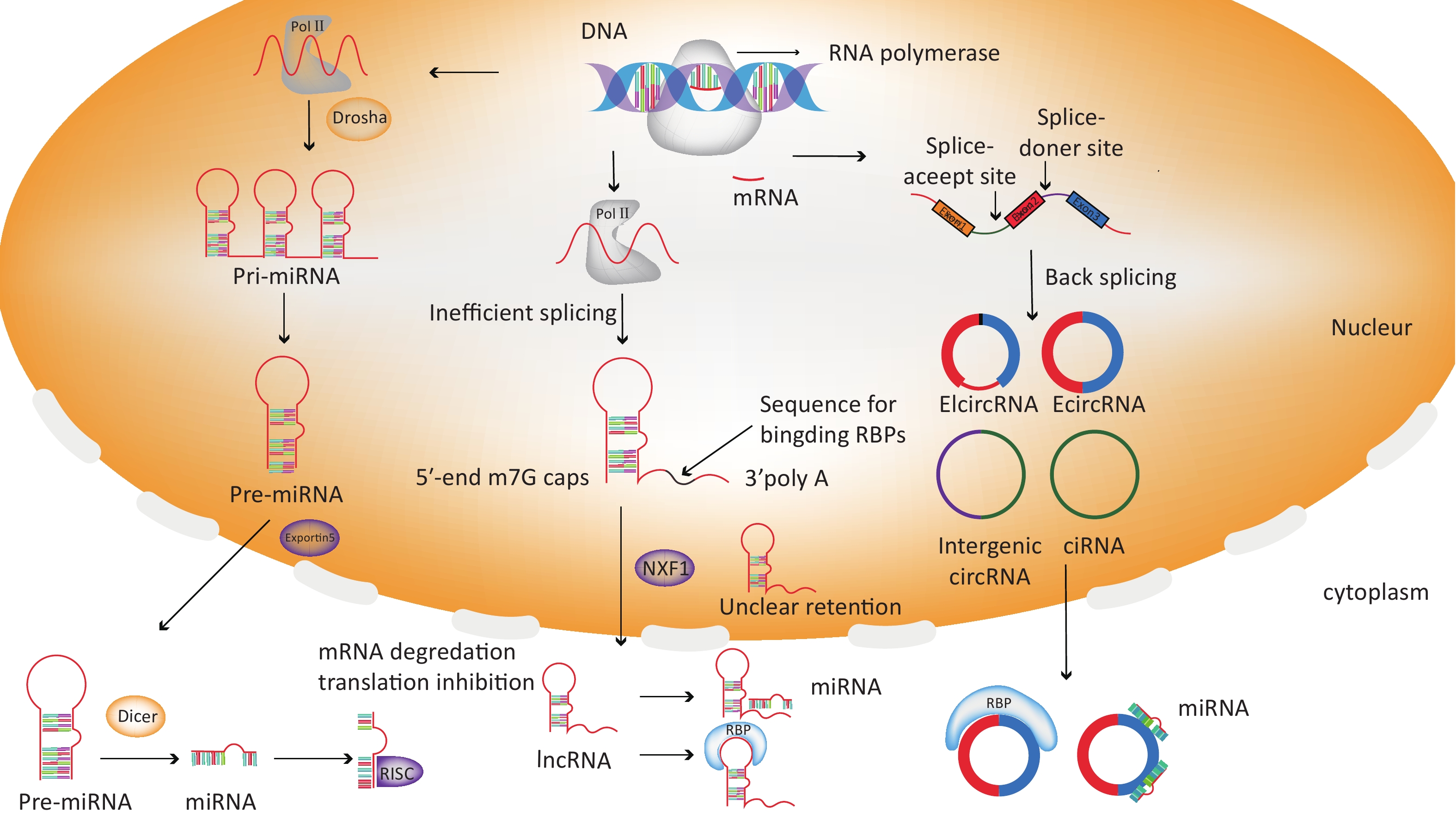
The formation process of miRNAs, lncRNAs, and circRNAs are shown in Figure 2.

Figure 2. The forming process of miRNAs (a), lncRNAs (b), and circRNAs (c). The genes of ncRNAs are firstly transcribed into linear RNAs by DNA polymerase in nucleus. Then, (a) miRNAs were processed from pri-miRNAs to pre-miRNAs through RNA polymerase II (Pol II) and Drosha, thereby transporting into the nucleus by export5. In the cytoplasm, mature miRNAs are formed by Dicer and subsequently bind to the silent effector complex (RISC), finally inhibited the expression of target gene. (b) Most lncRNAs are remained in the nucleus under the inefficient processing of Pol II and added a cap at the 5' end and a poly (A) tail at 3'. A small part of lncRNAs is then transported into the nucleus through NFX1 and further combined with miRNAs to release the inhibitory effects of miRNAs on target genes. (c) Four circRNAs are formed by trans-splicing, and subsequently act as miRNAs sponge or combine with RBP to regulate gene expression.
-
The protective effects of miRNAs on periodontitis are mainly concentrated on inhibiting pro-inflammatory cytokine production, improving cell functions and preventing the bone loss (Table 1). Functions MiRNAs LncRNAs CircRNAs Suppress Apoptosis, Inflammation, Proliferation miR-218 (target Mmp9)[49] miR-205-5p (target XBP1/ IL6ST)[50,51] miR-543-3P (target KLF6)[52] miR-212-5p (target Myd88)[53] miR-17-5p (target VEGFA)[54] miR-28-5p (target SPHK1)[55] miR-223-3p (target NLRP3-Caspase 1-GSDMD)[56] miR-466l (regulate IL-10)[57] lncRNA H19 (regulate PI3K/AKT)[58] IncRNA TUG1 (sponge miR-498/RORA)[59] lncRNA GAS5 (regulate NF-κB pathway)[60] IncZFY-AS1 (sponge miR-129-5p/DDX3X)[61] lncRNA PTCSC3 (target TLR4)[62] lncRNA MAFG-AS1 (target TLR4)[63] lncRNA LOXL1-AS1 (target IL-1β)[64] Iinc-RAM (target FGF2)[65] lncRNA JHDMID-AS1 (target DNAJC10)[66] IncRNA TUG1 (sponge miR-132)[67] IncRNA FGD5-AS1 (sponge miR-142-3p/SOCS6)[68] lncRNA DCST1 AS1 (sponge miR-21precursor/PLAP-1)[69] lncRNA MALAT1 (sponge miR-155-5p/ETS1)[70] lncRNA DLX2-AS1 (sponge miR-330-3p)[71] Circ 0062491 (sponge miR-498/SOCS6)[72] Circ_0085289 (sponge let-7f-5p/SOCS6)[73] Circ_0081572 (sponge miR-378h/RORA)[74] CircLRP6 (sponge miR-145a-5p/Zeb2)[75] hsa_circ_0003948 (sponge miR-144-3p/NR2F2)[76] Circ_00668819 (sponge miR-144-5p/RORA)[77] Bone Protection miR-21 (target Spry1)[78] miR-383-5p (target HDAC9)[79] miR-10a (Treg differentiation)[80] miR-17 (target HDAC9)[81] miR-203 (regulate AP-1/ICAM-1)[82] miR-200c (target IL-6, IL-8, and CCL-5)[83-85] miR-101 (regulate EZH2 /VCAM‐1)[86] miR-335-5p (unclear)[87] miR-543 (target TOB2)[88] miRNA-26a-5p (target Wnt5a)[89] IncRNA Nron (regulate NKRF)[90] lncRNA OIP5-AS1 (sponge miR-92a-3p)[91] lncRNA GAS5 (regulate GDF5 and p38/JNK)[92] lncRNA ANRIL (sponge miR-7/ NF-κB; miR-7-5p/IGF-1R)[93,94] IncRNA TWIST1 (regulate TWIST1)[95] IncRNA TUG1 (sponge Lin28A/miR-222-3p)[96] lncRNA XIST (sponge miR-214-3p) [97] lncRNA MEG3 (sponge miR-27a-3p/IGF1)[98] LINC00707 (sponge miR-490-3p/FOXO1)[99] CircMAP3K11 (sponge miR-511/ TLR4)[100] Circular RNA CDR1as (sponge miR-7/GDF5/SMAD0)[101] CircRNA FAT1 (sponge miR-4781-3p/SMAD5)[102] Circ 0062491 (sponge miR-142-5p/IGF1 axis)[103] Note. Mmp9: matrix metallopeptidase 9, XBP1: X-box binding protein 1, KLF6: kruppel-like factor 6, Myd88: myeloid differentiation primary response gene 88, IL6ST: interleukin 6 signa transducer, VEGFA: vascular endothelial growth factor A, SPHK1: sphingosine kinase 1, PI3K/AKT: phosphatidylinositol3-kinase/threonine protein kinase, TLR4: toll-like receptor 4, RORA: RAR-related orphan receptor A, NF-kB: Nuclear factor-kappa B, DDX3X: DEAD box polypeptide 3, IL-1β: β interleukin 1, FGF2: fibroblast growth factor 2, DNAJC10: a heat shock protein 40 family member, SOCS6: suppressor of cytokine signaling 6, PLAP-1: periodontal ligament-associated protein 1, ETS1: v-ets erythroblastosis virus E26 oncogene homolog 1, HIF-1α: hypoxia inducible factor 1 alpha, Zeb2: zinc finger E-box binding homeobox 2, NR2F2: nuclear receptor subfamily 2 group F member 2, Spry1: sprouty homolog 1, HDAC9: histone deacetylase 9, AP-1: activator protein 1, IGF1: insulin like growth facto 1, SMAD5: small mothers against decapentaplegic 5, GDF5: growth differentiation factor 5, SMAD: small mothers against decapentaplegic, TLR4: toll-like receptor 4, FOXO1: forkhead Box 1, IGF1: insulin-like growth factor-1, TWIST1: twist-related protein 1, NKRF: NF-κB repressing factor, JNK: jun N-terminal kinase. Table 1. Protective ncRNAs in the development of periodontitis
-
Burgeoning findings have revealed that miRNAs expression was dysregulated in periodontal tissues with periodontitis, resulting in abnormal expression of target genes[104]. For example, 91 miRNAs were differentially expressed in periodontal inflamed gingival tissues by gene sequencing technology, in which hsa-miR-126* and hsa-miR-20a were among the miRNAs with the most significant alteration[105]. The same conclusion was showed in a mouse study of periodontitis, which demonstrated that the miRNA expression was significant changed between healthy and periodontitis animals[106].
Emerging convincing studies suggested that miRNAs could control the progression of inflammation[16,107]. For example, exosomal miR-205-5p derived from PDLSCs relieved the inflammation by directly targeting XBP1 to influence the Th17/Treg balance in periodontitis rat model[50]. In periodontitis transgenic mice overexpressing miR-335-5p (335-Tg), 335-Tg group successfully relieved gingiva inflammation compared with wild-type mice[87]. Moreover, miR-17-5p suppressed abnormal angiogenesis by targeting vascular specific markers (CD31 and VEGFA), thereby controlling inflammatory infiltrates[54]. Besides, miRNAs could delay the development of periodontitis by elevating cell viability, inhibiting apoptosis and abnormal proliferation. For example, in LPS-induced cell apoptosis and viability inhibition, the overexpression of miR-543-3p, miR-212-5p and miR-203 reversed the destructive effects of LPS on PDLSCs functions by targeting KLF6, Myd88 and AP-1/ICAM-1 respectively[52].
PDLSCs possess multilineage differentiation potential and could differentiate into osteoblasts, thus initiating bone repair[108]. Many miRNAs including miR-383-5p, miR-543, and miR-21 were upregulated during the osteogenic differentiation of PDLSCs and interacted with HDAC9, TOB2, and Spry1, respectively, promoting the osteogenic differentiation of PDLSCs[78,79,88]. Similarly, numerous researchers used carriers in animal model to intuitively prove the ability of miRNAs in facilitating bone formation. For example, the PDLSCs transfected with miR-26a-5p mimics, which were subsequently loaded with hydroxyapatine-tricalcium phosphate (HA-TCT) and implanted into mice, promoted bone formation mechanically by targeting wnt5a[89]. Tadkamol Krongbaramee et al. found the local injection of PEI-plasmid miR-200 nanoplexes into the gingiva in periodontitis rat model effectively reduced alveolar bone loss[85]. In the subsequent study of miR-200c, Liu Hong et al. found that the bone protective effects of miR-200c were fulfilled by precisely targeting the 3’UTR of IL-6, IL-8, and CCL-5[83]. In addition, miR-10a combined the application of IL-2 and TGF-β could also prevent bone loss in a mouse periodontal disease model via nanofibrous spongy microspheres[80]. Some studies also indirectly proved the protective roles of miRNAs in bone loss. For example, in a rat model of periodontitis, miR-17 and miR-101 which separately targeted HDAC9 or EZH2/VCAM-1 ameliorated crestal bone loss[81,86].
-
Recent studies have confirmed the necessity of lncRNAs in the development of periodontitis and the interaction with miRNAs in regulating the cell functions of PDLSCs[109,110]. For example, 8925 lncRNAs were found to be differentially expressed in patients with periodontitis by microarray analysis[111]. Another study predicted that there were 84 lncRNAs involved in inflammatory processes, among them, AC000120.7, MZF1-AS1, FGD5-AS1, HOTAIR, and OIP5-AS1 were verified to downregulate in periodontitis patients[112]. Furthermore, the ceRNA networks conducted by lncRNA-miRNA-mRNAs also regulated the inflammatory process in periodontitis[113]. Those studies all proved that lncRNAs were central for the periodontitis development.
Similar to miRNAs, lncRNAs also account for the pathophysiological alteration of periodontitis. Several lncRNAs are negatively correlated with the degree of inflammatory state of periodontitis. For example, lncRNA GAS5 was downregulated in a mouse ligature-induced periodontitis model and its overexpression alleviated the expression of cytokines by regulating the NF-κB signaling pathway[60]. In LPS-stimulated inflammatory environments, the overexpression of lncRNA TUG1 and lncRNA DLX2-AS1 decreased the production of inflammatory cytokines by separately targeting miR-498 or miR-330-3p, thereby modulating downstream targets genes RORA or RO60[59,71]. Several lncRNAs are negatively correlated with the physiological functions of cells. For example, in PDLSCs from rats under oxidative stress, lncRNA JHDM1D-AS1 overexpression significantly blocked the promoting effect of H2O2 on apoptosis by negatively regulating DNAJC10[66]. In periodontal tissue from periodontitis, the knockdown of lncRNA FGD5-AS1 and lncRNA TUG1 that respectively targeted miR-142-3p and miR-132 exacerbated the inhibitory effects of LPS on PDLSCs, whereas overexpression rescued cell viability, blocked apoptosis and cytokine secretion[67,68]. lncRNA H19 was found to be upregulated in the mouse periodontitis model and closely associated with the activation of autophagy by inhibiting PI3K/AKT pathway, thereby achieving self-renewal of cells[58].
The abnormally increased proliferation of PDLSCs is considered to be associated with the disease progression of periodontitis[114]. The role of lncRNAs in proliferation inhibition was fullfed by targeting different genes. For example, lncRNA LOXL1-AS1, lncRNA DCST1-AS1 and Linc-RAM lncRNA could target TLR4, miR-21 precursor and FGF2 respectively, inhibiting the proliferation of PDLSCs[64,65,69]. Similarly, lncRNA PTCSC and lncRNA MAFG-AS1 could target TLR4 to inhibit the proliferation of PDLSCs as well[62,63]. Interestingly, the likelihood of periodontitis recurrence and proliferation rates of PDLSCs was inversely related to lncRNA MORT expression[115].
Many reports to date have indicated that lncRNAs could promote the expression of osteogenic transcription factors, thereby initiating bone repair[116]. For example, the overexpression of lncRNA-TWIST1 and LncRNA TUG1 upregulated the expression of Runx2, ALP, and OCN by targeting its downstream genes TWIST1 or lin-28 homolog A (Lin28A) respectively, consequently accelerating the formation of mineralized nodules[95,117].
The role of ceRNA formed by lncRNA-miRNA-mRNA in bone repair have been demonstrated by several studies. For example, in inflammatory environments, LncRNA OIP5-AS1 targeted miR-92a-3p directly to promote the osteogenic differentiation of PDLSCs[91]. In non-inflammatory environments under osteogenic induction, the silence of lncRNA TUG1, lncRNA XIST, lncRNA MALAT1, LIN00707 and lncRNA MEG3 decreased the osteogenic differentiation ability by separately sponging miRNA-222-3p, miR-214-3p, miR-155-5P, miR-490-3p and miR-27a-3p[70,96-99]. Similarly, lncRNA ANRIL could target several mechanisms simultaneously to promote the osteogenic differentiation of PDLSCs including miR-7/ NF-κB and miR-7-5p/IGF-1R[93,94]. Another two experiments validated the bone protective effects of lncRNAs at animal level. One of them was periodontitis model using lncRNA Nron knockout transgenic mice, in which TRAP staining results showed lncRNA Nron knockout significantly decreased the number of osteoclasts[90]. In another in vivo transplantation experiment, the HE staining results showed the silence of FoxO1 which negatively regulated lncRNA-POIR inhibited the formation of osteoids, indirectly suggesting the protective role of lncRNA-POIR in bone absorption[118]. Although lncRNAs play key role in PDLSCs osteogenic differentiation, studies on the role in inflammatory environments are rare, which requires further exploration.
-
The latest research has found that circRNAs involved in cell apoptosis, cell migration and oxidative stress are also differentially expressed in periodontitis[119]. For example, 70 differentially expressed circRNAs were identified in gingival tissues from periodontitis patients, among which circPTP4A2, chr22:23101560-23135351+, circARHGEF28, circBARD1 and circRASA2 played crucial role in the diagnosis of periodontitis[119]. Fifty-three circRNAs were differentially expressed in PDLSCs under hypoxia, in which lncRNA-FTX/circRNA-FAT1-hsa-miR-4781-3p-SMAD5 and circRNA LPAR1-hsa-miR-342-3p-ADAR might be involved in the progression of periodontitis[120].
circRNAs usually acted as miRNAs sponges to inhibit inflammation and improve cellular functions[121]. For example, Jie Li et al. first identified circ_0062491 as the sponge of miR-584, and they found it was downregulated in periodontitis patients[122]. Later, the roles of circ_0062491 in periodontitis were further studied. circ_0062491 was first described to directly regulate miR-498/SOCS6 to prevent PDLSCs from LPS-induced apoptosis and inflammation[72]. Another researcher found miR-142-5p/IGF1 axis was directly regulated by circ_0062491, promoting osteogenic differentiation of PDLSCs[103]. In periodontal tissues from periodontitis, the expression of Circ_0081572 and Circ_0066881 were downregulated and they were found to provide a sponge effect on miR-378h and miR-144-5p through targeting 3’ -UTR of RORA, which in turn restored the PDLSCs viability and inhibit apoptosis[74,77]. Likewise, Circ_ 0085289 and hsa_circ_0003948 enhanced cell viability and delayed progression of periodontitis by upregulating the expression of SOCS6 or NR2F2 through forming a competitive ring with let-7f-5p and miR-144-3p respectively, thereby achieving protective effects[73,76]. In periodontitis mouse model, tissues with si-circMAP3K11 accelerated the apoptosis and promoted the proliferation of PDLSCs by negatively regulating miR-511-3p[100]. Nowadays, the efficient bioinformatics prediction greatly helped investigators figuring out the potential role of circRNAs in promoting osteogenic differentiation. For example, Liangkun Xie et al. found 69–557 differentially expressed exosomal circRNAs during osteogenesis induction at day 0, 3, 7 by RNA sequencing. In the following qRT-PCR experiments, hsa_circ_0087960 and hsa_circ_0000437 were validated to be upregulated in the osteogenic differentiation of PDLSCs, meaning the upregulation of these two circRNAs could promote the osteogenic differentiation of PDLSCs[123]. In addition, 139 circRNAs were differentially expressed at day 14 of osteogenesis induction, VENN analysis further revealed that there were 52 circRNAs showed the same trends in all osteogenic stages[124]. Except for the bioinformatics analyses, Yu Ye et al. found the silencing of circFAT1 inhibited the bone formation in rat skull defect model mechanically through competitively binding miR-4781-3p and blocking SMAD5 expression[102]. What’s more, another animal experiment has shown that Circular RNA CDR1as deficiency could also inhibit the bone formation[101]. Consistently, the overexpression of circRNA CDR1as was proportionally related to the stemness of PDLSCs, which greatly enhanced the cell ability of self-renewal[125].
-
Some ncRNAs are positively correlated with the manifestation and pathophysiology of periodontitis. (Table 2).
Functions MiRNA LncRNA CircRNA Promote Apoptosis, Inflammation, Proliferation miR-143-3p (through droving M1 macrophage polarization or targeting KLF5)[126,127] IncRNA 01126 (sponge miR-518a-5p/HIF-1α/MAP or regulate MEK/ERK signaling pathway)[128,129] lncRNA MALA1(target miR-20a/TLR4 or miR-769-5p/HIF3A)[130, 131] lncRNA AWPPH (unclear)[132] lncRNA NEAT1(sponge miR-200c-3p/TRAF6)[133] LINC00616 (sponge miR-370/TFRC)[134] Circ_0138959(sponge miR-527/caspase-5)[135] Circ_0097010 (sponge miR-769-5p/KLF6)[136] Circ_0138960 (sponge miR-518a-5p/HDAC6)[137] Circ_0099630(sponge miR-212-5p/SPRY1 or miR-940/TRAF6)[138-140] Bone Destruction miR-130a (Unclear)[141] miR-325-3 (target Runx2)[142] miR-25a-3p (target CD69 mRNA)[143] miR-138(osteocalcin OC)[144] miR-30a-5p (target Runx2) [145] miR-23a (BMPR1B)[146] miR-214 (target ATF4)[147] miR-6512-3p (target SNHG7)[148] miR-223 (target FGFR2/ TGFβR2)[149] miR-302b (unclear)[150] lncRNA-ANCR (sponge miRNA-758/ Notch2 or regulate canonical WNT signaling pathway) [151, 152] lncRNA DANCR (unclear)[153] Circ_0138959(sponge miR-495-3p/TRAF6)[154] Circ CDK8(regulate ER stress/autophagy)[155] Note. KLF5: Kruppel-like factor 5, HIF-1α: Hypoxia-inducible factor-1alpha, TLR4: Toll-like receptor 4, HIF3A: hypoxia-inducible factor (HIF) 3A, TRAF6: TNF receptor associated factor 6, TLR4: Toll like receptor 4, HDAC6: histone deacetylase 6, SPRY1: sprouty homolog 1, KLF6: Kruppel-like factor 6, ATF4: activating transcription factor 4, TFRC: transferrin receptor, SNHG7: Small nucleolar RNA hostgene 7, FGFR2: fibroblast growth factor receptor 2, TGFβR2: transforming growth factor beta receptor 2. Table 2. Destructive ncRNAs in the development of periodontitis
-
The upregulation of a certain miRNA in periodontitis sometimes exerted destructive role in disease progress. For example, Young Hwa Lee et al. found miR-181b, miR-19b, miR-23a, miR-30a, miR-let7a, and miR-301a in periodontitis tissue were upregulated, which positively related with disease progression. MiR-143-3p was first found to be the most highly expressed miRNA through next generation sequencing (NGS) technology, which was validated to be positively correlated to clinical symptoms[156]. In the subsequent functional experiments, miR-143-3p was demonstrated to inhibit osteogenic differentiation of PDLSCs by targeting KLF5[127]. Recently, exosomal miR-143-3p was proved to enrich in inflammation-affected PDLSCs and could exacerbated inflammatory infiltrates by modulating macrophage polarization in periodontitis mouse model[126]. In LPS-induced inflammation environment, Tetramethylpyrazine protected PDLSCs from apoptosis and cell viability inhibition, however, miR-302b mimic reversed the protective effects[150].
Some miRNAs could inhibit bone formation by directly targeting osteogenic factors (Runx2, OPG, OCN and BMP), thereby limiting the effects of target gene[157]. For example, miR-30a-5p and miR-325-3p weakened bone formation by directly targeting Runx2, and miR‐325‐3p inhibitor rescued the formation of alveolar bone in rats[142,145]. Likewise, miR-23a and miR-138 inhibited osteogenesis of PDLSCs by individually targeting BMPR1B and OCN[144,146]. Additionally, miR-155-3p and miR-223 targeted Kctd1 and GFβR2/FGFR2 respectively, resulting in reduced differentiation of cementoblast[149,158]. Last but not least, miR-214 overexpression leads to a massive inhibition of osteogenic differentiation by directly targeting ATF4 and CTNNB1[147,159].
-
Five lncRNAs are involved in impairing the biological activities of periodontal cells by sponging miRNAs. Specifically, the upregulated expression level of lncRNA NEAT1 in LPS-treated PDLSCs caused the disordered expression of miR-200c-3p and TRAF6. This study further reported lncRNA NEAT1 silencing enhanced cell viability and repressed apoptosis in PDLSCs through miR-200c-3p/TRAF6[133]. In hypoxia environment, LIN01126 promoted apoptosis and inflammatory cytokines release of PDLSCs via sponging miR-518a-5p and inactivating MAPK signaling pathway[128]. Recently, one study reported the relationship between LINC00616 and ferroptosis, and found LINC00616 could promote ferroptosis by interacting with miR-370/TFRC, thereby speeding up the development of periodontitis[134]. LncRNA MALAT1 upregulation in LPS-stimulated human gingival fibroblasts (HGFs) and PDLSCs enhanced inflammatory cytokine production through interacting with miR-20a/TLR4 or miR-769-5p/HIF3A, which was reported by two studies[130,131]. Like lncRNA MALAT1, lncRNA ANCR could also block the osteogenic differentiation of PDLSCs through multiple pathways including activated Wnt signaling pathway and sponged miRNA-758[151,152].
Three lncRNAs was demonstrated to play destructive role in human bio-samples and animal model. For example, lncRNA AWPPH was positively correlated with the recurrence rate of periodontitis according to the analysis of blood samples from 80 patients with periodontitis[132]. LncZFY-AS1, which was elevated in Porphyromonas gingivali oral infected periodontitis mice, knock-down refrained oxidative stress and inflammation in mice with periodontitis by modulating miRNA-129-5p/DDX3X[61]. LncRNA-01126 was the most significantly increased lncRNA in periodontitis patients through lncRNA microarray, which was verified in the gingival tissues of periodontitis mice as well. Further functional experiments proved that lncRNA-01126 overexpression reduced the migration of PDLSCs by inhibiting MEK/ERK signal pathway[129].
-
The destructive role of circRNAs in periodontitis concentrated in disrupting the normal physiological functions of PDLSCs. For example, Jingjing Zheng et al. explored the regulation mechanism of CircCDK8 on osteogenic differentiation under hypoxia, and found CircCDK8 activated endoplasmic reticulum stress, autophagy, and apoptosis in PDLSCs through mTOR signaling[155].
Surely, the critical role of ceRNA formed by circRNA and miRNA is indisputable. Circ_0138959 was identified as a functional sponge of miR-527 that directly targeted the apoptosis proteins caspase-5 to block the effect of apoptotic protein on cells, ultimately promoting the apoptosis[135]. Wenjuan Deng et al. found circ_0138959 overexpression inhibited osteogenic differentiation, promoting apoptosis and cytokine release by interacting with miR-495-3p and TRAF6, leading to devastating outcomes[154]. Circ_0097010 and circ_0138959 inhibition could block LPS-mediated apoptosis and reduce inflammation by forming ceRNA with miR-769-5P/KLF6 and miR-518a-5p/HDAC6 respectively[136,154]. Moreover, silencing of circ_0099630 absorbed miR-940 consequently increased the mRNA level of TRAF6, promoting cell viability and inhibiting apoptosis and inflammatory response in LPS-induced PDLCs[139]. Consistent with a bioinformatics analysis, has_circ_0099630 overexpression played destructive role in the osteogenic differentiation of human periodontal ligament fibroblasts probably through interacting with miR-182, miR-1200, miR-338, miR-576 and miR-623[140]. Those experiments all proved the potential mechanism of circRNAs in accelerating the development of periodontitis. -
At present, the incidence of periodontitis is extremely high, and its pathogenesis remain ambiguous. Many ncRNAs have been proved to be differentially expressed in periodontitis patients and considered as biomarkers to predict the prognosis and recurrence of periodontitis[115,132,141]. Currently, to enhance the therapeutic effects, researchers have been investigating to implant functional ncRNAs into animals through scaffold or adenovirus or directly inject into gingiva[80,89,142,160]. However, there are still critical problems we are facing: Firstly, the expression of ncRNAs is easily affected by external stimuli, which makes it difficult to exert the therapeutic effects. For example, miR-146a significantly increased the activity of ALP and the expression of OCN and Runx2 on day7 and day14 after LPS stimulation[161]. Contrarily, miR-146a suppressed the activity of ALP and inhibited osteogenic differentiation of PDLSCs under mechanical forces[162]. Secondly, periodontitis is a complex infection caused by multiple bacteria. Up to now, research has shown that bacterial pathogens could suppress host miRNA expression for their survival[163]. Whether interaction between periodontal pathogens influence the expression of ncRNAs and whether there are bacteria-related biomarkers that can be used to diagnose periodontitis remain unclarified. Thirdly, ncRNAs are differentially expressed in divergent periodontal tissues. For example, the expression of miR-199a-5p was up-regulated in salivary level and decreased in plasma level among patients with periodontitis[164]. The miR-146a upregulation and miR-155 downregulation occurred only in gingiva after LPS stimulation instead of dental pulp and periodontal ligament fibroblasts from the same donor[165]. These uncertainties in ncRNAs expression led to challenges including the specificity, delivery and tolerability of ncRNA-based therapies. More importantly, many studies only focused on in vitro experiment, especially lncRNAs and circRNAs, which is different from the actual in vivo environmental state at the time of onset.
However, the future applications of ncRNAs still have great potential in periodontitis. We believe that ncRNAs could be applied as biomarkers in diagnose and typing of periodontitis. Additionally, ncRNAs could also be used as indicators for the treatment decision in periodontitis. In periodontitis treatment, the functional ncRNAs that processed into tablets or thin films could combine with classic periodontal surgery to enhance the therapeutic effectiveness. Surly, for genetic-related periodontitis, it is also a promising choice to develop vaccines based on ncRNAs.
Altogether, research on ncRNAs in periodontitis contains not only challenges but also opportunities, more preclinical and clinical studies are deserved to be done in this field, contributing to elucidate the underlying mechanisms in periodontitis as early as possible.
The Dual Role of Non-coding RNAs in the Development of Periodontitis
doi: 10.3967/bes2023.079
- Received Date: 2022-10-26
- Accepted Date: 2023-04-18
-
Key words:
- Periodontitis /
- MicroRNA /
- Long non-coding RNA /
- Circular RNA /
- Non-coding RNA
Abstract: This review aims to sum up how Non-coding RNAs (ncRNAs) regulate the development of periodontitis and provides a new perspective for understanding the pathogenesis of periodontitis. We explored the ncRNA's dual role in the development of periodontitis by summarizing evidence from previous in vivo and in vitro studies as well as clinical samples. In our review, the downregulation of 18 miRNAs, 22 lncRNAs and 10 circRNAs demonstrates protective roles in periodontitis. In contrast, the expression of other 11 miRNAs, 7 lncRNAs and 6 circRNAs are upregulated in periodontitis, which promote the progression of periodontitis. These dysregulated ncRNAs exert their protective or destructive roles by mainly influencing cell proliferation, differentiation and apoptosis via cross-talking with various molecules or signaling pathways. Our findings suggested which and how ncRNAs promote or delay the progression of periodontitis, which may greatly contribute to diagnose and therapy development of periodontitis based on ncRNAs in the future.
Conceptualization and writing-original draft: XU Xi and HAN Ying Ying; Writing-review and editing: all authors; Supervision and final approval of manuscript to be published: HAN Ying Ying.| Citation: | XU Xi, LANG Guang Ping, CHEN Zhong Lan, WANG Jing Le, HAN Ying Ying. The Dual Role of Non-coding RNAs in the Development of Periodontitis[J]. Biomedical and Environmental Sciences, 2023, 36(8): 743-755. doi: 10.3967/bes2023.079 |







 Quick Links
Quick Links
 DownLoad:
DownLoad: