-
Cryptosporidium is a protozoan parasite belonging to the phylum Apicomplexa that causes self-limiting diarrhea in immunocompetent individuals, and it may also cause chronic and life-threatening diarrhea in those that are immunocompromised[1]. The two main routes of Cryptosporidium transmission are via water and food. At least 30 Cryptosporidium species have been confirmed, including C. andersoni, with more than 70 genotypes of undefined species. C. andersoni causes chronic infections in adult cattle and humans[2, 3]. Although over 100 chemotherapeutic and immunotherapeutic agents have been examined for anti-Cryptosporidium efficacy, Nitazoxanide [NTZ; N-(5-nitrothiazoly) salicylamide acetate] is the only medicine licensed in the USA and Central and South America to treat cryptosporidiosis in immunocompetent children and adults. However, this medicine is not effective in immunocompromised individuals. Furthermore, transplant recipients and individuals undergoing cancer chemotherapy are not encouraged to use NTZ to control infection because of its severe side effects. Risk of infection and absence of vaccine demand sustained efforts to develop new anti-Cryptosporidium medicines with fewer side effects for immunocompromised individuals. These medicines may originate from natural plant products.
Ginkgolic acids, extracted from Ginkgo biloba sarcotesta, are used in traditional Chinese medicine. Ginkgolic acids are a mixture of several 2-hydroxy-6-alkylbenzoic acids with straight alkyl chains. Many studies have investigated ginkgolic acids as possible treatments for parasites. For instance, ginkgolic acids significantly inhibit the growth of Toxoplasma gondii. We have reported that a mixture of ginkgolic acids is efficacious against C. andersoni in human adenocarcinoma (HCT-8) cell culture[4]. There are multiple ginkgolic acid monomers. The monomer GA has fifteen carbon atoms with the alkyl chain containing one carbon-carbon double bond (GA15:1), whereas the monomer GB has thirteen carbon atoms and a saturated hydrocarbon chain (GB13:0). However, the role of each component in the anti-Cryptosporidium activity of ginkgolic acids needs to be elucidated. In this regard, this study aimed to investigate the anti-Cryptosporidium activity of the pure ginkgolic acid monomers GA and GB in HCT-8 cell cultures. This study used a quantitative real-time PCR (RT-PCR)-based method to evaluate the anti-Cryptosporidium activities of these monomers in vitro in order to screen a new medicine to treat Cryptosporidium infections in animals and humans. A proliferation assay was also performed to determine the toxicity to cell viability. In our experiments, NTZ, which is generally considered as an effective medicine for cryptosporidiosis, was used as a positive control.
Ginkgolic acids are a mixture of different compounds extracted from G. biloba sarcotesta. The purified monomers GA and GB were purchased from Tauto Biotech Co., Ltd., Shanghai, China. The medicine NTZ was donated by Professor HE Guo Sheng of Shanghai Veterinary Research Institute, Chinese Academy of Agricultural Sciences, China. C. andersoni oocysts (Sakha 101 isolate) were originally obtained from a naturally infected calf with diarrhea. Before cells were infected, the oocysts were purified in a discontinuous sucrose gradient and infective sporozoites were released, following the procedures of Wu et al.[5]. HCT-8 cells were obtained from Shanghai Generay Biotechnology Co. Ltd., China. HCT-8 cells (1 × 104 cells/well) were seeded on 96-well cell culture plates for 24 or 48 h. Effects of GA, GB, GA + GB (1:1), and NTZ on cell proliferation were assayed using a cell counting kit (CCK-8; Dojindo Molecular Technologies, Japan) for proliferation assay, as described by Perry et al.[6]. The maximum safe concentration of each medicine was calculated by a probabilistic method[4].
HCT-8 cells (5 × 104 cells/well) were grown in 6-well culture plates in RPMI 1640 medium containing 10% fetal bovine serum (Hyclone Inc., USA) and infected with 1 × 103 infective sporozoites for 8 h. Next, the medium was replaced with fresh culture medium to remove noninfectious parasites, and the cells were then ready for further treatment with medicines. On the basis of the maximum safe concentrations of every medicine, the treated cells at 37 ℃ in an atmosphere containing 5% CO2 were harvested after 48 h to extract total DNA using a DNA Purification Kit (Generay Biotechnology Co., Ltd, Shanghai). By using the forward primer 5′-CACCTCCCAACCCTGAATGTC-3′ and the reverse primer 5′-TGCTGGCAAATACTGGA-3′ of C. andersoni oocyst wall protein (COWP) gene (GenBank no. AB514044.1), we cloned the COWP-pGEM-T Easy vector (Promega, Madison, WI, USA) as a standard curve. The copy number of the COWP gene in each sample was determined for three times by RT-PCR (ABI PRISM 7, 000 sequence detection system, Singapore) and expressed as mean ± SD. Statistical analysis was conducted by analysis of variance followed by Fisher's least significant difference test using the GraphPad Prism 7 software. P-values < 0.05 were considered significant compared with the control.
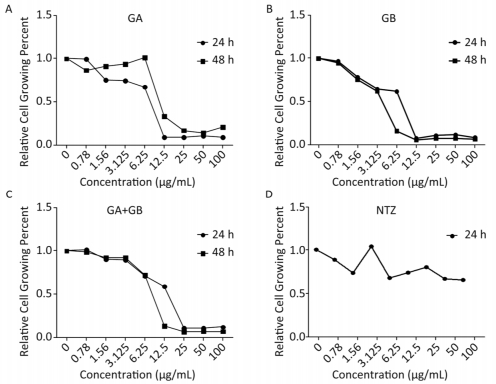
Because medicines may influence the proliferation of host cells, we conducted a cell proliferation assay to determine the maximum safe concentration of the study medicines. In parallel, we used the anti-Cryptosporidium medicine NTZ as a control. We observed that the proliferation of HCT-8 cells was dependent on the dose of the test medicines (Figure 1). The maximum safe concentrations for both GA and GA+GB were 3.125 µg/mL for 24 h or 48 h, whereas those of GB were 2.500 and 2.000 µg/mL for 24 and 48 h, respectively. Therefore, we adopted 3.125 µg/mL as the maximum concentration of all medicines tested in this study.
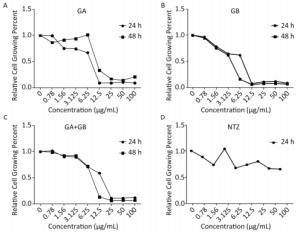
Figure 1. Proliferation of HCT-8 cells was affected by treatment with different concentrations of medicines for 24 or 48 h. A, B, C, and D show the effects of GA, GB, GA+GB, and NTZ (as s positive control), respectively, on the growth of HCT-8 cells. Data are representatives of at least three independent experiments.
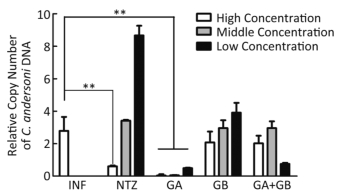
HCT-8 cells were infected with C. andersoni for 8 h and then treated with medicines for 48 h. Next, the copy number of the C. andersoni COWP gene was quantified by RT-PCR to determine parasite abundance. The GA and GB concentrations tested were 3.125 (high concentration), 1.56 (middle concentration), and 0.78 µg/mL (low concentration) (Figure 2). The results showed that GA at each concentration was efficacious in decreasing the copy number of the COWP gene relative to infected cells compared with the no-medicine group (all P < 0.01). GB at high concentration decreased the copy number of the COWP gene compared with the no-medicine group, but the decrease was not significant. NTZ at high concentration, as a positive control, significantly decreased the parasite gene copy number compared with that of the infected group with on-medicine treatment (P < 0.05).
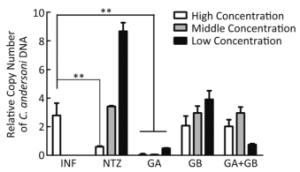
Figure 2. Anti-Cryptosporidium efficacy of the tested medicines in vitro. HCT-8 cells were infected with C. andersoni sporozoites for 8 h and then treated with medicines for 48 h. The efficacy of the medicines on the copy number of the C. andersoni oocyst wall protein (COWP) gene was compared with that of the infected group with no-medicine treatment. Medicine concentrations were 3.125 (high), 1.56 (medium), and 0.78 µg/mL (low), which were chosen on the basis of their safe range. Values are expressed as means. Data are representatives of at least three independent experiments. *P < 0.05, **P < 0.01 compared with the no-medicine group. INF, infection.
Crude ginkgolic acids extracted from G. biloba sarcotesta contain different types of monomers. Preliminary studies showed that crude ginkgolic acids inhibit the growth of C. andersoni in vitro[4]. To explore the relative efficacy of ginkgolic acid monomers, we chose GA (15:1) and GB (13:0) on the basis of their availability, and observed their anti- Cryptosporidium activities in vitro using HCT-8 cells.
As a reference, previous studies showed that NTZ at 10 µg/mL significantly reduces the growth of C. parvum and C. andersoni in a cell culture[4]. In this study, the same effect was observed against the proliferation of C. andersoni in HCT-8 cell culture. In particular, we observed that NTZ at 3.125 µg/mL significantly inhibited the growth of C. andersoni. Thus, different Cryptosporidium species have different sensitivities to this medicine (unpublished data).
Ginkgolic acid monomers have been the subject of intense interest in medicinal chemistry because of their various biological activities[7-9]. In our study, GB was less toxic than GA to the growth of host cells, whereas an equal proportion mixture of the two monomers (GA+GB), at 3.125 µg/mL, was not highly toxic to the growth of HCT-8 cells. A significant anti-Cryptosporidium efficacy was shown by GA alone at high, medium, and low concentrations (P < 0.01). This effect could be attributed to our use of purified ginkgolic acid monomers instead of the mixtures of unpurified ginkgolic acids used in previous studies[4].
GB at 3.125 µg/mL inhibited the growth of host cells, whereas GB at the other two concentrations, which were below the safe dose for host cell growth, exerted no inhibitory effect against Cryptosporidium in host cells. The mixture of the two monomers (GA+GB) showed a mild anti-Cryptosporidium efficacy, which was not statistically significant at low concentrations. The anti-Cryptosporidium activity of GA may have been damped when the two ginkgolic acid monomers were combined. These results indicated that there could be a structure-activity relationship in the mode of action of each monomer.
Our data indicated that the carbon chain length of the ginkgolic acid monomer was the most important factor affecting anti-Cryptosporidium activity. We conjectured that higher membrane permeability was associated with longer alkyl chain of GA, which made medicine delivery more effective. We previously reported that ginkgolic acids significantly inhibit the growth of T. gondii[8], an important apicomplexan parasite, and postulated that ginkgolic acids could interfere with protein and DNA synthesis in the parasite.
We adopted an in vitro culture technique to study C. andersoni infection because of the difficulty in infecting mice with oocysts obtained from cattle. In addition, other reports showed that it is impossible to transmit bovine-derived oocysts to neonatal BALB/c mice, adult BALB/c mice, SCID mice, guinea pigs, rats, rabbits, or goats[9]. The in vitro method has proved reliable for studying potentially useful therapeutic agents against C. andersoni[5] and C. parvum[10]. In this study, the development of the parasite was evaluated by quantifying copies of the COWP gene using RT-PCR[4] because this method provides more accurate and objective results compared with counting the parasites under a microscope, which could be subjective. Moreover, our in vitro technique was a convenient and relatively inexpensive method for medicine screening.
In conclusion, we showed that the ginkgolic acid monomer GA exerted a more potent anti-Cryptosporidiumactivity compared with GB, and it could be developed as a potent medicine for treating Cryptosporidium infections. To further understand the role of the substitution degree of the alkyl groups in the activity of ginkgolic acids against Cryptosporidium, studies on other ginkgolic acid monomers are needed.
No conflict of interest to declare.
CAO Jian Ping, SHEN Yu Juan, and Chidiebere E. UGWU conceived and designed the study, analyzed the data, and wrote and revised the manuscript. JIANG Yan Yan performed the experiments and revised the manuscript. Chidiebere E. UGWU, WU Liang, XU Yu Xin, YIN Jian Hai, DUAN Li Ping, CHEN Sheng Xia, LIU Hua, PAN Wei, and QUAN Hong performed the experiments. CAO Jian Ping and SHEN Yu Juan contributed reagents and materials.
In vitro Screening of Ginkgolic Acids for Antiparasitic Activity against Cryptosporidium andersoni
doi: 10.3967/bes2019.040
the National Natural Science Foundation of China 81772225
the National Natural Science Foundation of China 81371841
the Chinese Special Program for Scientific Research of Public Health 201502021
the Fourth Round of Three-Year Public Health Action Plan of Shanghai, China 15GWZK0101
- Received Date: 2018-01-04
- Accepted Date: 2018-09-25
| Citation: | Chidiebere E. UGWU, JIANG Yan Yan, WU Liang, XU Yu Xin, YIN Jian Hai, DUAN Li Ping, CHEN Sheng Xia, LIU Hua, PAN Wei, QUAN Hong, SHEN Yu Juan, CAO Jian Ping. In vitro Screening of Ginkgolic Acids for Antiparasitic Activity against Cryptosporidium andersoni[J]. Biomedical and Environmental Sciences, 2019, 32(4): 300-303. doi: 10.3967/bes2019.040 |







 Quick Links
Quick Links
 DownLoad:
DownLoad: