-
The threat of bacteria and pathogens to human health and environmental safety has become a major public concern. In the fight against pathogenic bacteria, much effort has been expended on designing new antibacterial agents[1]. To date, various materials such as metals, oxides, N-halamines, polymers, chitosan, and graphene have been used as antibacterial agents[2, 3]. Among them, silver-based materials in which Ag+ ions are generally the active species exhibit a highly bactericidal effect on a broad range of bacteria[4]. Recently, silver halides have been recognized as potent antimicrobial agents, with the release of Ag+ ions being a crucial aspect of their antibacterial function[5]. However, a challenge associated with the application of silver halides is their instability, and much work has been devoted to embedding them in a silica/polymer matrix to enhance their stability.
N-chloramines are considered to be the most powerful of the biocidal agents due to their chlorine oxidation state[6]. N-chloramines are known to have long-term stability, high durability, and regenerability, which make them attractive as disinfectants for food, water, and textiles. Recently, much effort has been devoted to synthesizing hybrid nanoparticles (NPs) as a means achieving improved antibacterial performance[7]. In this paper, we present a one-pot procedure for preparing AgCl embedded in silica (AgCl@S) NPs, in which we graft 1-allylhydantoin (AH) as a precursor onto the surface of AgCl@S (AgCl@S/AH) NPs through radical polymerization followed by chlorination to obtain an N-chloramine-decorated AgCl (AgCl@S/AHC) antibacterial agent.
The innovative aspect of this work is the synergistic antibacterial effect between AgCl and N-chloramine in the AgCl@S/AHC antibacterial agent. In this work, the cetyltrimethylammonium chloride (CTAC), silver nitrate (AgNO3), tetraethylorthosilicate (TEOS), hypochlorite (NaClO), potassium persulfate (KPS), 1-allylhydantoin (AH), and methyl methacrylate (MMA) used in the experiments were of analytical grade and used without any further purification. The nutrient broth (NB, BR) and nutrient agar (NA, BR) were purchased from the Beijing Aoboxing Biotech Company (Beijing, China). Aqueous solutions were prepared with double distilled water.
The preparation of the AgCl@S/AHC sample included preparation of AgCl cores, coating with the silica shell, grafting the AH, and chlorination (Supplementary Figure S1 available in www.besjournal.com). The AgCl cores were prepared using the precipitation method. In a typical synthesis, we dissolved CTAC (0.18 mmol) in water (97.0 mL) while vigorously stirring, then added AgNO3 solution (0.5 mmol) and NaCl solution (0.5 mmol). After reacting at 40 °C for 3 h, AgCl cores were obtained. Then, we used HCl solution (0.1 mol/L) to adjust the pH value of the AgCl solution to 3.0 and added 2.0 mL of TEOS dropwise to coat the AgCl cores (AgCl@S). Finally, we performed the conjugation of AH and chlorination according to the protocols described in our previous work[7].
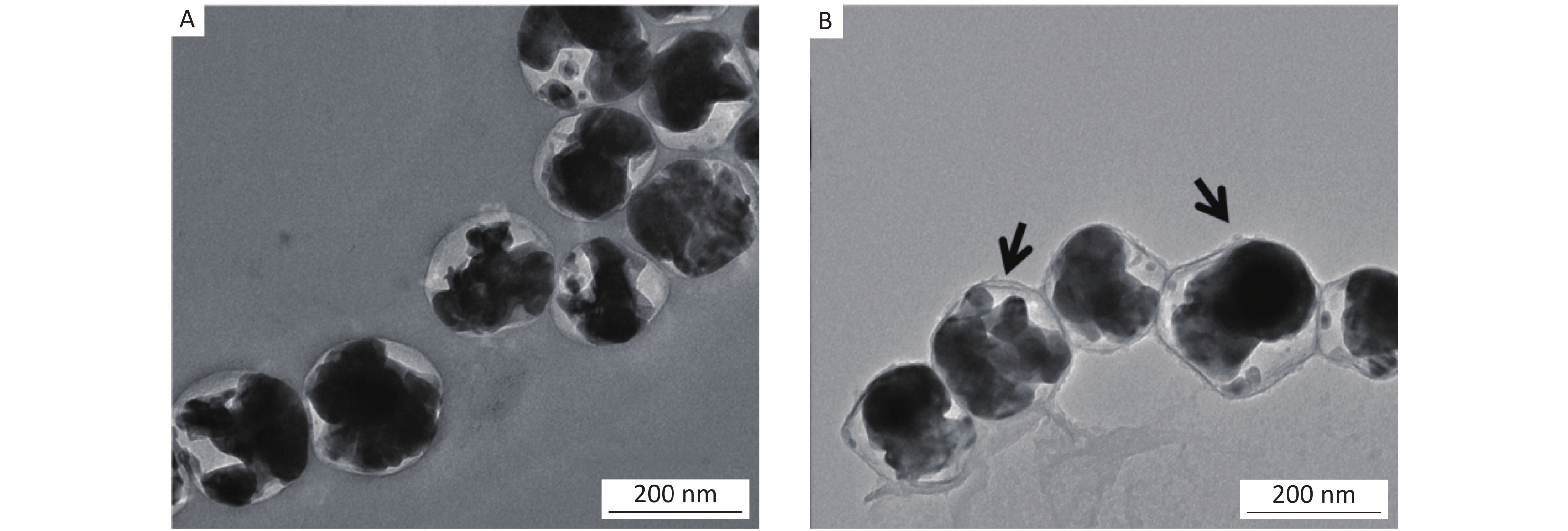
The morphologies of the prepared samples were examined by transmission electron microscopy (TEM, JEOL-2100) at an acceleration voltage of 200 kV. Figure 1 shows TEM images of the prepared AgCl@S and AgCl@S/AH samples. The AgCl@S sample had a well-defined spherical shape and narrow size distribution of 180–200 nm. The core/shell structure was clearly distinguishable and each nanosphere contained several AgCl cores with a thin silica shell (Figure 1A). After coating the AH, there was no obvious change in its particle size or morphology, but the sample had a somewhat rough surface or double layer structure (Figure 1B), which indicates the successful introduction of AH.
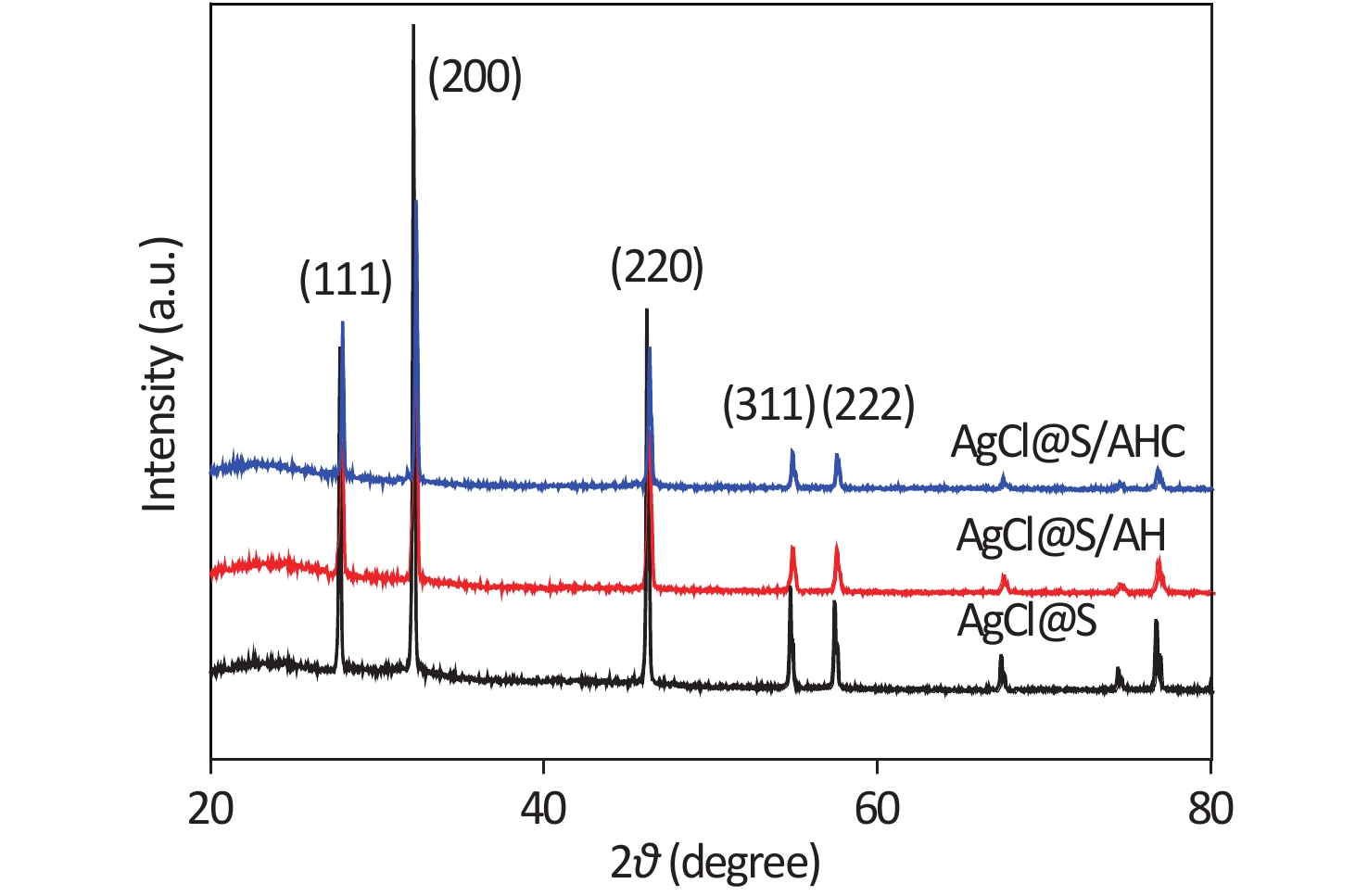
Supplementary Figure S2 (available in www.besjournal.com) shows the X-ray diffraction (XRD) patterns of the AgCl@S and AgCl@S/AH samples. The diffraction peaks at 2θ of 27.7°, 32.1°, 46.2°, 54.7°, and 57.4° are attributed to the (111), (200), (220), (311), and (222) reflections, respectively, from the cubic phase of AgCl (JCPDS: 31-1238)[8]. In addition, a small and broad amorphous peak can be observed in the 2θ range of 20°–30°, which may be due to the amorphous silica-coating layer. After coating the AH and chlorination, all of the diffraction peaks could be clearly identified, which suggests that the silica shell effectively enhanced the stability of the AgCl cores.
Supplementary Figure S3 (available in www.besjournal.com) shows the FT-IR spectra of the AgCl@S, AgCl@S/AH, and AgCl@S/AHC samples. For the three samples, the typical absorption band at 967 cm−1 is ascribed to the stretching vibrations of Si–OH, and the bands at 1,078 cm−1 and 802 cm−1 correspond to the stretching vibrations of the Si–O–Si frameworks and indicate the presence of the silica shell.[9] For the AgCl@S/AH sample, a stretching vibration of C-N occurs at 1,100–1,250 cm−1 and that of C=O at 1,727 cm−1, which indicates the presence of hydantoin[10]. For the AgCl@S/AHC sample, there is a peak at 743 cm−1, which is ascribed to the N-Cl bond[6]. The FTIR analysis further suggested that AH was successfully conjugated on the surface of AgCl@S.
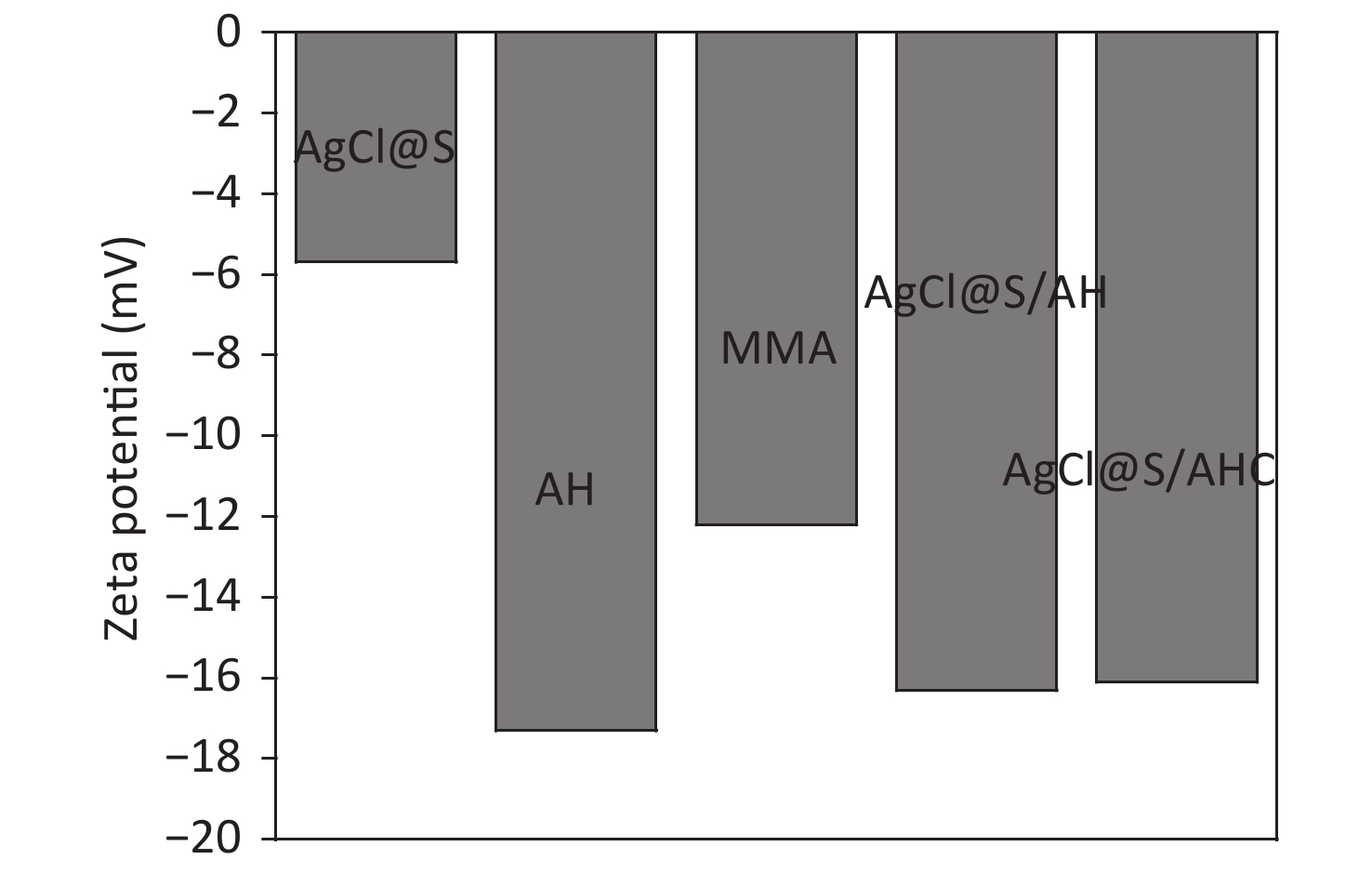
Thermogravimetric (TG) analysis was employed to estimate the mass ratios of the inorganic and organic components in the hybrid sample. Supplementary Figure S4 (available in www.besjournal.com) shows the TG curves of the AgCl@S and AgCl@S/AH samples. The AgCl@S sample exhibits a successive process of loss of mass caused by the evaporation of adsorbed water and the dehydroxylation of Si–OH. For the AgCl@S/AH sample, the mass-loss process involved two regions. The mass loss (25%) in the range of 150–400 °C is mainly attributed to the thermal decomposition of the organic moiety, indicating that the organic content was about 25 wt%. The subsequent mass loss at temperatures above 400 °C is attributed to the dehydroxylation of silanol. Supplementary Figure S5 (available in www.besjournal.com) shows the zeta potentials of the hybrid samples. The potential value of the AgCl@S sample was −5.7 mV due to the surface silanol groups. After the AH had been coated, the potential value decreased from −5.7 mV to −16.3 mV, indicating the conjugation of the AH precursor.
Gram-positive S. aureus (ATCC 25923) and gram-negative E. coli (ATCC 25922) were selected as indicators for our antibacterial experiments. Prior to the experiments, all glassware and culture media were sterilized in an autoclave. The antibacterial activity of AgCl@S/AHC was evaluated by its disinfection rate according to the procedure described below.
Using the serial dilution method, the AgCl@S/AHC sample was dispersed in sterilized distilled water to produce serial suspensions with different concentrations. Then, we mixed 50 μL of the bacterial suspension (108 CFU/mL) with 450 μL of the sample suspension and incubated it under constant shaking at 37 °C. To study the bacterial reduction with contact time, at predetermined contact times, excessive Na2S2O3 solution (0.03 wt%) was used to remove the Cl+ ions and terminate the sterilization process. Then, we extracted 20 μL of the suspension and spread it evenly on the surface of the agar in a Petri dish and incubated it at 37 °C for 24 h. Lastly, the number of the surviving colonies was counted to determine the bacteria concentration.
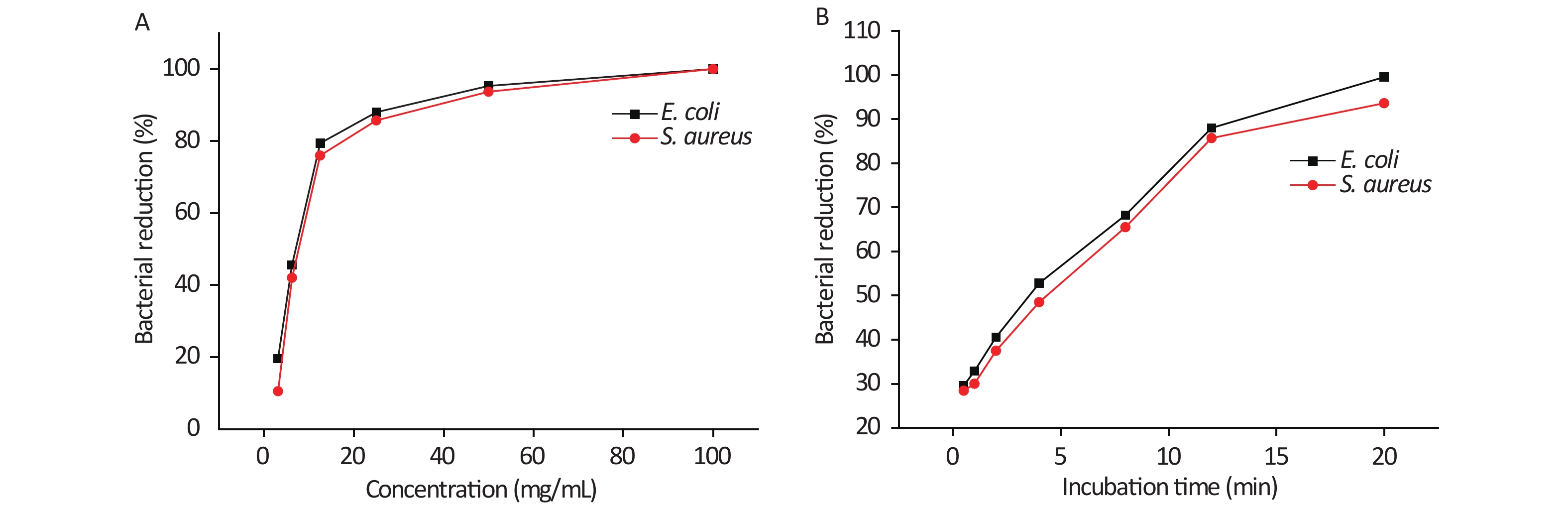
Figure 2A shows the concentration–bacterial reduction curves of AgCl@S/AHC against both E. coli and S. aureus, in which we can see that the bacterial reduction increases with increases in the sample concentration. When the AgCl@S/AHC concentration was 100 mg/mL, all bacteria were killed after 12 h of treatment. Figure 2B shows the time–bacterial reduction curves of AgCl@S/AHC against both strains, in which it is clear that the number of surviving colonies decreases with increases in the contact time. For example, the AgCl@S/AHC sample inactivated 32.89% of the E. coli after 1 h. After 20 h of contact, the sample had killed all the bacteria. Generally, N-chloramines achieve rapid sterilization and Ag-based materials have a lasting inhibition effect. For the two strains, the hybrid sample showed a similar tendency, which suggests a synergistic antibacterial effect between AgCl and N-chloramine.

Figure 2. Bactericidal effect of AgCl@S/AHC at different concentrations for 12 h (A) and at different times with 25.00 mg/mL (B).
To better understand this synergistic effect, Table 1 lists antibacterial data of the three samples for different contact times. It can be seen that the AgCl@S sample was able to inactivate both strains and its degree of bacterial reduction increased with the contact time. For example, the AgCl@S sample inactivated about 0.7% of the E. coli and 0.2% of the S. aureus after 0.5 h. When the contact time was 8 h, the AgCl@S sample killed 18.7% of the E. coli and 16.0% of the S. aureus. After 20 h of contact, the sample had killed about 50% of the E. coli and 44.7% of the S. aureus. For the AgCl@S sample, Ag+ ions are believed to be the active component and the sample exhibited a sustained biocidal effect due to the continuous release of Ag+ ions. It is clear that AgCl@S and AgCl@S/AH exhibit similar long-lasting antibacterial activity. Significantly, AgCl@S/AHC shows drastic and long-lasting antibacterial activity against both strains. For example, AgCl@S/AHC was able to kill 29.6% of the E. coli and 28.4% of the S. aureus after 0.5 h, which is more effective than the other samples. After 20 h of contact, the sample had killed nearly 100% of both strains and its strong biocidal effect is related to the synergistic antibacterial effect of the Ag+ ions and oxidative chlorine.
Contact time (h) Bacterial reduction % (E. coli) Bacterial reduction % (S. aureus) AgCl@S AgCl@S/AH AgCl@S/AHC AgCl@S AgCl@S/AH AgCl@S/AHC 0.5 0.7 1.1 29.6 0.2 1.8 28.4 1 2.7 2.4 32.8 1.8 2.3 30.0 2 6.4 7.8 40.5 5.5 7.6 37.5 4 9.8 10.0 52.8 7.2 10.1 48.5 8 18.7 17.5 68.2 16.0 19.7 65.5 12 32.7 31.9 87.9 31.5 32.7 85.6 16 44.1 45.2 99.5 42.1 42.8 93.6 20 49.3 51.3 100.0 44.7 45.7 100.0 Note. Initial E. coli and S. aureus concentrations were 108 CFU/mL. The sample concentration was 25.00 mg/mL. Table 1. Bacterial concentrations of E. coli and S. aureus treated for different contact times
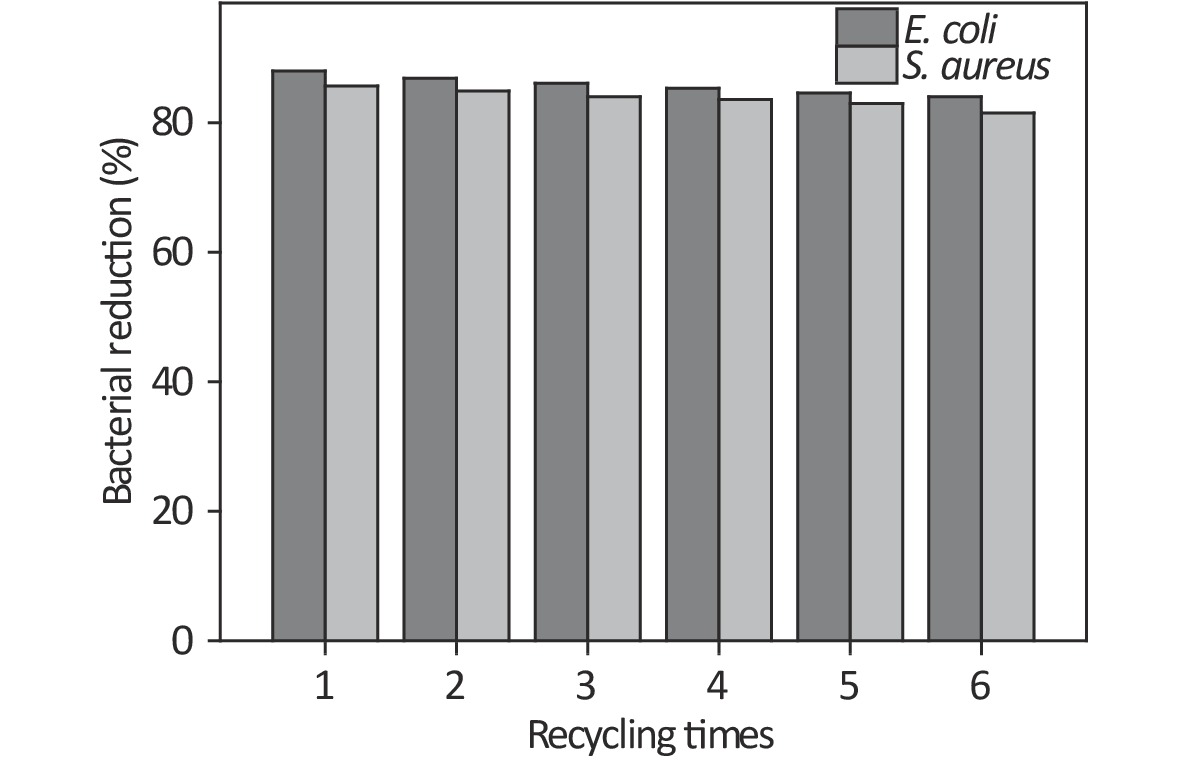
N-chloramine is a regenerable antibacterial agent that can revert to its precursor state upon making contact with a microorganism, and oxidative chlorine can be regenerated by chlorination. To achieve regeneration, the AgCl@S/AHC sample was treated with Na2S2O3 solution to remove all Cl+ ions, and this treated AgCl@S/AHC sample was then dispersed into HClO solution (10%) for chlorination at room temperature for 1 h. Lastly, the recovered AgCl@S/AHC sample was collected, washed, and dried. Supplementary Figure S6 (available in www.besjournal.com) shows the antibacterial activity of the recovered AgCl@S/AHC sample. For the recyclable assay, the concentration of the recovered AgCl@S/AHC sample was 25 mg/mL, the treatment time with the bacteria suspension was 2 h, and the bacterial concentration was determined by the plate culture count method. It is clear that the antibacterial performance of AgCl@S/AHC remained stable even after six experimental cycles. In addition, the morphologies of the healthy and treated bacteria were examined by SEM. Supplementary Figure S7 (available in www.besjournal.com) shows SEM images of healthy and treated E. coli and S. aureus. It can be seen that the healthy E. coli and S. aureus exhibit integrated and regular morphologies, whereas the cell walls of the E. coli and S. aureus treated with the AgCl@S and AgCl@S/AHC samples appear to be damaged and disorganized. These morphological changes indicate the occurrence of interaction between the hybrid sample and the bacterial cells.

Figure S6. Antibacterial activity of AgCl@S/AHC against E. coli and S. aureus during six antibacterial cycles.

Figure S7. SEM images of E. coli and S. aureus before and after being treated with samples, arrows represent damaged bacteria.
In conclusion, the AgCl@S/AHC sample exhibited a rapid and sustained antibacterial effect against E. coli and S. aureus due to the synergistic effect of Ag+ and oxidative chlorine. We can conclude that AgCl@S/AHC exhibits a rechargeable antibacterial performance and its antibacterial activity remains stable even after six experimental cycles.
All the authors approved the final manuscript and have no conflicts of interest to declare.
Preparation of N-chloramine-Decorated AgCl Nanoparticles with Enhanced Bactericidal Activity
doi: 10.3967/bes2020.095
- Received Date: 2019-12-12
- Accepted Date: 2020-06-02
| Citation: | XU Jia Rong, ZHAO Yan Bao. Preparation of N-chloramine-Decorated AgCl Nanoparticles with Enhanced Bactericidal Activity[J]. Biomedical and Environmental Sciences, 2020, 33(9): 723-726. doi: 10.3967/bes2020.095 |














 Quick Links
Quick Links
 DownLoad:
DownLoad: