-
Obesity is a major condition characterized by abnormal or excessive fat accumulation, leading to an increased risk of type 2 diabetes, cardiovascular diseases, and cancer[1-3]. In recent decades, gut microbiota has been investigated in various diseases, including obesity. Evidence has shown that gut dysbiosis, which usually manifests as decreased bacterial diversity[4], gene richness[5], and a lower ratio of Bacteroidetes to Firmicutes[6], contributes to the pathogenesis of obesity and its associated metabolic disorders[7,8]. Transplanting gut microbiota from obese co-twins could increase body mass and adiposity in germ-free mice compared to gut microbiota from lean co-twins[9], suggesting that the gut microbiota is directly involved in the development of obesity. However, most studies have focused on the role of bacteria in obesity without further analysis of the non-bacterial components of the microbiota. In addition to bacteria, viruses, fungi, and archaea are important components of the gut microbiota; however, knowledge about them is relatively limited. Recent evidence has shown that the gut virome and fungi play a role in obesity[10,11]. However, the role of Archaea in obesity remains unexplored; thus, it is important to investigate their roles in the pathogenesis of obesity.
Among the diverse microbial inhabitants of the gut, archaea are largely unexplored compared with other components. Recently, Archaea have garnered increasing attention for their potential contribution to host metabolism and health. Archaea are a group of single-celled prokaryotic organisms with cellular characteristics distinct from those of bacteria, such as possessing three RNA polymerases, lacking peptidoglycans, and having membranes that enclose lipids with hydrocarbons rather than fatty acids[12]. Several archaeal species have been successfully isolated from various sites on the human body, including the skin, oral cavity, respiratory tract, gastrointestinal tract, and vagina, highlighting their widespread presence and potential importance in human health[13]. Although there is no direct evidence that specific archaea can cause or promote human diseases, studies have shown that gut archaea participate in methanogenesis, heavy metal transformation, trimethylamine metabolism, and immune modulation[14]. For example, alterations in the composition of the gut archaea have been observed during colorectal tumorigenesis, as evidenced by the enrichment of halophiles and depletion of methanogens in patients with colorectal cancer[15]. Moreover, a recent study reported that methanogenic archaea in the lower gastrointestinal tract play important metabolic roles by converting bacterial end-products into methane, correlating with obesity and certain cancers[16]. Additionally, Archaea can interact with bacteria, thus optimizing the metabolism of fermentative bacteria in the gut[17]. Be that as it may, the role of archaea in metabolic activities and interactions within the gut ecosystem remains poorly under-investigated, particularly in the context of obesity and weight loss interventions.
Bariatric surgery, an effective therapeutic option for severe obesity, can induce profound alterations in the gut microbial communities[18,19]. However, the impact of bariatric surgery on the gut archaea and its implications for metabolic health remain unknown, underscoring the need for comprehensive investigations to elucidate the dynamics of the gut archaea in obese patients undergoing bariatric surgery and their potential role in metabolic recovery post-surgery.
In this study, we analyzed the composition of gut archaea using fecal shotgun metagenomics and assessed alterations in the gut archaeome, bacteriome, and archaeal-bacterial correlations between obese and lean subjects. Next, we determined the compositional alterations of archaea in obese individuals, along with decreased correlations with gut bacteria, and further validated them in two additional cohorts. Moreover, we found that the inter-kingdom interaction between archaea and bacteria increased in obese patients after bariatric surgery.
-
Shotgun metagenomic sequencing data were retrieved from three published cohorts comprising 191 obese participants and 184 lean controls. The study by Liu et al.[20], which had the largest sample size and included obese patients undergoing bariatric surgery, was designated the discovery cohort. The bariatric surgery subgroup (n = 23) represented a subset of obese participants from cohort 1, with longitudinal samples collected before and 3 months after surgery. The other two studies conducted by Nie et al.[21] and Karlsson et al.[22] were designated as the validation cohorts. Across all three cohorts, obesity was consistently defined as BMI ≥ 30 kg/m2, ensuring comparability of the obese phenotype. For the two Chinese cohorts (PRJEB12123 and PRJNA597839), the original clinical protocols used Asian/Chinese BMI classifications, in which BMI 18.5–23.9 kg/m2 is considered the healthy range. For the European cohort PRJEB1786, which was primarily defined by glucose tolerance status, the WHO/European BMI classification was applied (healthy range BMI < 25 kg/m2). Thus, 22 of the 49 controls had a BMI between 23 and 25 kg/m2, which is within the normal weight range for European populations and consistent with the original study design. The detailed cohort characteristics are summarized in supplementary table S1.
-
Metadata and sequencing reads were obtained from the National Center for Biotechnology Information Sequence Read Archive (NCBI SRA) database (ncbi.nlm.nih.gov/bioproject/) under the BioProject identifiers PRJEB12123, PRJNA597839, and PRJEB1786. Raw reads were processed according to the following steps: first, low-quality reads were filtered out, and adapters were trimmed using Fastp (version 0.20.0) with default parameters[23]; second, human reads were removed using KneadData (version 0.11.0, github.com/biobakery/kneaddata). Reads that did not align with the human genome and that passed quality controls were retained as clean reads. Subsequently, clean reads were subjected to taxonomic annotations. Read counts were determined using Kraken2 (version 2.0)[24], which was implemented in conjunction with a custom bacterial and archaeal database (RefSeq bacterial and archaeal genomes version 206 were downloaded from NCBI in June 2021). Various Linux tools, including Singularity (version 4.1.0)[25] and the GNU Parallel software (version 4.3.1)[26] were used for data processing.
-
The read count and metadata tables were imported into PhyloSeq (version 1.44.0) in R (version 4.3.1). Samples with archaeal read counts of less than 1,000 were discarded for downstream analysis. The read counts were transformed into a relative abundance table for downstream analysis. Linear discriminant analysis Effect Size (LEfSe) was used to identify the taxa that differentiated between the groups. Taxa with an LDA (Linear Discriminant Analysis (LDA) score greater than 2 were considered significant. Archaeal genera were ranked using the LDA score and used as candidate markers for subsequent discriminative analyses.
-
Alpha and beta diversity indices were calculated using the R package vegan (version 2.6.4)[27]. Two-tailed Wilcoxon’s rank sum test was used to determine statistically significant differences between the two groups. Permutational multivariate analysis of variance (PERMANOVA) was performed for Bray-Curtis dissimilarity. To evaluate the discriminative potential of the archaeal signatures, logistic regression models were fitted using the top n archaeal genera (n = 2–33) ranked by the LDA score, and model performance was summarized using the area under the receiver operating characteristic curve (AUC). For each marker set size, the models were fitted separately within each cohort to assess the cross-cohort consistency. The R package pROC (version 1.18.5)[28] was used to display and analyze the Receiver Operating Characteristic (ROC) curves. Correlations between bacteria and archaea were calculated using the sparse correlation for the compositional data (SparCC) algorithm[29]. Data processing and visualization were performed using the R package tidyverse (version 2.0.0)[30] and base R (version 4.3.1)[31], unless otherwise stated. Heatmaps were generated using the R package pheatmap (version 1.0.12)[32]. All statistical tests were conducted in R, and a p-value < 0.05 was considered statistically significant.
-
To validate the robustness of our database, we initially examined the differences in gut microbiota between obese subjects and lean controls. Regarding alpha diversity indices, a significant decrease was observed in the gut bacterial Chao1 index in obese subjects compared to that in lean controls (P < 0.001; Supplementary figure S1A). However, the Shannon diversity index did not differ significantly between the two groups (Supplementary figure S1B). We then compared beta diversity analysis employing Principal Coordinates Analysis (PCoA) by bacterial genus, which revealed that the two groups formed distinct clusters (R2 = 0.038, p = 0.001; Supplementary figure S1C). Further investigations into alterations in taxonomic distribution between the groups were conducted. Our findings align with those of previously published studies. Notably, obese subjects manifested an increased relative abundance of Firmicutes and a reduced abundance of Bacteroidetes compared to lean controls (Supplementary figure S1D). Additionally, significant shifts in bacterial taxa between the groups were observed, with 22 taxa increasing and 69 taxa decreasing in abundance in the obese group compared to the lean controls (Supplementary figure S1E). Overall, our analysis effectively replicates the established research findings that obesity is strongly associated with altered gut bacterial microbiota.
-
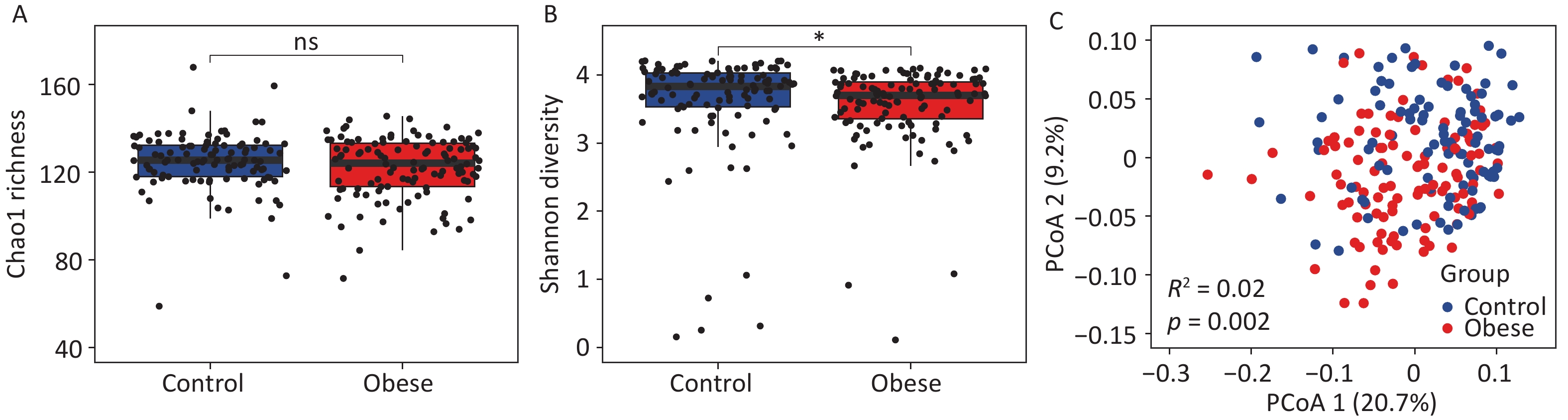
To investigate the changes in gut archaea associated with obesity, we analyzed the alpha diversity indices of obese subjects and lean controls. We observed no significant change in the gut archaeal richness, as measured by the Chao1 index (Figure 1A). However, gut archaeal diversity, as quantified by the Shannon index, was significantly lower in obese subjects than in lean controls (P < 0.05, Figure 1B). Furthermore, Principal Coordinate Analysis (PCoA) based on gut archaeal genera distinctly segregated obese subjects from lean controls into separate clusters, indicating distinct gut archaeal communities between the two groups (R2 = 0.02, P < 0.01, Figure 1C). Next, we compared the alpha diversity indices between groups using two additional independent cohorts. No significant changes in either Chao1 or Shannon indices were observed in the validation cohorts (Supplementary figure S2). These results suggested that the gut archaeal community in obese subjects differed markedly from that in lean controls. Additionally, heterogeneity between different cohorts significantly affected gut archaeal diversity.
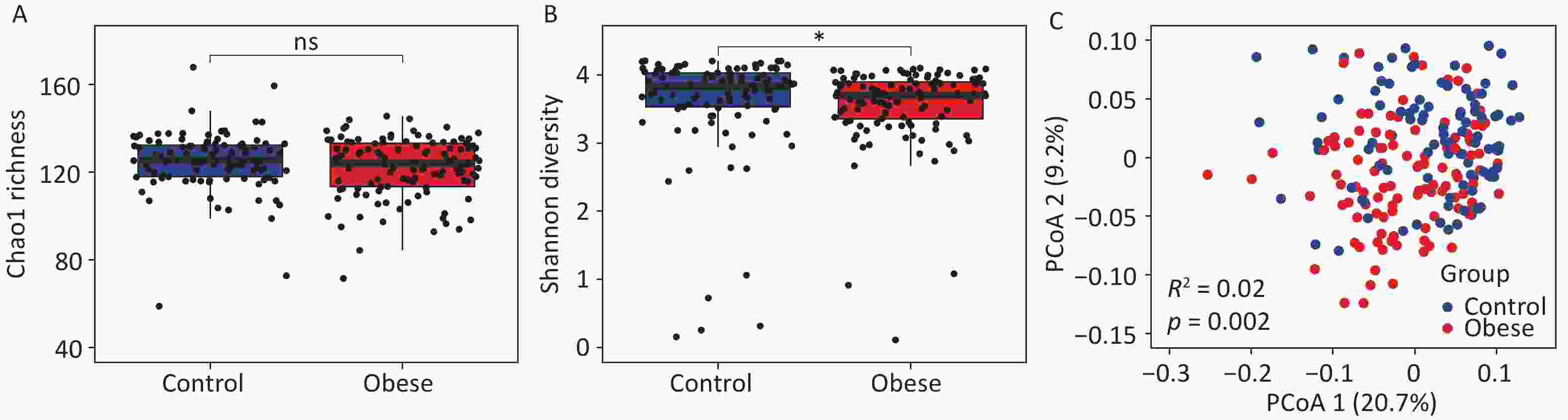
Figure 1. Alterations of gut archaea in obese subjects. (A) Chao1 richness and (B) Shannon diversity for gut archaeal genera between obese subjects and lean controls. Groups were compared using the Wilcoxon rank-sum test. (C) Principal coordinate analysis of gut archaeal genera separates obese subjects from lean controls. Groups were compared using PERMANOVA (permutational multivariate analysis of variance). * P < 0.05.
-
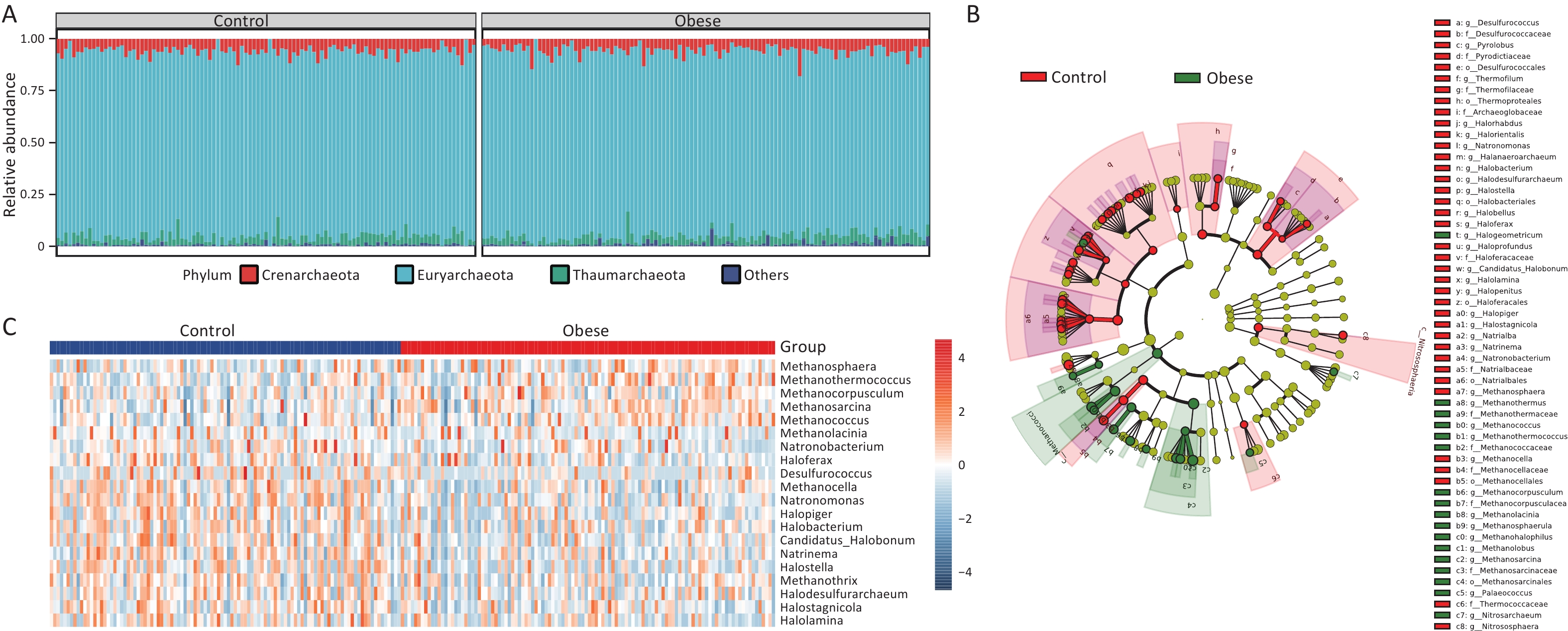
Subsequently, we explored the taxonomic distribution associated with obesity. Initially, we examined the gut archaeal profiles at the phylum level to discern alterations in the taxonomic composition between obese subjects and lean controls. At this level, Euryarchaeota emerged as the most abundant phylum in both obese and lean groups, followed by Crenarchaeota and Thaumarchaeota (Figure 2A). Subsequently, we identified 57 differential taxa between the two groups, spanning from the phylum to the species level, using Linear Discriminant Analysis Effect Size (LEfSe) (Figure 2B and Supplementary figure S3). This indicated significant shifts in the archaeal taxonomic composition of obese individuals relative to that of lean controls.
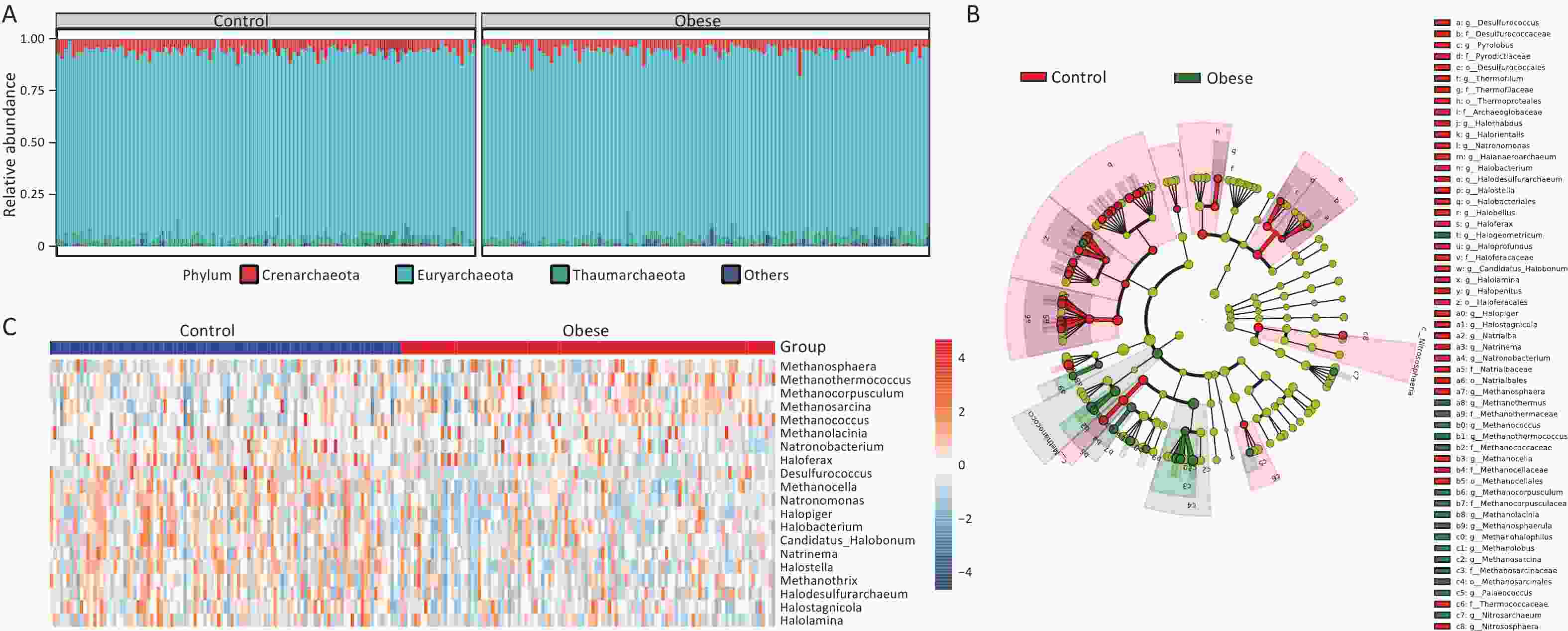
Figure 2. Gut archaeal taxonomic composition of obese subjects remarkably differs from lean controls. (A) Relative abundance of gut archaeal phyla. (B) Cladogram showing different archaeal compositions in the taxonomic lineage. (C) Heatmap showing log transform fold changes of the differential gut archaeal genera for obese subjects compared with lean controls in the discovery cohort. Groups were compared using LEfSe (Linear discriminant analysis Effect Size). The top 20 genera measured by LDA (linear discriminant analysis) effect size were selected for visualization.
To gain further insight into the alterations in taxonomic composition at more refined taxonomic levels, we subsequently explored the differential genera and species between obese subjects and lean controls. At the genus level, 11 genera were found to be increased in obese subjects compared to lean controls, whereas 22 genera were decreased (LDA > 2, Figure 3C and Supplementary figure S4). Notably, among the increased Archaea in obese subjects, Methanosarcina, Methanococcus, and Methanosphaera represented the taxa with the highest mean relative abundance. Conversely, Haloferax, Methanocella, and Natronomonas had the highest mean relative abundances among the genera that decreased in obese subjects. Additionally, 25 species, including Methanococcus maripaludis, Methanolacinia petrolearia, and Methanosarcina barkeri, were increased, whereas 61 species, including Methanobrevibacter smithii, Haloferax volcanii, and Halostagnicola Natrialbaceae archaeon XQ INN 246, were lower in obese subjects than in lean controls (LDA > 2, Supplementary figure S5). These findings collectively underscore the remarkable divergence in gut archaeal composition between obese subjects and lean controls.
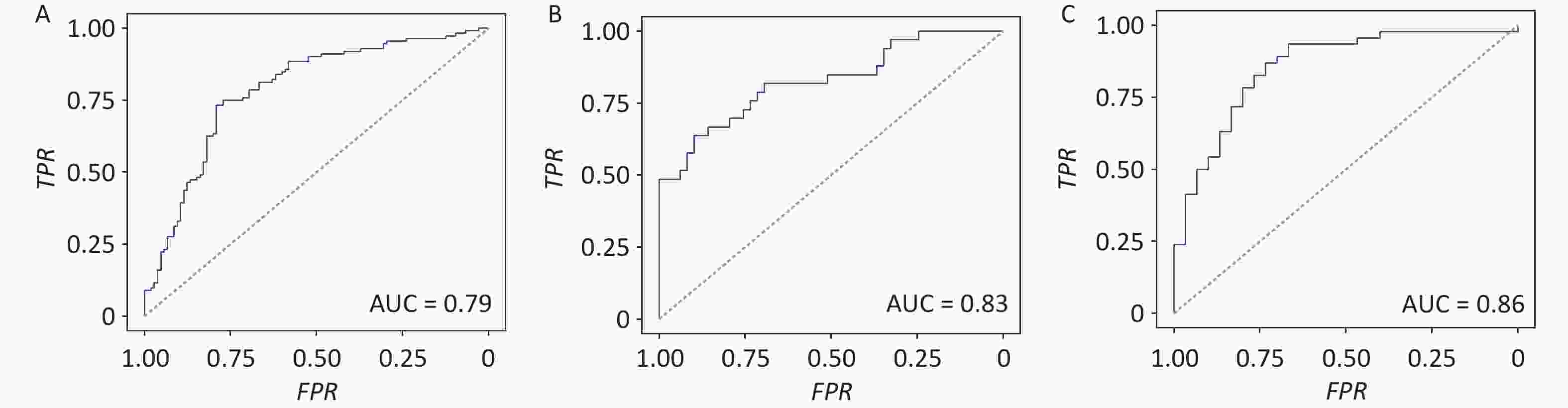
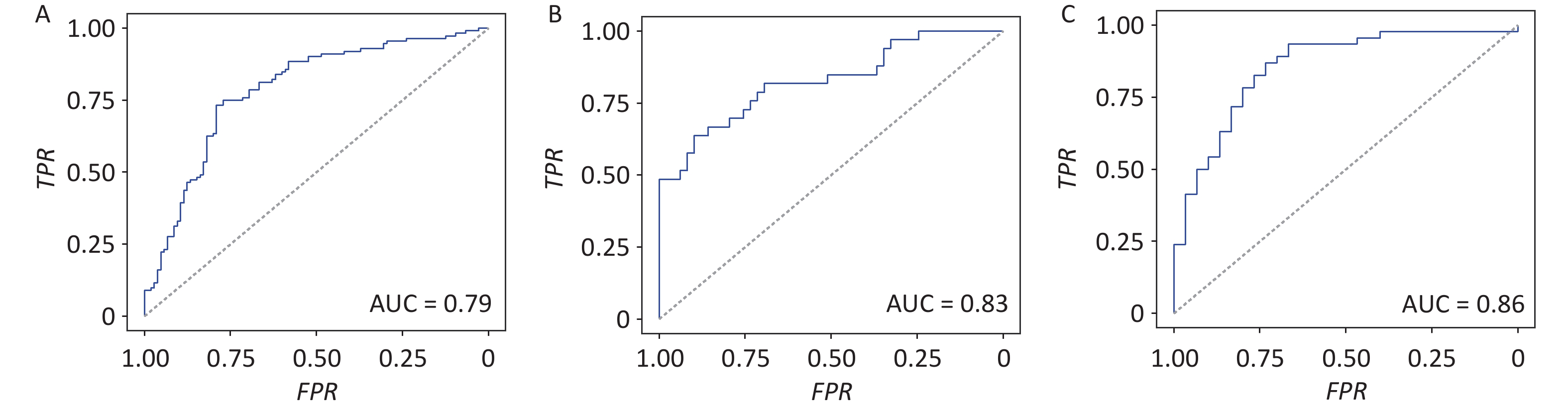
Figure 3. Gut archaeal genera classify obese subjects from lean controls. Predict model for (A) discovery cohort. Predict model for validated cohorts (B) PRJNA597839 and (C) PRJEB1786. Models were trained by the top 20 genera filtered by LDA values from the discovery cohort. Receiver operating characteristic (ROC) curves showing discrimination between obese and lean subjects based on 20 archaeal genera (AUC = 0.79, 0.83, and 0.86 in Cohorts 1–3, respectively).
-
To test the feasibility of using the identified gut archaeal alterations to distinguish obese patients, we developed a logistic regression model to differentiate between obese subjects and lean controls using differentially abundant archaeal markers. At the genus level, the top 20 genera exhibiting differential abundance between obese and lean groups were incorporated. These genera were chosen because model performance increased progressively with the inclusion of additional genera and reached a clear plateau at approximately 18–22 genera across all cohorts (Supplementary figure S6), indicating that 20 genera provided a stable and parsimonious marker set for downstream analyses. Among these, six genera were the most abundant in obese subjects: Methanosarcina (family Methanosarcinaceae), Methanococcus and Methanothermococcus (family Methanococcaceae), Methanosphaera (family Methanobacteriaceae), Methanolacinia (family Methanomicrobiaceae), and Methanocorpusculum (family Methanocorpusculaceae), all within the phylum Euryarchaeota. These taxa include methanogens with diverse methanogenic pathways that may mechanistically link archaeal activity to host energy harvesting and obesity. Our findings indicate that these top 20 differentially abundant archaeal genera can effectively discriminate obese subjects from lean controls with an AUC of 0.79 (95% CI 0.72–0.85, Supplementary figure 3A).
To assess the consistency of obesity-related gut archaea across different populations, the top 20 differential archaeal genera identified in the discovery cohort were applied to two additional cohorts. Cohort-specific logistic regression models were fitted using the relative abundances of these differential genera in two independent cohorts. We achieved an AUC of 0.83 (95% CI 0.73–0.92) and 0.86 (95% CI 0.77–0.94) for these two cohorts, respectively (Figure 3B and 3C). Additionally, a similar logistic regression model was fitted to gut archaeal species. We found that the prediction model at the species level achieved AUCs of 0.87, 0.91, and 0.88 for the discovery cohort and the two independent cohorts, respectively (Supplementary figure S7).
Taken together, our findings emphasize the association between obesity and changes in gut archaea across various cohorts, indicating that archaeal composition plays a vital role in the obesity-related gut microbiome. Methanogenic archaea may be implicated in the development of obesity.
-
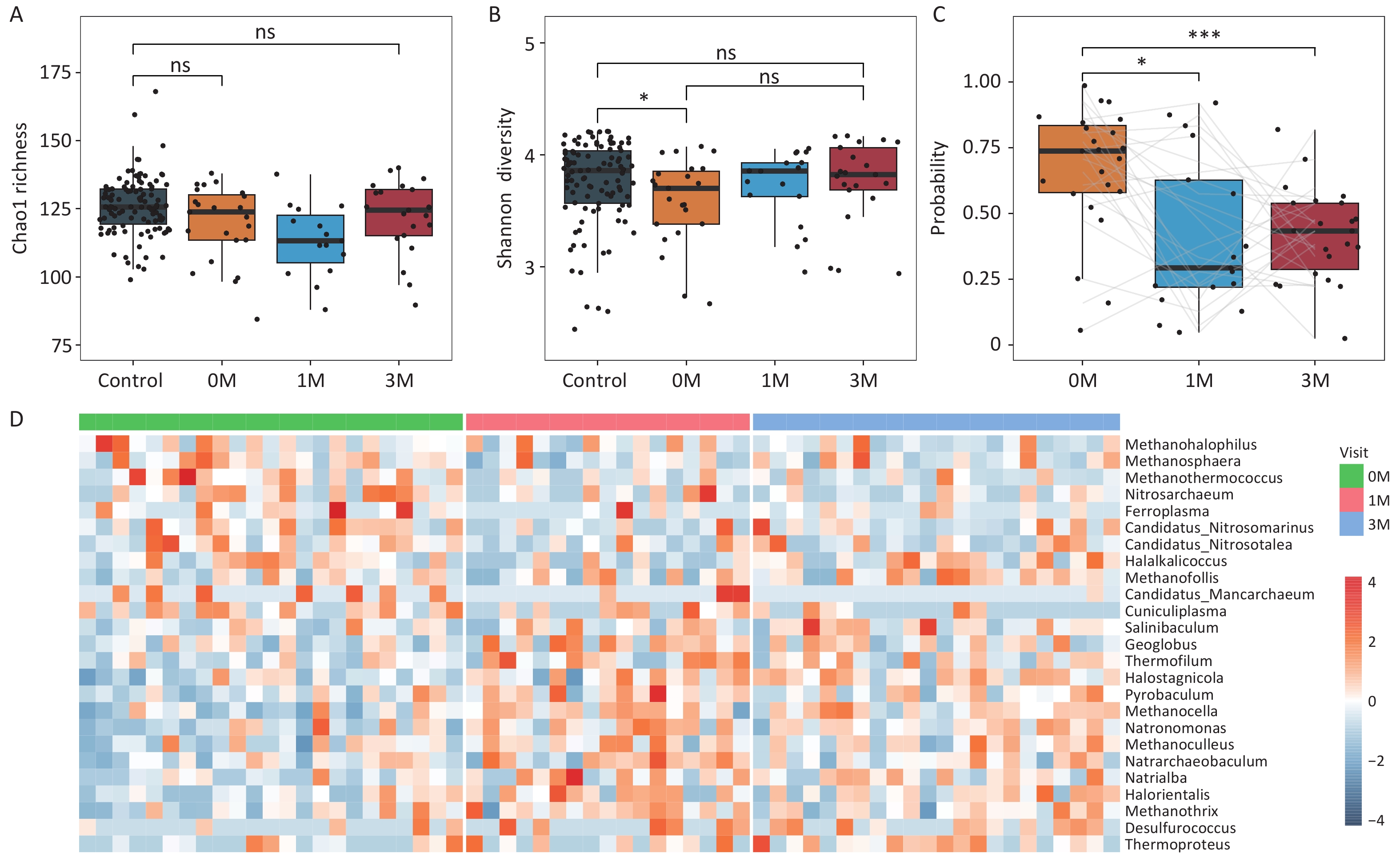
Previous studies have demonstrated significant alterations in the gut microbiome of obese individuals following bariatric surgery. Here, we explored the characteristics of the gut archaea in 23 obese patients who underwent bariatric surgery (sleeve gastrectomy). No significant changes in Chao1 richness or Shannon diversity were observed in obese subjects at 1 and 3 months after surgery compared to their preoperative baseline values (Figure 4A and Figure 4B). A prediction model based on differential gut archaeal genera suggested a decreased probability of obesity in patients after bariatric surgery. This indicated that the gut archaeal profiles in obese subjects after bariatric surgery were more similar to those of lean controls (Figure 4C).
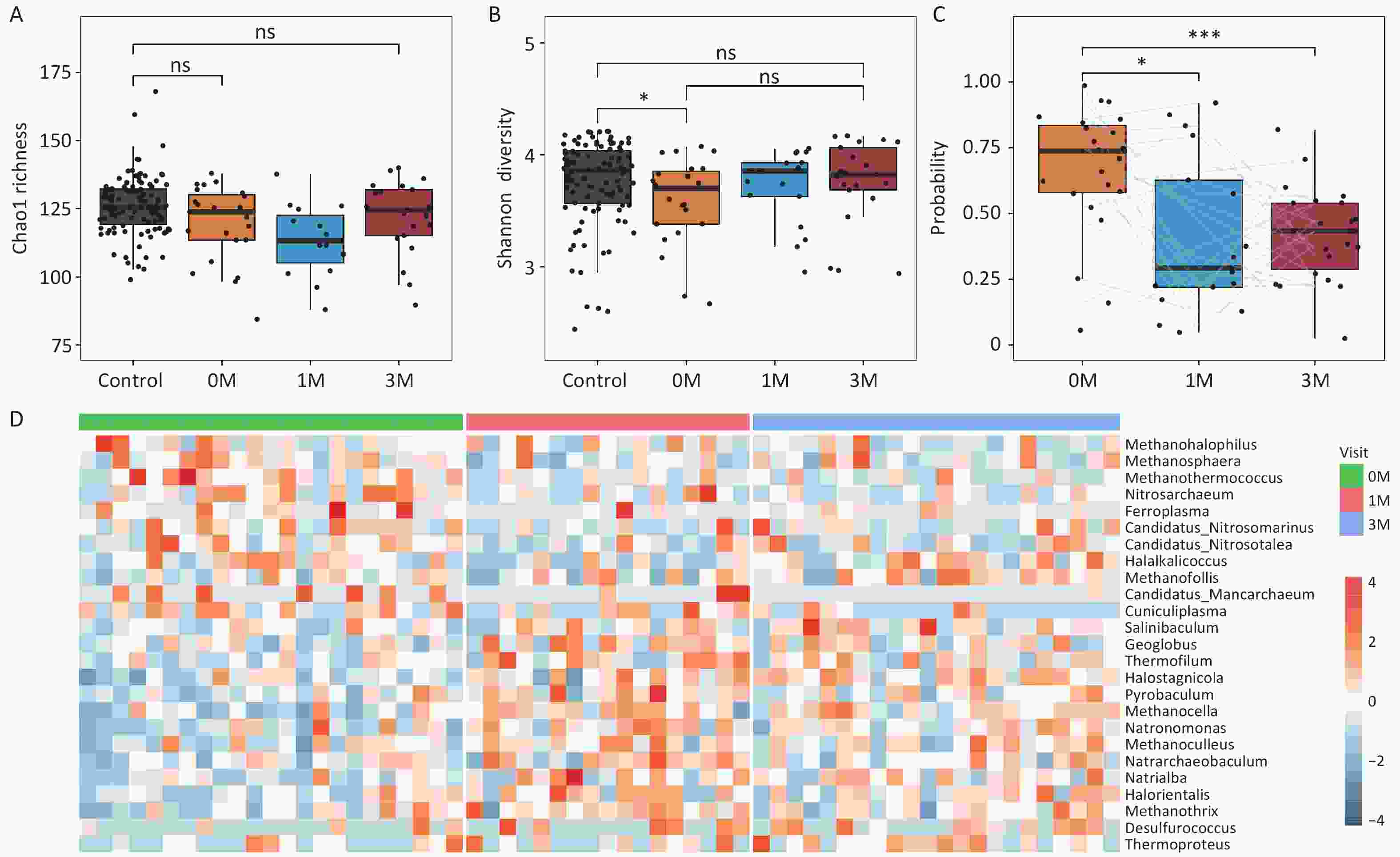
Figure 4. Bariatric surgery mitigates gut archaeal dysbiosis in obese subjects. (A) Chao1 richness and (B) Shannon diversity for obese subjects before and after surgery. Groups were compared using the Wilcoxon rank-sum test. (C) The probability of obesity in obese subjects before and after surgery. (D) Heatmap showing fold changes of the differential gut archaeal genera for obese subjects post-bariatric surgery compared with baseline. Groups were compared using LEfSe (Linear discriminant analysis Effect Size). S0: before surgery. S1: 1 month after surgery. S3: 3 months after surgery. Groups were compared using the Wilcoxon rank-sum test. * P < 0.05,** P < 0.01,*** P < 0.001.
Furthermore, we observed that at 1-month post-surgery, 11 obesity-related gut archaeal genera exhibited increased abundance, whereas six genera displayed decreased abundance. Similarly, at 3 months post-surgery, eight genera increased, and five genera decreased. Importantly, these changes in the genera showed a consistent upward or downward trend at 1- and 3-month surgery (Figure 4D). Specifically, Methanothermococcus, which was enriched in the obese subjects at baseline, significantly decreased at both 1- and 3-months post-surgery, whereas Methanosphaera exhibited a significant reduction at 1-month (Supplementary figure S8). In contrast, taxa that were initially depleted in the obese state, including Methanocella, Natronomonas, and Halostagnicola, significantly increased in abundance at both postoperative time points (Supplementary figure S9). These bidirectional shifts indicated that bariatric surgery effectively reshaped the gut archaeome, aligning it more closely with that of lean controls within 3 months.
-
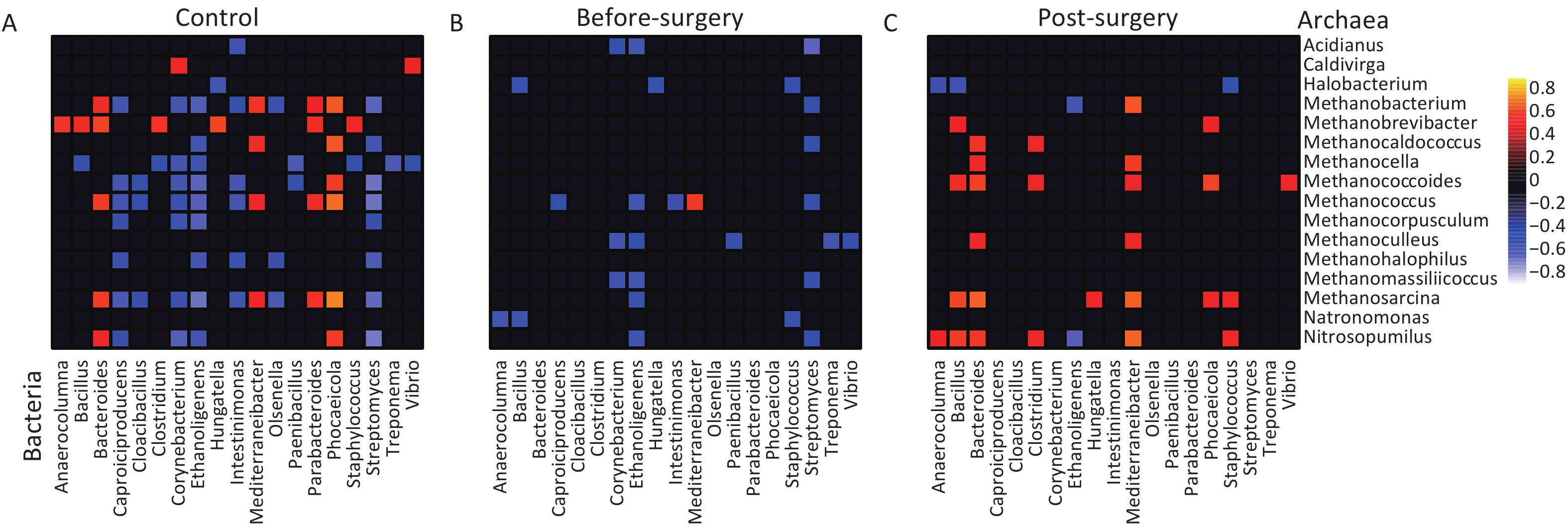
Studies have indicated that altered ecological interactions in the gut microbiota are associated with metabolic diseases. To investigate the potential interplay between gut archaea and bacteria in obese subjects, we used Sparse Correlations for Compositional data (SparCC) to unveil the correlations between gut archaea and bacterial genera. Strong correlations between bacterial and archaeal genera were observed in the lean controls (Figure 5A). However, the number of archaeal-bacterial correlations was remarkably lower in obese subjects than in lean controls (27 vs. 77, P < 0.001, Figure 5A and 5B). Importantly, both positive and negative correlations were notably diminished in obese individuals, indicating a weakening of inter-kingdom interactions between bacteria and archaea (Supplementary figure S10). Intriguingly, following bariatric surgery, the number of positive archaea-bacterial interactions, which had previously diminished, recovered (Figure 5C and Supplementary figure S7). These correlations suggest that decreased bacterial-archaeal interactions may be closely intertwined with the development of obesity. Notably, bariatric surgery partially restored these interactions, particularly the positive co-occurrence relationships, indicating a rebound in inter-kingdom connections.
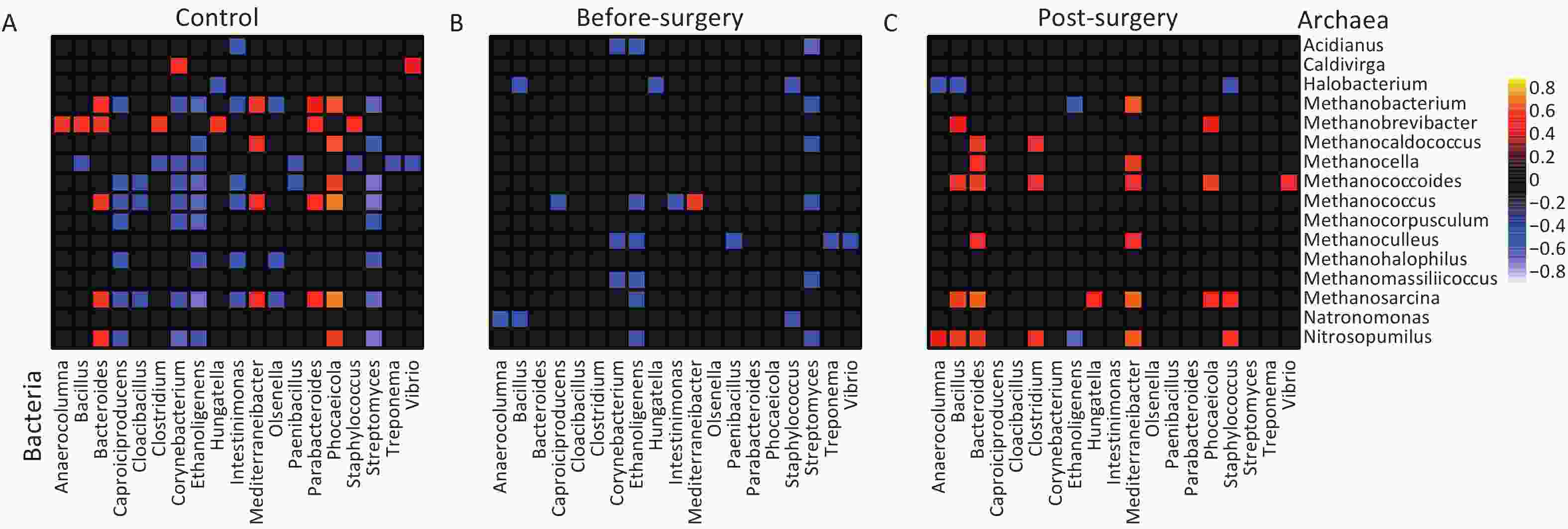
Figure 5. Decreased bacterial-archaeal correlations are found in obese subjects but restored post-bariatric surgery. (A) Lean controls, (B) Obese subjects before surgery, and (C) Obese subjects after surgery. Correlation coefficients were estimated and corrected for compositional effects using the SparCC algorithm. A subset of correlations with coefficient strengths of > 0.5 or < -0.5 and P < 0.05 were considered significant. Any taxa with correlation pairs ≥ 2 were kept in the heatmaps.
-
Archaea are the key components of the human microbiome. Alterations in gut archaea associated with metabolic diseases remain poorly understood. In this study, we utilized high-throughput metagenomic sequencing data to explore the changes in the gut archaea of obese subjects for the first time. Significant alterations in diversity, taxonomic composition, and inter-kingdom correlations were observed in obese subjects compared with lean controls. Additionally, marked changes in gut archaea have been found in obese subjects after bariatric surgery. Prediction models based on these differential gut archaeal taxa have revealed a significantly lower probability of obesity in patients following weight loss surgery. These findings suggest a close correlation between gut archaea and the development of obesity. Therefore, reshaping the archaeal composition of the gut may represent a novel avenue for obesity management.
In the discovery cohort, we found a decreased Shannon diversity index in obese subjects compared to lean controls. However, we did not observe significant changes in the other two validation cohorts. This discrepancy can be attributed to several factors. First, the three cohorts represent distinct geographical and demographic backgrounds. Large-scale meta-analyses have shown that gut microbial diversity varies substantially across populations and is influenced by ethnicity, climate, and local environment[33]. Second, dietary patterns differed markedly between cohorts, and diet is a primary determinant of microbial community structure. Variations in macronutrient composition, fiber intake, and habitual foods can profoundly reshape the microbial diversity and metabolic functions. Third, the variety of sequencing techniques and platforms across studies may introduce bias in microbial composition[34,35]. Nevertheless, our prediction model, based on differential gut archaeal signatures, effectively identified obese subjects in both the discovery and validation cohorts, suggesting consistency in obesity-related archaeal taxa across different cohorts.
In this study, we identified a range of changes in the gut archaeal taxa of obese subjects. Among these differential taxa, several methanogens were significantly increased in the obese subjects. Previous studies have implicated methane-producing microbes in various diseases, including inflammatory bowel diseases, metabolic diseases, and colorectal cancer. Turnbaugh et al. found that the gut microbiome of obese individuals exhibited an augmented capacity for energy harvesting, which correlated with a higher abundance of methanogens[36]. Similarly, Samuel et al. demonstrated that methanogens can disturb the host energy balance by influencing hydrogen transfer and fermentation processes in the gut[37]. Moreover, studies have revealed that in patients with constipation-predominant irritable bowel syndrome, elevated methane production is associated with more severe symptoms and delayed intestinal transit time[38,39]. This phenomenon may be attributed to methane acting as a neuromuscular transmitter capable of diminishing intestinal contraction activity and consequently affecting transit time. Furthermore, Methanogens can compromise cardiovascular health by metabolizing trimethylamine oxide (TMAO)[40].
We also observed significant increases in two Methanogens, Methanosphaera and Methanothermococcus, in obese subjects compared with lean controls. This aligns with the findings of Pimentel et al. who demonstrated that methanogens, through methane production, could impede intestinal transit and enhance nutrient absorption, potentially contributing to obesity[41]. Interestingly, Methanosphaera and Methanothermococcus significantly decreased in obese patients following bariatric surgery. This finding highlights that effective weight loss intervention may be associated with a reduced abundance of methane-producing microbes and associated metabolic pathways.
Correlations among multiple kingdoms of the gut microbiome, including bacteria, fungi, and viruses, are crucial for maintaining the balance of the gut microbial ecology[42-44]. Researchers have revealed significant alterations in the ecological networks between archaea and bacteria in patients with colorectal cancer (CRC)[15]. Specifically, a conspicuous negative correlation was observed between CRC-enriched Archaea and CRC-depleted bacteria, along with a positive correlation between CRC-enriched Archaea and CRC-enriched bacteria. In our study, we observed a significant reduction in bacterial-archaeal correlations in obese subjects compared to those in lean controls, suggesting that bacterial-archaeal correlations are closely associated with the maintenance of gut homeostasis in non-obese individuals. Notably, after bariatric surgery, the number of positive correlations between archaea and bacteria significantly increased, reaching levels comparable to those observed in lean controls. This indicates that effective weight loss interventions, such as bariatric surgery, could partially restore the network connections between the gut archaea and bacteria.
While this study elucidated alterations in gut archaea and archaeal bacterial correlations in obese subjects, it is subject to limitations that warrant further investigation. First, the sensitivity of shotgun sequencing for archaeal detection remains low, as archaea typically constitute < 0.1% of reads, and reference genome coverage is still incomplete. Increased sequencing depth, archaeal-enriched extraction, and targeted archaeal amplicon sequencing are required to obtain reliable archaeal profiles for obesity[45]. Second, the functional interpretation is constrained by the sparse annotation of archaeal metabolic pathways. Methanogenic archaea can modulate bacterial fermentation through interspecies hydrogen transfer and alter short-chain fatty acid production, potentially affecting host energy harvesting. Further integration of metatranscriptomics and metabolomics may help to clarify their metabolic roles. Finally, although archaeal-targeted interventions were not examined in this study, such approaches may represent future directions. Modulating archaeal–bacterial syntrophy through prebiotic, probiotic, or fecal microbiota transplantation strategies could influence host energy metabolism and complement existing obesity treatments.
-
In conclusion, we found that obesity is characterized by decreased gut archaeal diversity and bacterial-archaeal inter-kingdom correlations. Obesity-related gut archaeal taxa discriminated obese subjects from lean controls in both the discovery and independent validation cohorts. Following weight loss treatment by bariatric surgery, a significant change in the gut archaeal composition was observed, and the prediction model utilizing archaeal taxa showed a substantially lower probability of obesity in obese subjects postoperatively than at baseline. These results suggest that the gut archaea play an important role in the development and management of obesity. Further studies are needed to explore the potential benefits of modulating the gut archaea to enhance the efficacy of weight loss interventions.
-
Not applicable. This study utilized publicly available data and did not involve the collection of new data involving human subjects; therefore, ethical approval was not required.
-
Not applicable. This study utilized publicly available data and did not involve new data collection involving human subjects; therefore, informed consent was not required.
HTML
Patients and Sample Information
Shotgun Sequencing Reads Processing and Taxonomy Annotation
Differentially Abundant Taxa Identification
Statistical Analysis
The Established Research Findings That Obesity is Intensively Associated with Altered Gut Microbiota were Replicated by Our Analytic Methods
Reduced archaeal diversity is observed in obese subjects compared with lean controls
Gut Archaeal Composition of Obese Subjects Remarkably Differs From Lean Controls
Archaeal Signatures Could Be Predictive Markers for Obesity
Bariatric Surgery Could Reverse the Obesity-Associated Archaeal Alteration and Align Closely with Lean Controls
Decreased Bacterial-Archaeal Correlations are Found in Obese Subjects but Restored After Bariatric Surgery
Competing Interests The authors declare no conflict of interest.
Authors’ Contributions K. L. Y. and J. N. Z.. and JW.Y. contributed equally to this study. Conceptualization: KL.Y. and LL. C. and J.Y.; Methodology: K. L. Y.. and J.Y.; and software, K. L. Y.. and J.Y.; validation, J. N. Z.; and formal analysis, K. L. Y.. and J. Y.; investigation, N. Z.. and QC.W.: Resources, QC.W. and H.W.; data curation: JW. Y. and HM.W.; writing and original draft preparation, KL.Y. and J.Y.; writing, review, and editing, J. N. Z. and L. L. C.; visualization, K. L. Y.; supervision, J.Y. and L. C.; project administration, K. L. Y.. and LL.C.; Funding acquisition: K. Y. and J. N. Z. and J.Y.
Data Availability Statement and Ethics: The data and materials used in this study are publicly available from the NCBI SRA under BioProject identifiers.
&These authors contributed equally to this work.







 Quick Links
Quick Links
 DownLoad:
DownLoad: