-
Sleep is a biological phenomenon with highly conserved evolutionary characteristics. The American Academy of Sleep Medicine and the Sleep Research Society recommend that adults get at least 7 hours of sleep per night[1]. However, the stress caused by fast-paced life often leads to sleep deprivation (SD). SD is strongly associated with damage to the auditory system[2,3]. Obstructive sleep apnea-hypopnea syndrome (OSAHS) is a common sleep disorder. Clinical observations indicate that some patients with OSAHS experience persistent hearing loss accompanied by tinnitus and other symptoms[4]. More than 61.8% of patients with sudden deafness experienced SD[5].
Currently, various paradigms are used for SD in animal models, including modified multiple platforms, rotating rods, gentle stimulation, forced exercise, and pharmacological sleep deprivation methods. However, these paradigms generally do not achieve sufficient SD. Additionally, the phenotypic validation of SD animal models established using these paradigms is relatively stable in the central nervous system but less effective in peripheral systems and organs[6]. Sang et al.[6] showed that the use of the curling prevention by water (CPW) paradigm for prolonged SD can keep mice awake for approximately 96% of their time. Furthermore, SD was found to induce the accumulation of prostaglandin D2 (PGD2) in the brain, leading to excessive production of pro-inflammatory cytokines, ultimately resulting in a systemic cytokine storm and multiple organ dysfunction syndrome. Therefore, a high-efficiency CPW paradigm was used to establish a mouse SD model, and the regulatory effects of prolonged SD on the inner ear auditory function were investigated.
SD is closely associated with auditory system damage. Animal studies have shown that SD induces structural and morphological changes in the cochlea of rats, leading to auditory impairment[3]; however, the underlying mechanisms remain unclear. To investigate the adverse effects of prolonged SD on the auditory system, we used the CPW paradigm to induce prolonged SD in mice and evaluated the changes in auditory function and protein expression levels. Finally, the mechanisms via which oxidative stress and inflammation regulate the effect of prolonged SD on auditory function were determined.
Previous studies have shown that SD is a stress factor in animals. After 72 h of SD, the change in body weight was significantly greater in the SD group than in the control group (Supplementary Figure S1B). The 72 h SD group also exhibited a significant increase in corticosterone (CORT) levels, the main steroid hormone of the hypothalamic-pituitary-adrenal axis (Supplementary Figure S1C). In the 96 h SD model, the mortality rate in mice was as high as 70%, which was higher than that (11%) observed in the 72 h SD group (Supplementary Figure S2). We observed that 96 h SD was not suitable for animal models of hearing loss, as it led to high mortality in mice.
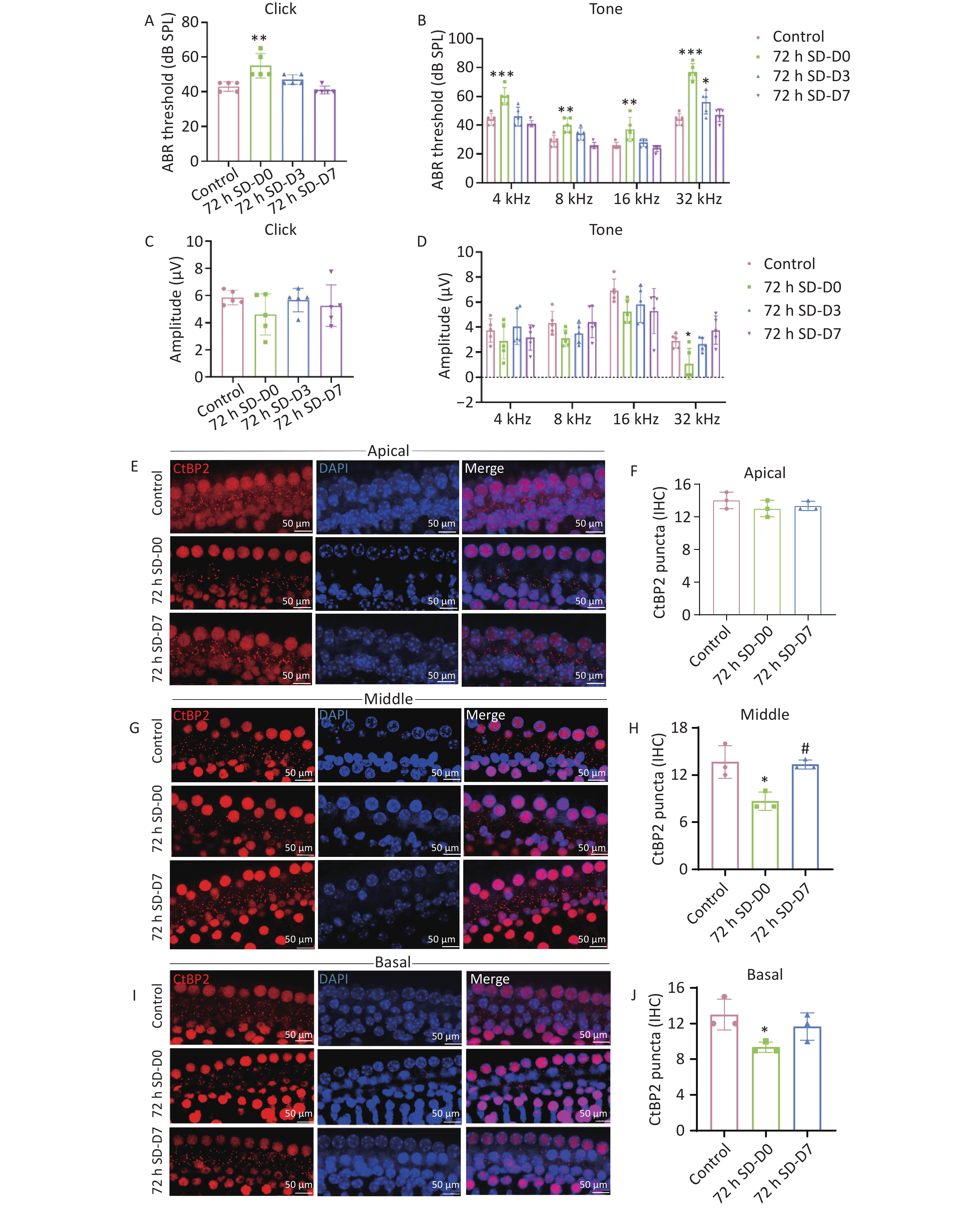
Clinical studies have shown that SD impairs the auditory system, leading to tinnitus and sudden deafness[7]. A retrospective study of primary tinnitus revealed that tinnitus severity correlated positively with poor sleep quality, suggesting that poor sleep quality may influence the onset and maintenance of tinnitus[8]. In addition, Li et al.[9] found that repeated SD disrupts the autophagy-lysosomal system of the inner ear and aggravates cochlear damage caused by noise exposure. To investigate the effect of SD on the auditory threshold of mice, auditory brainstem response (ABR) testing was performed immediately after SD (D0) and on days 3 (D3) and 7 (D7) post-SD (Figure 1). At all frequencies, the hearing threshold in the D0 group after 72 h of SD was significantly higher than that in the control group. The hearing threshold of the 72 h SD group recorded on D0 increased by an average of 13 dB (click), 14 dB (4 kHz), 8 dB (8 kHz), 7 dB (16 kHz), and 31 dB (32 kHz) (Figure 1C, D). Over time, the hearing function of mice in the 72 h SD group recovered. The hearing threshold in the 72 h SD group at D3 was significantly higher than that in the control group, and it returned to the control level; however, the hearing threshold at 32 kHz was still significantly higher than the control measurement. The hearing thresholds at D7 did not differ significantly from that of the control. Subsequently, the amplitudes of ABR I waves induced by each stimulus frequency were analyzed (Figure 1E). At 32 kHz, the ABR I wave amplitude was significantly lower than the control measurement at D0, with an average decrease of 1.83 μV (32 kHz) (Figure 1F). Over time, the ABR I wave amplitudes at D3 and D7 returned to normal levels at each frequency. In addition, we measured hearing in mice immediately after 96 h of SD. At all frequencies, the hearing threshold at D0 after 96 h of SD was significantly higher than that of the control, with average increases of 16 dB (click), 18 dB (4 kHz), 30 dB (8 kHz), 29 dB (16 kHz), and 25 dB (32 kHz) (Supplementary Figure S3A). The ABR I wave amplitudes were significantly lower than those measured in the control group (P < 0.05). The amplitudes of the ABR I waves decreased by 5.24 μV (click), 1.52 μV (4 kHz), 1.58 μV (8 kHz), 2.40 μV (16 kHz), and 1.73 μV (32 kHz) (Supplementary Figure S3B). However, the 96 h SD method is not suitable for animal models of hearing loss due to the large damage to the mouse body. Therefore, the 72 h SD method was used to establish subsequent models. These findings are consistent with the ABR threshold change characteristics of temporary threshold shift (TTS), indicating that prolonged SD causes temporary hearing loss and a reversible reduction in cochlear synaptic function in mice. Additional evidence comes from the morphological examination of the cochlear sensory hair cells and ribbon synapses. Myosin VIIa staining was performed to evaluate hair cell survival (Supplementary Figure S4). In control animals, the cochlear surfaces showed an orderly arrangement of three rows of outer hair cells (OHCs) and one row of inner hair cells (IHCs). No reduction was observed in the number of OHCs and IHCs at any time point (D0, D3, and D7) after SD, indicating that SD did not cause significant damage to the cochlear hair cells. Subsequently, we examined the ribbon synapses that transfer signals from IHCs to spiral ganglion neurons. Immunostaining of the presynaptic (CtBP2) site showed that the synaptic density of the parietal gyrus of the cochlea did not change significantly on D0 after SD compared to that in the control group. In contrast, the synaptic ribbon density in the middle and basal gyri of the cochlear gyrus decreased significantly (Figure 1E–J). Over time, the synaptic ribbon density at any cochlear location in the 72 h SD group on D7 did not differ significantly compared with that in the control group. These results indicate that prolonged SD induces reversible structural and functional damage to cochlear ribbon synapses, demonstrating the extent of plasticity of ribbon synapses. This suggests that sleep recovery facilitates the restoration of synaptic signal transmission and initiates the regeneration of ribbon synapses, thereby increasing their number.
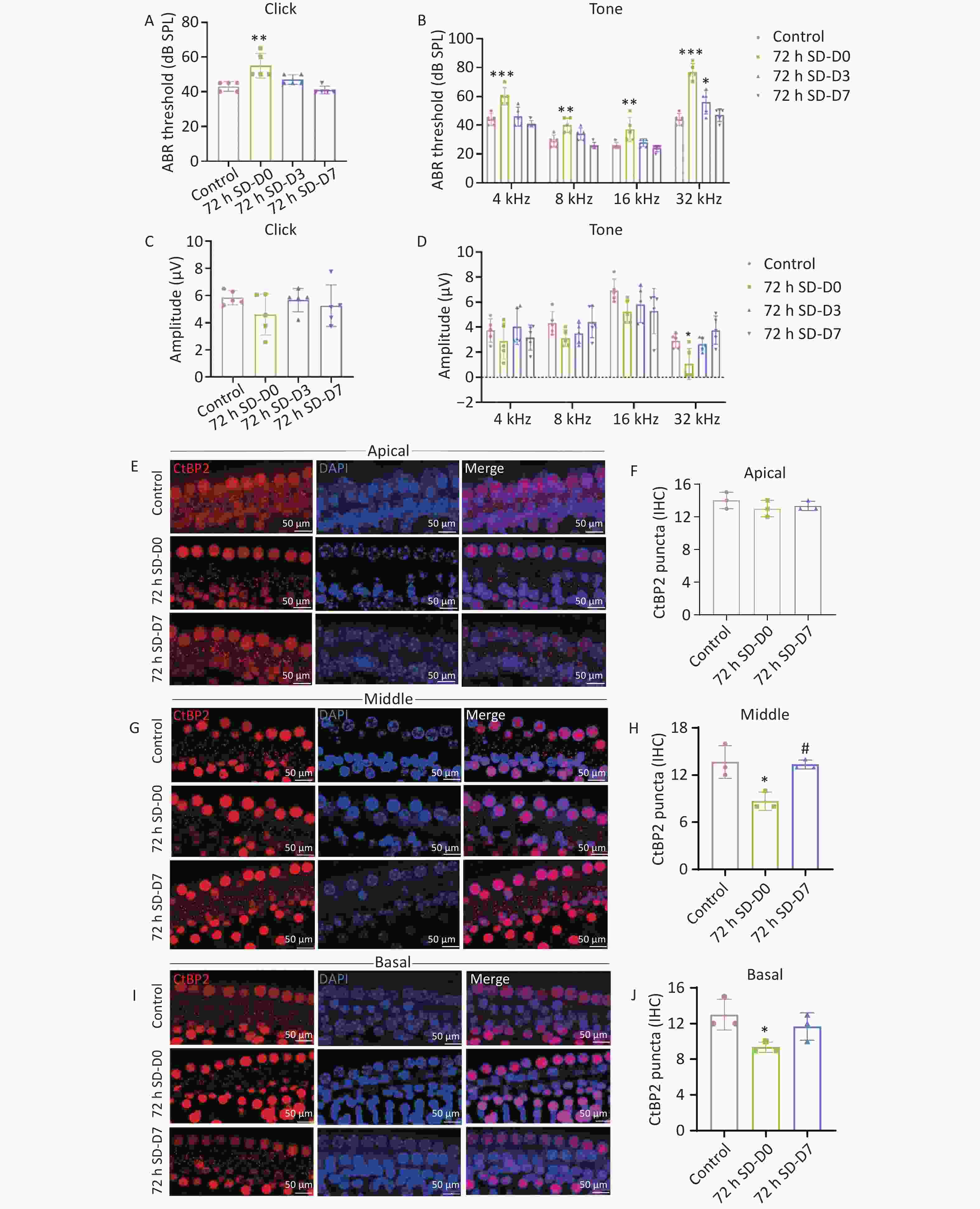
Figure 1. Cochlear functional and morphological evaluation. (A–B) ABR thresholds of mice were elicited after click or pure tone (4, 8, 16, and 32 kHz) stimuli. Seventy-two hours of SD induced a temporary threshold elevation of approximately 7–31 dB (n = 5). (C–D) The amplitude was elicited after click or pure tone (4, 8, 16, and 32 kHz) stimuli (n = 5). (E–J) Representative images of synaptic ribbons (red, anti-CtBP2; blue, DAPI) in the immunohistochemically stained area of the cochlear turn (n = 3). *P < 0.05, **P < 0.01, ***P < 0.001 compared with the control group and #P < 0.05 compared with the 72 h SD-D0 group. Scale bar = 50 μm. D0, immediately after 72 h SD. D3, Day 3 after 72 h SD. D7, Day 7 after 72 h SD. ABR, auditory brainstem responses; SD, sleep deprivation.
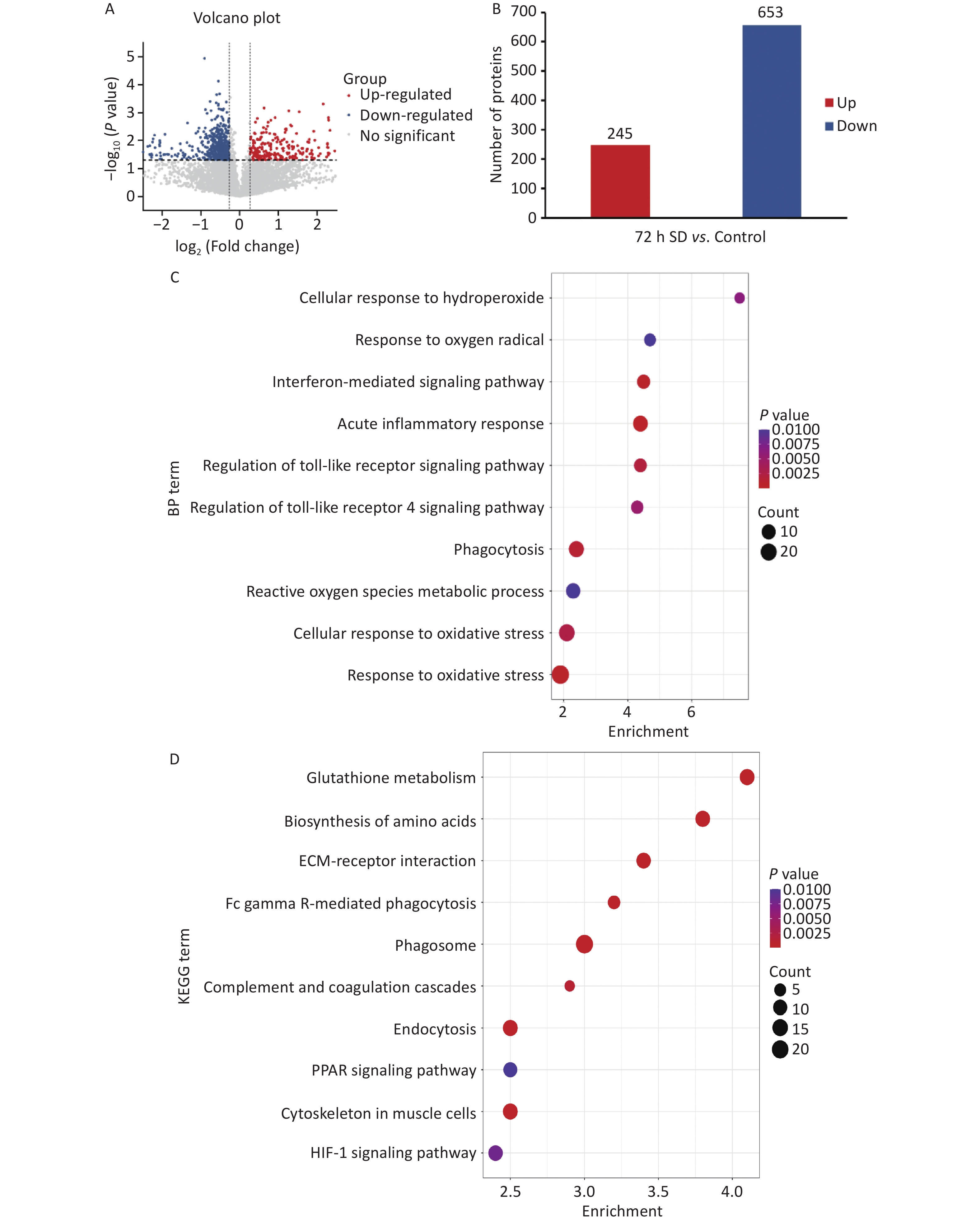
To further analyze the mechanism via which prolonged SD affects auditory function, data-independent acquisition proteomics was used to analyze protein expression in cochlear tissues. As shown in Figures 2A and 3B, after comparing the gene expression of the 72 h SD and control groups, 898 differentially expressed proteins (DEPs) were identified, of which 245 were upregulated and 653 were downregulated. The degree of aggregation among biological replicates within the group was high, indicating that the differences within the group were small and those between the groups were obvious, which could be used for follow-up omics research. Gene Ontology functional enrichment analysis revealed that most DEPs were distributed in biological processes related to oxidative stress, acute inflammatory responses, and regulation of the Toll-like receptor (TLR) 4 signaling pathway (Figure 2D). Kyoto Encyclopedia of Genes and Genomes pathway analysis revealed that DEPs were mainly enriched in signaling pathways related to oxidative stress, inflammatory responses, and immune defense mechanisms, such as the hypoxia inducible factor-1 and peroxisome proliferator-activated receptor signaling pathways (Figure 2E). These data suggested that prolonged SD triggered cochlear oxidative stress and inflammatory responses. Bioinformatics analysis showed that prolonged SD may affect auditory function in mice through mechanisms involving inflammatory responses and regulation of the TLR4 signaling pathway. The TLR4 signaling pathway is a classical pathway that mediates inflammatory immune responses, while the TLR4/NF-κB/NLRP3 pathway is a classic inflammatory pathway.
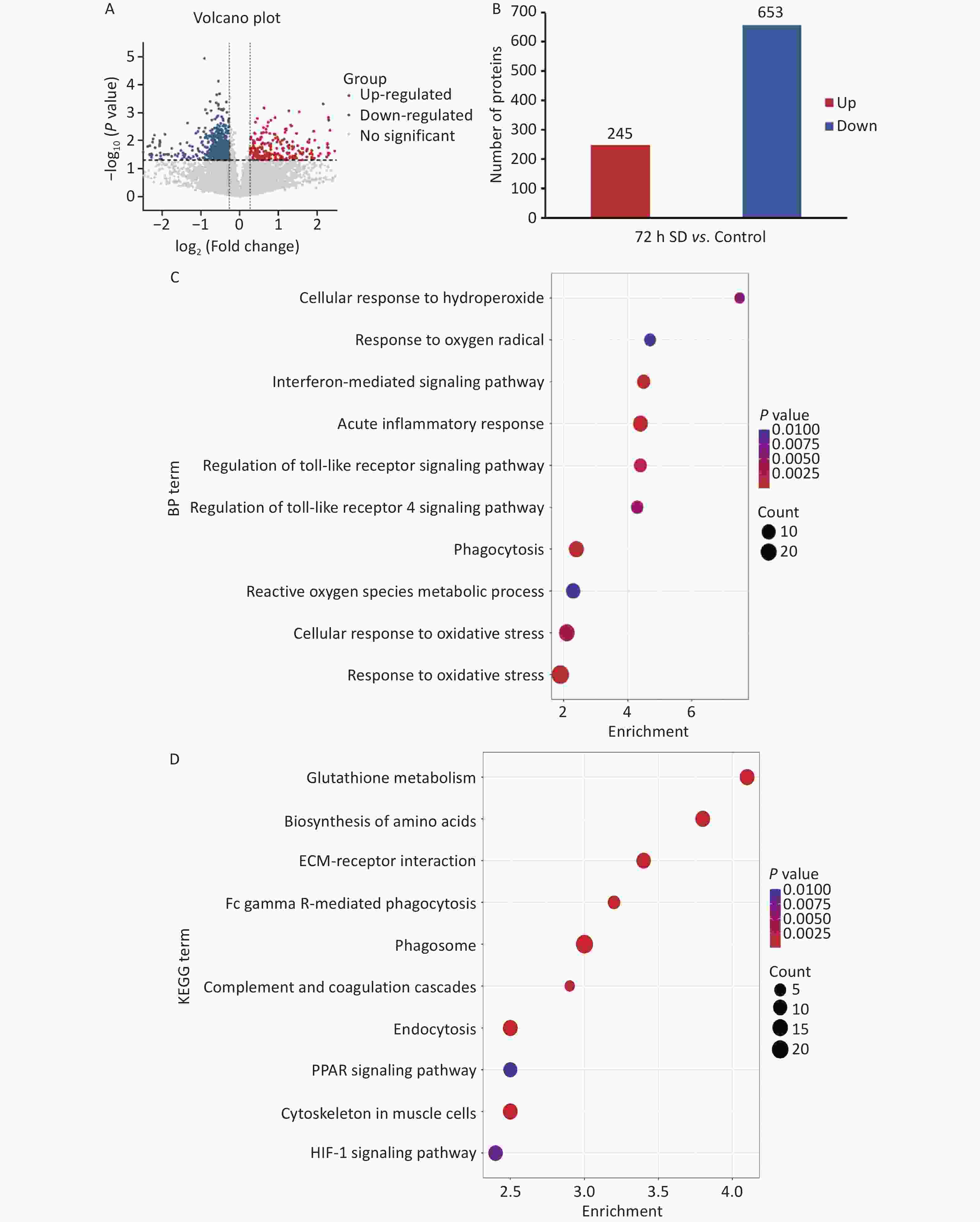
Figure 2. Comparison of protein levels between the 72 h SD and control groups. (A) Volcano plot of DEPs between the 72 h SD and control groups; (B) Statistical plot of DEPs between the 72 h SD and control groups; (C) Enrichment map of GO biological process functions of DEPs between the 72 h SD and control groups; (D) Scatter plot of KEGG pathway enrichment of DEPs between the 72 h SD and control groups. DEPs, differentially expressed proteins; GO, Gene Ontology; KEGG, Kyoto Encyclopedia of Genes and Genomes; SD, sleep deprivation.
To verify the effect of prolonged SD on the systemic inflammatory response in mice, the serum levels of inflammatory factors were measured. The serum concentrations of interleukin (IL)-1β, IL-6, and IL-17A were significantly increased after SD compared with that in the control group (Supplementary Figure S5A–C), confirming that SD elicits a systemic inflammatory response.
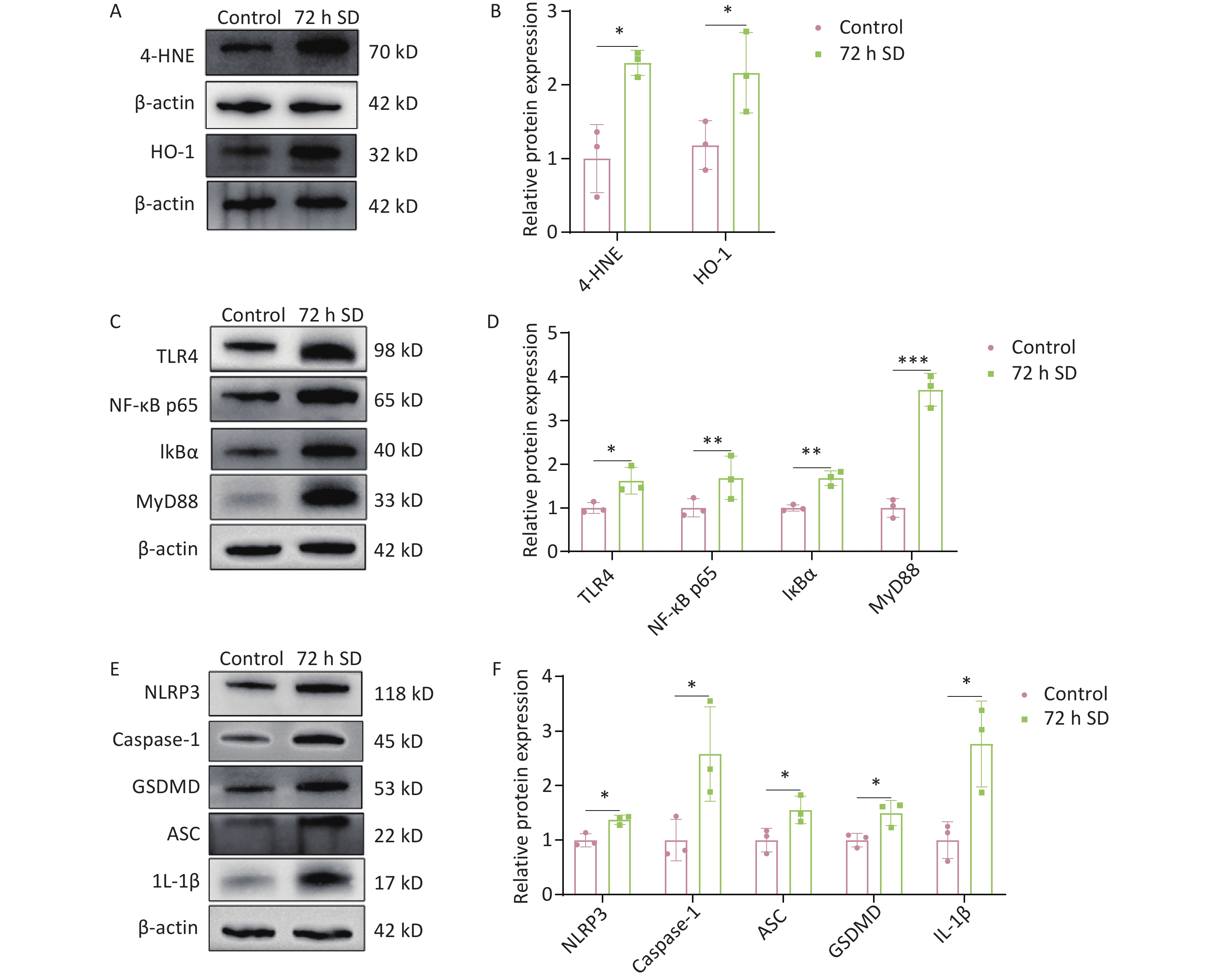
The cochlea is a metabolically active organ. Under stress conditions, cochlear damage induces oxidative stress. Previous studies have shown that cochlear tissue produces large amounts of reactive oxygen species (ROS) in response to factors such as noise exposure, which is one of the main causes of cochlear damage and hearing loss[10]. Clinical studies have shown that patients with OSAHS exhibit reduced rapid eye movement sleep, leading to altered neurotransmitter concentrations, inflammation, and oxidative stress, all of which affect auditory signaling[4]. The protein levels of 4-hydroxynonenal (4-HNE) and heme oxygenase-1 (HO-1) in the cochlea (Figure 3A–B) increased after SD. These results suggest that the activation of the cochlear oxidative system after SD leads to the release of lipid peroxides and activation of the antioxidant defense system to remove excess ROS from cells. Therefore, we hypothesized that antioxidant enzymes, such as HO-1, may continue to exert antioxidant effects and maintain the intracellular redox balance by removing excess ROS, thereby contributing to hearing recovery.
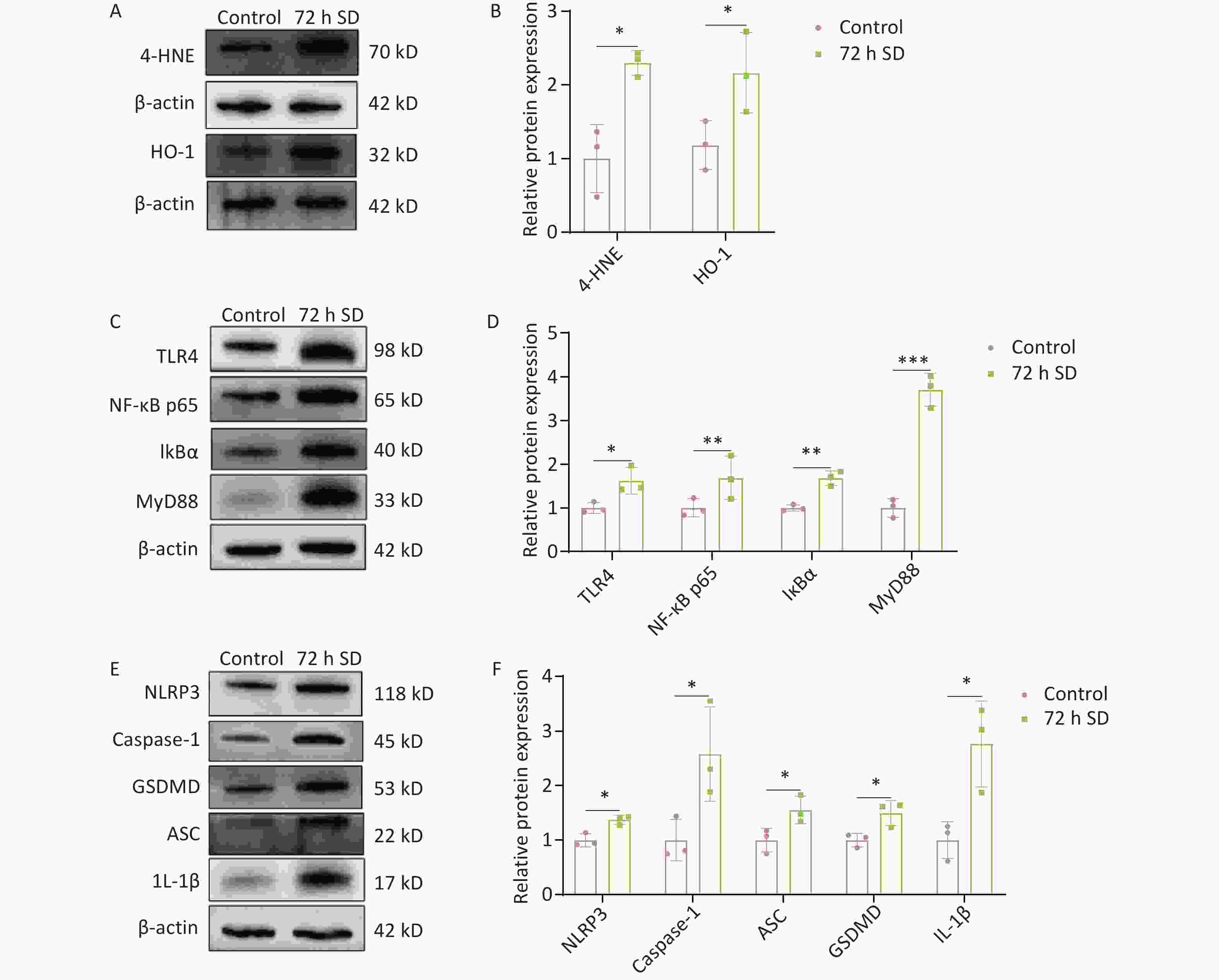
Figure 3. Effect of 72 h SD on the expression of oxidative stress marker proteins and the TLR4/NF-κB/NLRP3 signaling pathway-related proteins in the cochlea. (A) Western blot analysis of 4-HNE and HO-1 levels. (B) Expression level of 4-HNE. (C) Western blot analysis of TLR4, NF-κB p65, IκBα, and MyD88 levels. (D) Western blot analysis of TLR4, NF-κB p65, IκBα, MyD88 levels. (E) Expression levels of NLRP3, caspase-1, GSDMD, ASC, and IL-1β. (F) Expression levels of NLRP3, caspase-1, GSDMD, ASC, and IL-1β. (n = 3). *P < 0.05, **P < 0.01, ***P < 0.001 compared with the control group. 4-HNE, 4-hydroxynonenal; HO-1, heme oxygenase-1; SD, sleep deprivation. ASC, apoptosis-associated speck-like protein containing CARD; caspase-1, cysteine aspartate-specific protease1; GSDMD, gasdermin-D; IκBα, nuclear factor kappa B inhibitor alpha; IL-1β, interleukin-1β; MyD88, myeloid differentiation protein 88; NF-κB, nuclear factor kappa-B; NLRP3, NOD-like receptor thermal protein domain- associated protein 3; SD, sleep deprivation; TLR4, toll-like receptor 4.
To further analyze the effect of prolonged SD on the cochlear inflammatory response, the expression of TLR4 signaling pathway-related proteins in cochlear tissue was detected. The results showed that the expression levels of TLR4, NF-κB p65, IκBα, and MyD88 in the cochlea were significantly higher after SD than before SD (Figure 3C–D), indicating that SD activated the TLR4 signaling pathway. The NLRP3 inflammasome pathway is regulated by TLR4 signaling; therefore, the expression of NLRP3 inflammasome-associated proteins in the cochlea was measured following SD. The results showed that the expression levels of NLPR3, caspase-1, ASC, GSDMD, and IL-1β in the cochlea were significantly higher after SD than in the control group (Figure 3E–F). The expression levels of TLR4 signaling pathway-related proteins and NLRP3 inflammasome-associated proteins were also significantly higher in the cochlea after SD, suggesting that prolonged SD triggers cochlear inflammation by activating the TLR4/NF-κB/NLRP3 signaling pathway, as shown in Supplementary Figure S6.
Taken together, these results indicate that prolonged SD can induce auditory system damage by activating the TLR4/NF-κB/NLRP3 signaling pathway. The specific mechanism may involve triggering of a systemic inflammatory response after prolonged SD, followed by TLR4 receptor activation in the cochlea. Second, TLR4 activates NF-κB and IκBα via the MyD88-dependent pathway, promoting NLRP3 inflammasome expression and triggering cochlear inflammation. Additionally, prolonged SD induces an oxidative stress response in the cochlea. Eventually, these processes lead to TTS and cochlear synaptopathy. These findings suggest that clinicians should closely monitor the hearing of patients with sleep disorders.
Our findings indicates that SD affects auditory function in the inner ear. By analyzing its mechanisms of action, we elucidate the link between SD and auditory impairment. This study focused on acute exposure to SD, which is its limitation. Further research should assess the effects of chronic SD on the auditory system of mice and its underlying mechanisms. In addition, the current state of clinical research in human patients should be considered. Although this study utilized proteomic and pathway analyses for detailed elucidation, further verification of causal relationships was not performed. More experiments, such as key gene knockout, are still needed in the future to explore the deeper molecular mechanisms.
HTML
Competing Interests The authors declare no conflict of interest.
Ethics All animal experiments were conducted in accordance with the Guidelines for the Care and Use of Laboratory Animals and were approved by the Ethics Committee of the Military Medical Sciences Academy (Animal Ethics Number: IACUC of AMMS-04-2023-033).
Authors’ Contributions Xiaoqiong Song and Kefeng Ma conceived, designed, and performed the research, and wrote and revised the paper. Mengzhu Cheng, Cui Gu, Fenghan Wang, and Xinyu Dai collected the samples and performed the research. Chunping Wang performed the research and revised the manuscript. Bo Cui and Xiaojun She designed and supervised the study and reviewed the manuscript.
Data Sharing The supplementary materials will be available in www.besjournal.com.
&These authors contributed equally to this work.
Reference







 Quick Links
Quick Links
 DownLoad:
DownLoad: