-
Perfluorinated/polyfluorinated substances (PFAS) are synthetic organic compounds widely used in over 200 industrial and consumer applications since the 1950s. In recent years, numerous emerging PFAS alternatives such as hexafluoropropylene oxide dimer acid (HFPO-DA, GenX), 4,8-dioxa-3H-perfluorononanoic acid, and 6:2 chlorinated polyfluorinated ether sulfonate (6:2 Cl-PFESA, F-53B) have replaced restricted legacy PFAS. However, the environmental and human health effects of these alternatives remain poorly understood. Since they are resistant to degradation, these persistent compounds have been detected globally in water, soil, dust, and biological samples from wildlife, marine organisms, and humans. The detection of PFAS in maternal blood, umbilical cord blood, amniotic fluid, and breast milk raises concerns that humans may be exposed to these chemicals during vulnerable fetal developmental stages[1].
The fetal developmental period is a critical window during which the reproductive organs are highly sensitive to PFAS exposure. Anogenital distance (AGD), defined as the distance between the anus and genitalia, is an established biomarker of fetal androgen exposure in rodents and humans[2]. As a sensitive indicator of early reproductive development, AGD is regulated by fetal androgens (e.g., testosterone and dihydrotestosterone) and exhibits pronounced sexual dimorphism; it is markedly longer in males than in females[3]. In newborns, AGD not only reflects in utero hormonal exposure levels, but may also predict long-term reproductive health outcomes, such as sperm quality, fertility, and risks of hormone-related disorders[4]. Animal and epidemiological studies have linked prenatal PFAS exposure to AGD alterations, but the current evidence remains inconsistent. Epidemiological studies have reported conflicting associations (positive, negative, or null) between PFAS exposure and AGD in male and female infants and have primarily focused on legacy PFAS instead of emerging PFAS alternatives. Additionally, most studies have examined only single-sex cohorts or relied on a single AGD indicator, lacking comprehensive comparisons across offspring sexes and multiple AGD indicators.
In this study, the association between prenatal emerging PFAS exposure and AGD in newborns was explored in 375 mother–child pairs enrolled in the Jiashan Birth Cohort between September 2016 and April 2018. Eligible participants were pregnant women with a gestational age < 16 weeks, who attended initial prenatal care appointments at Jiashan Maternal and Child Health Hospital, Zhejiang Province, China, or Jiashan County First People’s Hospital, Zhejiang Province, China during this period. Exclusion criteria included: multiple pregnancies, fetal birth defects, pregnancy complications (e.g., preeclampsia or gestational diabetes), severe medical conditions, or incomplete questionnaire or blood sample data.
The demographic characteristics and medical histories of all participants were collected through questionnaires and clinical data. The demographic characteristics of the study population are summarized in Supplementary Table S1, and the collection and detailed definitions of the covariates are presented in Supplementary Text S1. Peripheral venous blood was collected from pregnant women at 12–16 weeks of gestation using a standard venous blood collection procedure. The PFAS were quantitatively analyzed using isotope-labelled internal standards. The analytical details of PFAS quantification are provided in the Supplementary Materials (Supplementary Text S2, Supplementary Tables S2–S4). Correlations among individual PFAS congeners (detection frequency, DF > 70%) were assessed using Spearman’s correlation analysis, and the results are presented in Supplementary Figure S1.
Two physicians measured the AGD of all neonates using a standardized caliper within 3 days after delivery, and the inter-rater agreement is presented in Supplementary Text S3. During the measurements, the newborns were maintained in a frog-leg position (hip flexion at 60–90°) by an assistant. The physicians performed independent AGD measurements from the anterior aspect using standardized digital calipers. For male neonates, AGDAP (from the anal midpoint to the anterior penile attachment at the pubic symphysis) and AGDAS (from the anal center to the scrotal-perineal border where rugated skin transitions to smooth perineum) were measured. Female neonates were similarly assessed for AGDAC (from the center of the anus to the clitoris) and AGDAF (from the center of the anus to the posterior convergence of the fourchette). The detailed measurement landmarks and neonatal AGD distributions are shown in Supplementary Figure S2 and Supplementary Table S5, respectively.
Statistical analyses were performed using R software (version 4.4.2) and SPSS (version 27.0; IBM Corp.). PFAS with DF values > 70% were included in the multivariate linear regression (MLR), restricted cubic spline (RCS), and Bayesian kernel machine regression (BKMR) models for analysis. The associations between individual PFAS concentrations and AGD in both sexes were estimated through MLR, with regression coefficients (β) and 95% confidence intervals (CI) calculated. Potential nonlinearity in exposure-response relationships for each PFAS was calculated through RCS modeling, and the mixture and individual effects of PFAS co-exposure on AGD were calculated through BKMR. BKMR and RCS modeling used the “bkmr” and “rms” packages in R, respectively. A P-value of < 0.05 was considered statistically significant.
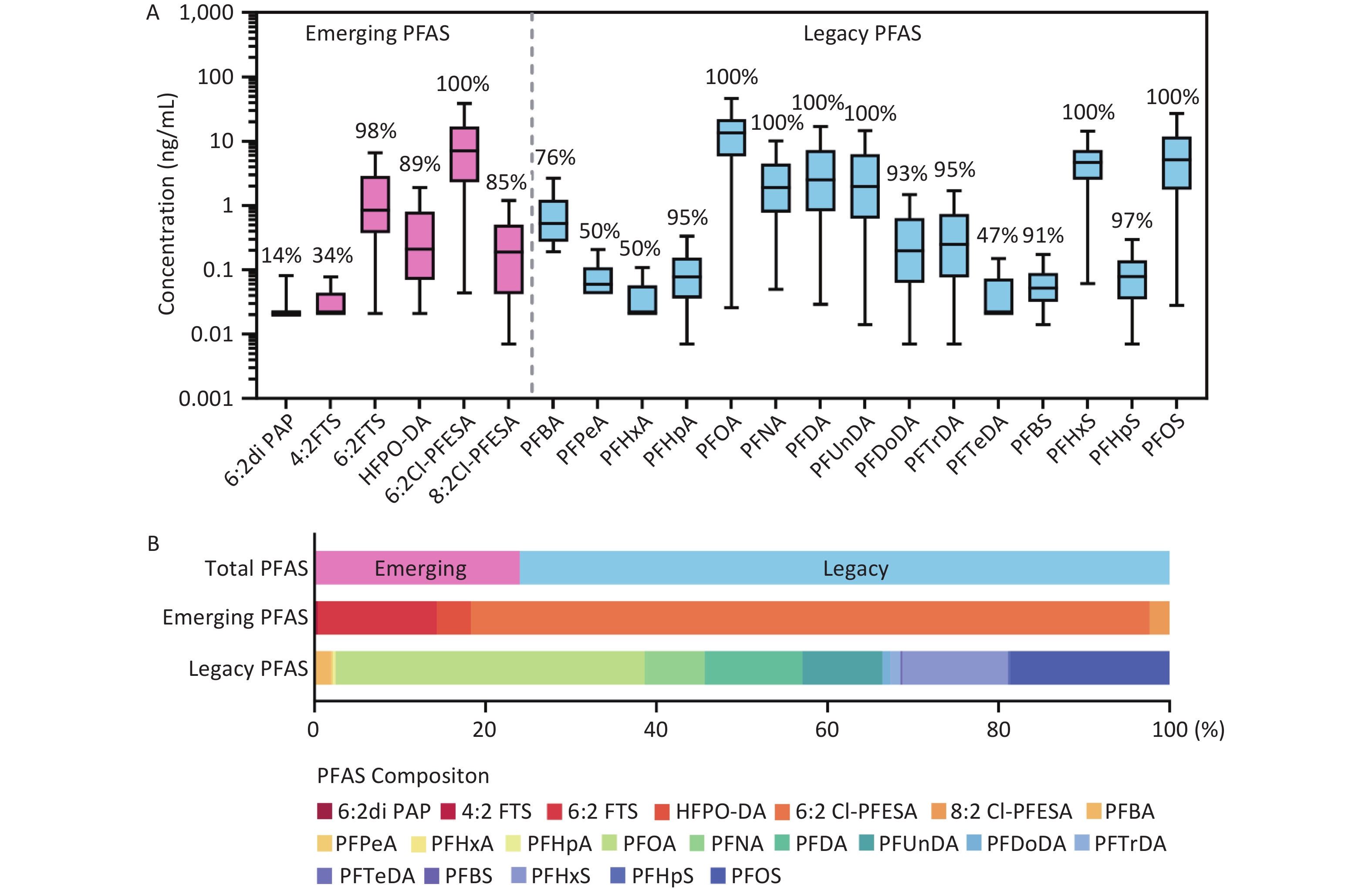
A total of 53 PFAS were measured in maternal plasma, including 22 legacy PFAS and 31 emerging alternatives. As shown in Figure 1 and Supplementary Table S6, 15 legacy PFAS and 6 emerging PFAS alternatives were detected with detection frequencies ranging from 14% to 100%. Perfluorooctanoic acid (PFOA) showed the highest median plasma concentration (13.58 ng/mL), followed by perfluorooctanesulfonic acid (PFOS), perfluorohexane sulfonic acid (PFHxS), perfluorodecanoic acid, perfluoroundecanoic acid (PFUnDA), and perfluorononanoic acid. Although PFOA and PFOS have been banned, they remain the predominant legacy PFAS components in maternal plasma due to their long half-lives. Among the emerging PFAS, the highest median plasma concentration was found for 6:2 Cl-PFESA (7.25 ng/mL), followed by that of 6:2 fluorotelomer sulfonic acid, HFPO-DA, and 8:2 chlorinated polyfluoroalkyl ether sulfonate. The high detection frequencies (> 85%) of these compounds indicated the bioaccumulation of emerging PFAS in the maternal body. Notably, 6:2 Cl-PFESA crosses the placental barrier, showing detectable levels in the umbilical cord blood with direct fetal exposure during pregnancy. However, the toxicity profile of 6:2 Cl-PFESA in fetuses remains poorly characterized.
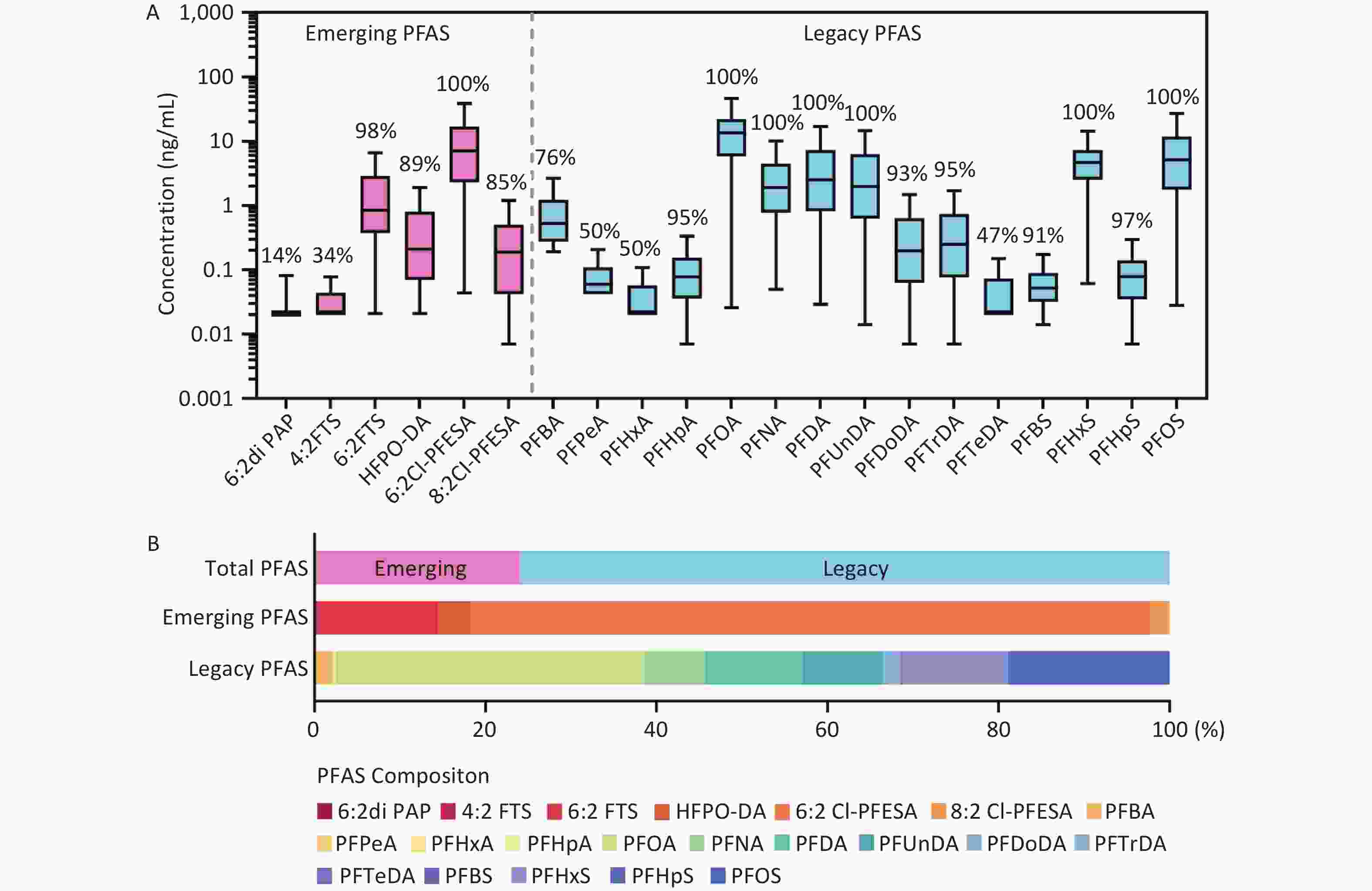
Figure 1. Concentrations (A) and composition (B) of PFAS (ng/mL) detected in plasma samples of the study participants. Boxes display 25th, 50th, and 75th percentiles for PFAS concentrations, and whiskers represent 10th and 90th percentiles, respectively. Values at the middle of the boxes represent the median concentrations, and values above boxes represent the detection rates. 6:2 diPAP, sodium bis(1H,1H,2H,2H-perfluorooctyl) phosphate; 4:2 FTS, 4:2 fluorotelomer sulfonic acid; 6:2 FTS, 6:2 fluorotelomer sulfonic acid; HFPO-DA, hexafluoropropylene oxide dimer acid; 6:2 Cl-PFESA, 6:2 chlorinated polyfluorinated ether sulfonate; 8:2 Cl-PFESA, 8:2 Chlorinated polyfluoroalkyl ether sulfonate; PFBA, perfluorobutanoic acid; PFPeA, perfluoropentanoic acid; PFHxA, perfluorohexanoic acid; PFHpA, perfluoroheptanoic acid; PFOA, perfluorooctanoic acid; PFNA, perfluorononanoic acid; PFDA, perfluorodecanoic acid; PFUnDA, perfluoroundecanoic acid; PFDoDA, Sodium perfluoro-n-dodecanoate; PFTrDA, perfluorotridecanoic acid; PFTeDA, perfluorotetradecanoic acid; PFBS, perfluorobutane sulfonic acid; PFHxS, perfluorohexane sulfonic acid; PFHpS, perfluoroheptane sulfonic acid; PFOS, perfluorooctane sulfonic acid.
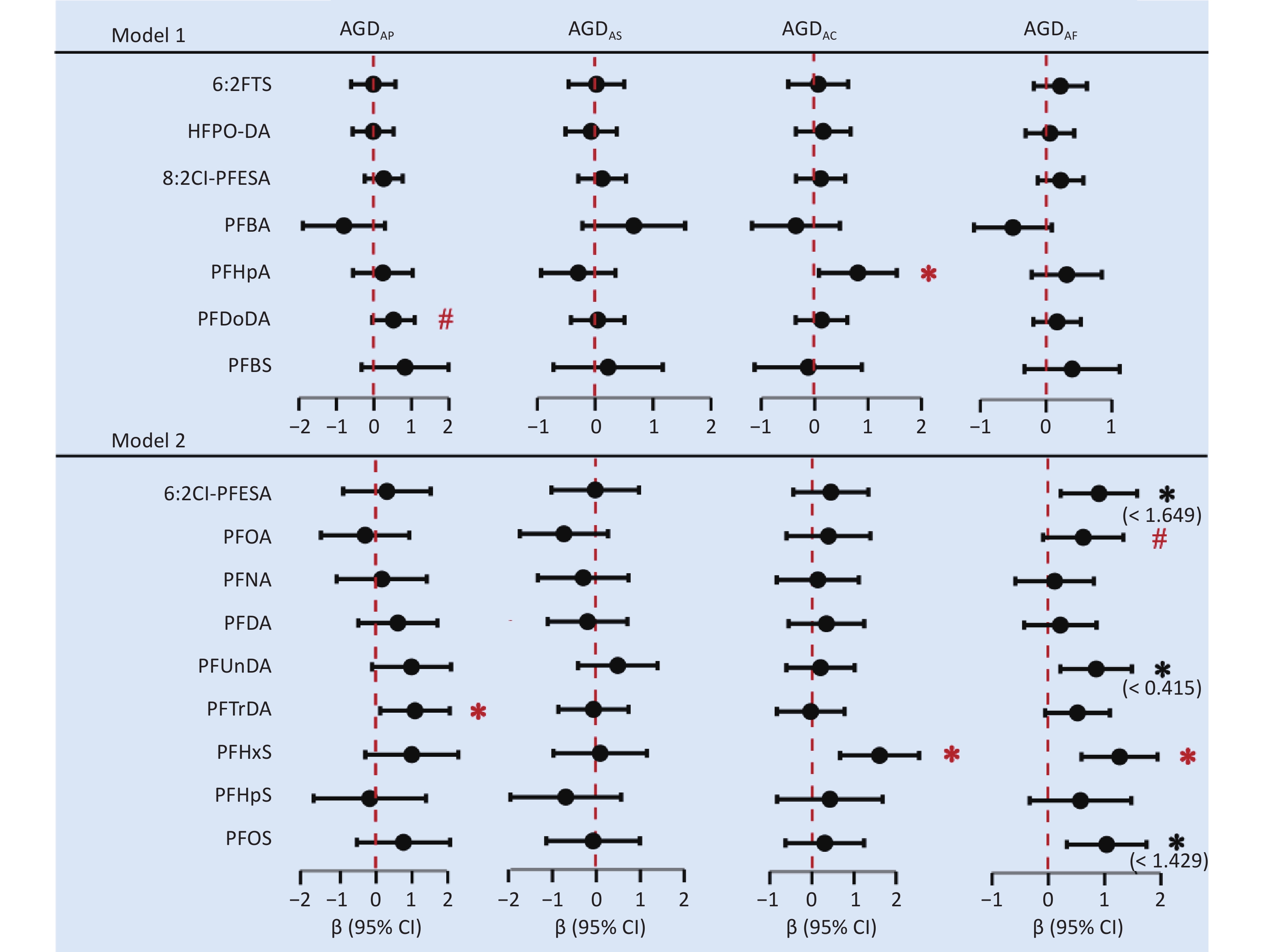
Nine PFAS showed considerable differences between male and female infants (Supplementary Table S7) and were divided into low- and high-exposure groups based on the median concentration (Supplementary Table S8). Sex-specific associations were observed between prenatal PFAS exposure and AGD (Figure 2 and Supplementary Table S9). In male infants, each one-unit increase in log-transformed maternal perfluorotridecanoic acid (PFTrDA) concentration was associated with a 1.08 mm (95% CI: 0.11, 2.06) increase in AGDAP, whereas no significant association was found between maternal PFAS concentrations and AGDAS. In female infants, each one-unit increase in log-transformed maternal concentrations of perfluoroheptanoic acid and PFHxS was associated with increased AGDAC values, and β values (95% CI) were 0.81 (0.08, 1.54) and 1.61 (0.67, 2.55) mm, respectively. A similar positive association was observed between PFHxS and AGDAF (β = 1.27 mm, 95% CI: 0.59, 1.94). Additionally, the results of the RCS model indicated that 6:2 Cl-PFESA, PFUnDA, and PFOS exhibited nonlinear relationships with AGDAF (Supplementary Figure S3); within certain concentration ranges, they showed significant positive associations with AGDAF (Supplementary Table S10). Long-chain PFAS have longer elimination half-lives and higher protein-binding affinities than short-chain PFAS. Therefore, the significant associations observed in this study were primarily concentrated in the long-chain PFAS.
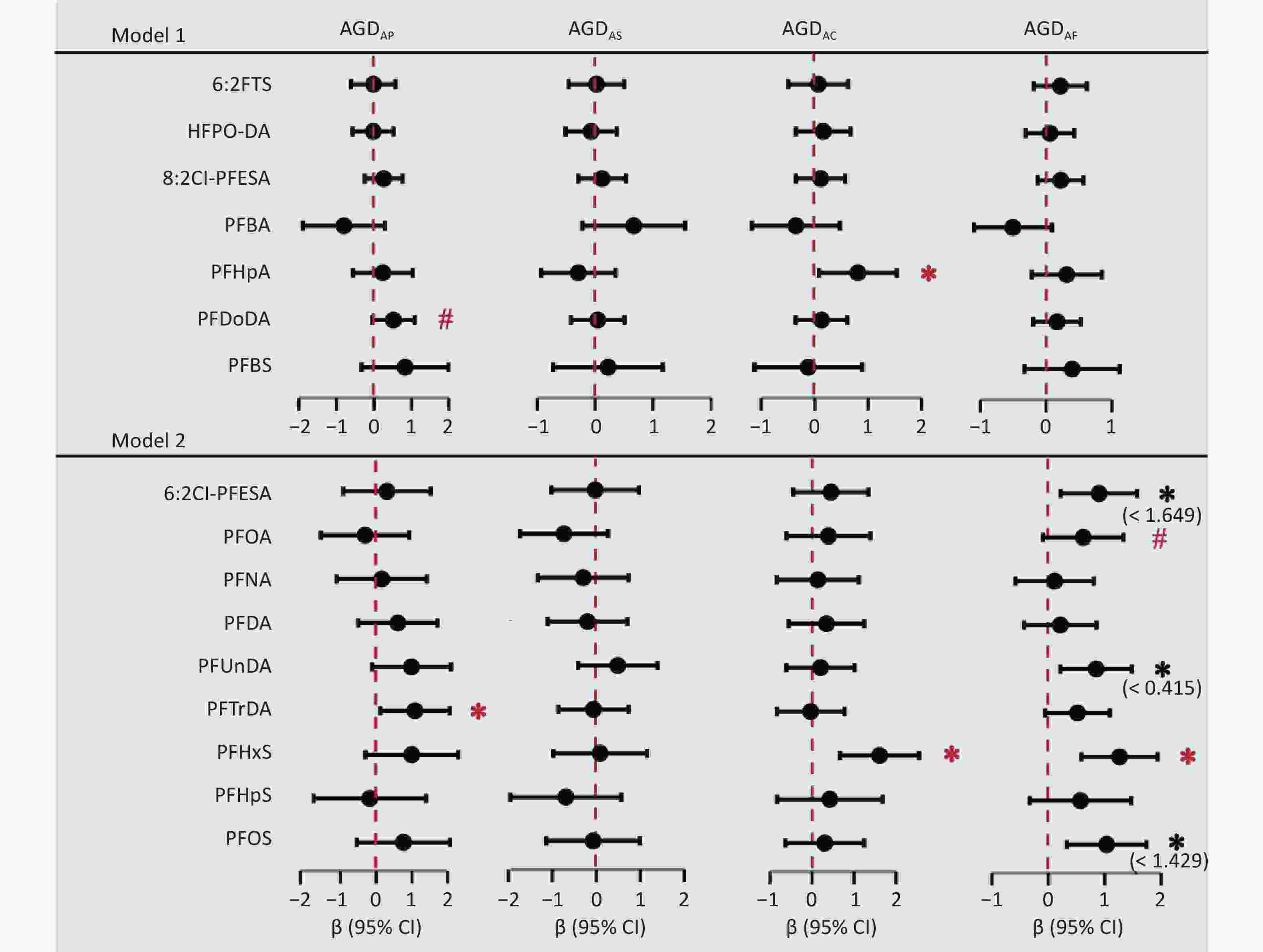
Figure 2. The forest plot displays the adjusted regression coefficients (β) and 95% confidence intervals (95% CIs) derived from separate multivariable linear regression models assessing the associations between each PFAS (log-transformed) congener and AGD. Solid dots represent point estimates (β), and horizontal lines represent the 95% CIs. Model 1 was adjusted for maternal age, gestational age, parity, educational attainment, pre-pregnancy BMI, pre-pregnancy passive smoking, and birth weight. Model 2 was further adjusted for exposure level. BMI, body mass index; CI, confidence interval; AGDAP, anopenile distance; AGDAS, anoscrotal distance; AGDAC, anoclitoris distance; AGDAF, anofourchette distance. 6:2 FTS, 6:2 fluorotelomer sulfonic acid; HFPO-DA, hexafluoropropylene oxide dimer acid; 6:2 Cl-PFESA, 6:2 chlorinated polyfluorinated ether sulfonate; 8:2 Cl-PFESA, 8:2 Chlorinated polyfluoroalkyl ether sulfonate; PFBA, perfluorobutanoic acid; PFHpA, perfluoroheptanoic acid; PFOA, perfluorooctanoic acid; PFNA, perfluorononanoic acid; PFDA, perfluorodecanoic acid; PFUnDA, perfluoroundecanoic acid; PFDoDA, Sodium perfluoro-n-dodecanoate; PFTrDA, perfluorotridecanoic acid; PFBS, perfluorobutane sulfonic acid; PFHxS, perfluorohexane sulfonic acid; PFHpS, perfluoroheptane sulfonic acid; PFOS, perfluorooctane sulfonic acid. * P < 0.05, # P < 0.1.
The association between PFAS exposure and fetal AGD demonstrates sex-specific effects that have also been reported for other endocrine-disrupting chemicals. These differential effects may be explained by the endocrine-disrupting properties and toxicokinetics of PFAS. Specifically, PFAS appear to increase female AGD through estrogenic effects and decrease male AGD through their anti-androgenic activity. In utero PFOS exposure considerably reduced AGD in male rat fetuses, and the concentrations of PFOS, PFDA, PFUnDA, and total PFAS have been associated with a short AGD in wild male minks[5]. However, existing epidemiological evidence shows conflicting associations (positive, negative, or null) between PFAS exposure and AGD in males.
Therefore, the positive associations observed in males in the present study warrant careful interpretation. This discrepancy may reflect differences in the PFAS composition or critical exposure windows during androgen-sensitive periods. Critical exposure windows were established differently across cohorts. For instance, the MIREC study[6] defined it as the first trimester (8–14 weeks), whereas the Faroese Birth[7] Cohort 5 study defined it as late pregnancy (within two weeks before delivery). Consistent with the Shanghai Minhang Birth Cohort Study (S-MBCS), we used maternal PFAS levels at 12–16 gestational weeks to represent exposure during the critical AGD development window (presumed masculinization period of 8–14 weeks). However, our findings are inconsistent with those of the S-MBCS owing to the differences in PFAS exposure levels. For example, our cohort exhibited a substantially higher median concentration of PFTrDA compared with the S-MBCS (0.25 vs. 0.12 ng/mL).
PFAS have been reported to exhibit both estrogen receptor (ER) agonist/antagonist properties and thyroid hormone receptor (THR)-disrupting activity, which can indirectly interfere with the programming of the fetal androgen axis. ER signaling may disrupt fetal Sertoli cell function and estrogen/androgen balance, leading to compensatory endocrine changes. In contrast, PFAS can affect fetal thyroid function either by binding to thyroid hormone transport proteins such as transthyretin or by directly interfering with THR, thereby disrupting the balance of hormonal homeostasis during critical periods of genital development. Additionally, long-chain PFAS may exhibit non-monotonic dose–response effects. At lower exposure levels, the activation of peroxisome proliferator activated receptor (PPAR) by PFAS could indirectly influence the expression of genes involved in steroid hormone synthesis (such as CYP17A1 and CYP19A1) through complex crosstalk among nuclear receptors, potentially leading to temporary or context-dependent enhancement of androgen signaling. In contrast, at higher doses, PFAS may trigger distinct toxicological pathways, resulting in inhibitory effects. However, the effects of PFTrDA on male AGD at higher concentrations require further validation through animal experiments and epidemiological studies.
Furthermore, prenatal PFAS exposure is associated with increased AGDAC or AGDAF in female neonates. This finding is consistent with the results of the S-MBCS and the Shanghai Birth Cohort Study (SBC). Our results showed that PFHxS was positively associated with AGDAC and AGDAF in female infants (P < 0.001), and that PFOS and its alternative 6:2 Cl-PFESA were significantly positively associated with AGDAF within certain concentration ranges (Supplementary Table S10). PFHxS and PFOS exhibits estrogenic effects[8] and may disrupt estrogen homeostasis through multiple mechanisms, including ER activation, enhanced ERα-dependent transcriptional activity, and upregulated expression of estrogen-responsive genes. Hence, prenatal PFAS exposure affects AGD via estrogenic pathways. In addition, in vivo and in vitro studies have shown that certain PFAS inhibit the activity of aromatase, which converts testosterone to estradiol (E2). Altered enzymatic activity may disrupt sex hormone homeostasis, thereby affecting the development of AGD.
A significant positive association was observed between 6:2 Cl-PFESA and AGDAF. 6:2 Cl-PFESA crosses the placental barrier more easily than PFOS, posing a potential threat to fetal development. Epidemiological studies have shown that maternal serum exposure to 6:2 Cl-PFESA is associated with an increased risk of preterm birth and low birth weight, and is accompanied by elevated levels of estrone and E2 in cord blood[9]. We hypothesized that this fetal estrogenic excess perturbs steroidogenic homeostasis, possibly dysregulating androgen metabolism or directly impairing genital tubercle development, thereby increasing AGD. Furthermore, multiple animal experiments have confirmed that long-term, low-concentration exposure to 6:2 Cl-PFESA can considerably impair the reproductive endocrine system through disruption of hormonal homeostasis, induction of oxidative stress, and cellular damage[10].
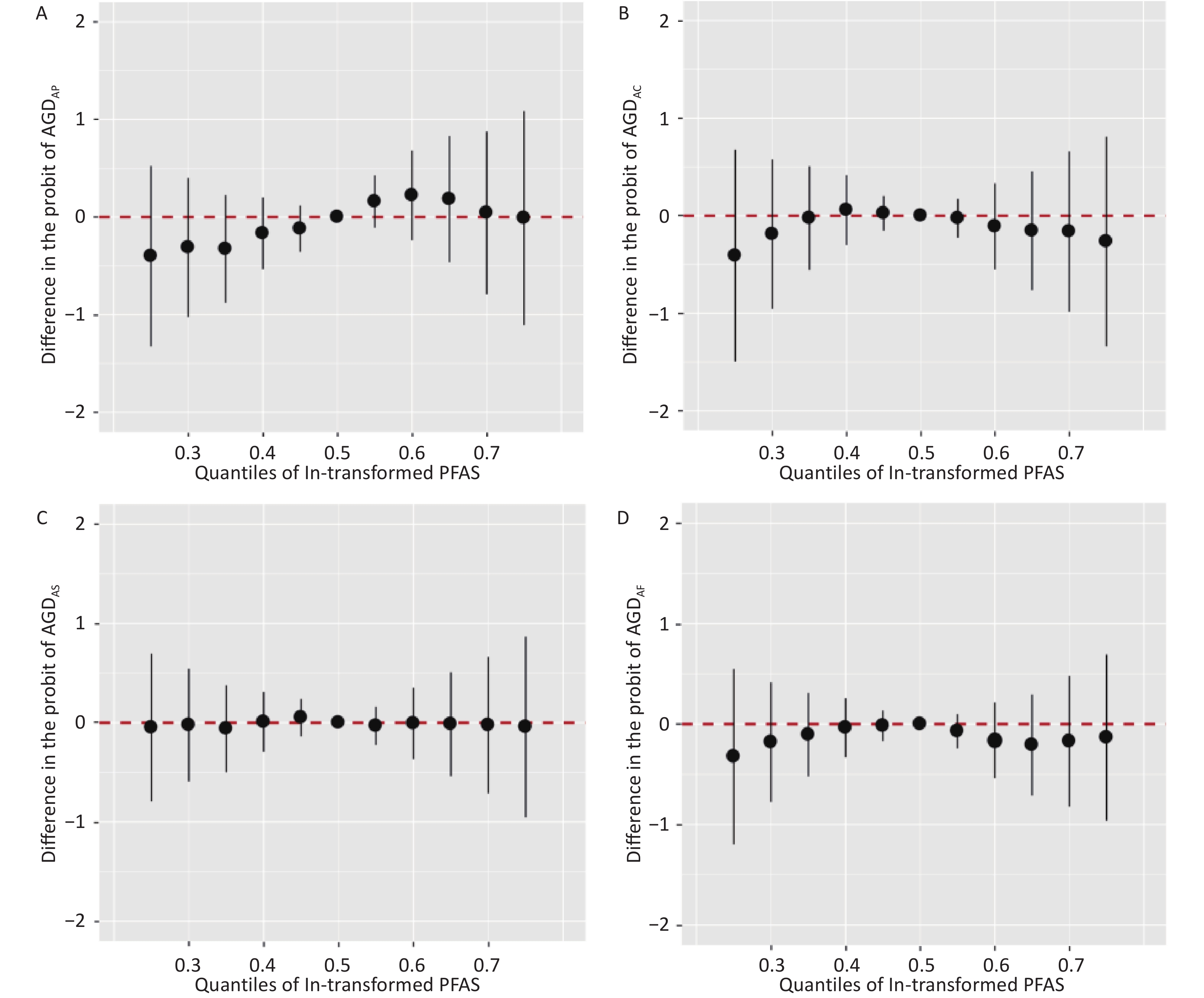
However, in the BKMR model, we did not find any statistically significant exposure-response relationship between mixed PFAS exposure and AGD in male or female infants (Figure 3). These seemingly contradictory results may reveal the complexity of mixture exposure. Different PFAS in the mixture exert opposing effects on reproductive endocrine pathways (e.g., anti-androgenic vs. estrogenic effects), resulting in an overall effect close to zero. In addition, we cannot discount the failure to detect the overall effect owing to the complex nonlinear and interaction patterns within the mixture or the statistical power limitation of the BKMR model under this sample size. Larger sample sizes are required to reliably estimate all potential nonlinearities and interactions.
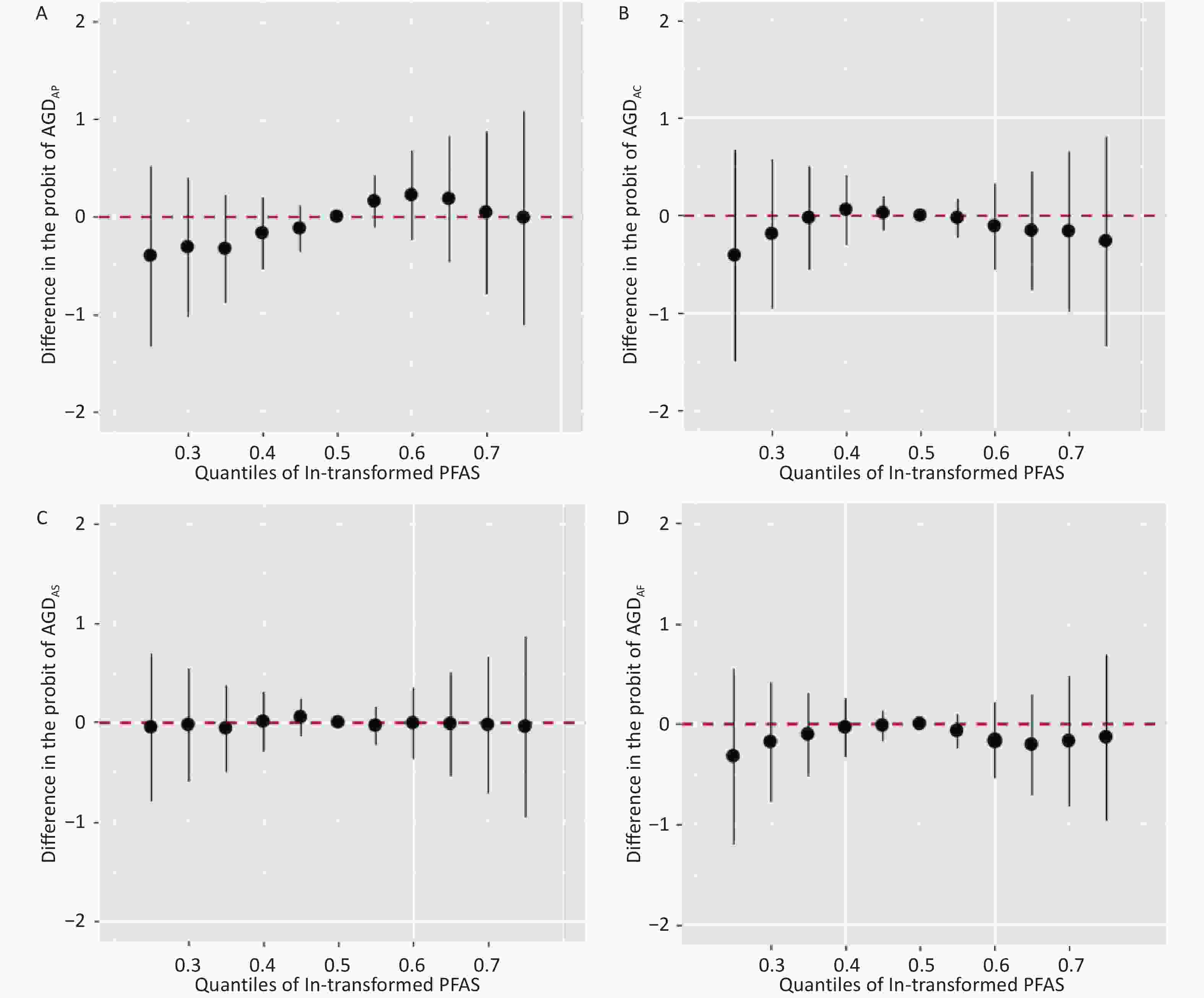
Figure 3. Overall effects of prenatal exposure to PFAS mixture on male or female AGD in BKMR. Overall effects plot the estimated difference in AGD when all 16 PFAS concentrations are fixed at the specified quantiles (0.25–0.75) compared to when all 16 PFAS’s concentrations are fixed at the 50th percentile. Models were adjusted for maternal age, gestational age, parity, educational attainment, pre-pregnancy BMI, pre-pregnancy passive smoking, and birth weight. AGDAP, anopenile distance; AGDAS, anoscrotal distance; AGDAC, anoclitoris distance; AGDAF, anofourchette distance; BKMR, Bayesian kernel machine regression models.
In conclusion, we observed a sex-specific association between PFAS exposure and neonatal AGD, further supporting the hypothesis that prenatal PFAS exposure may disrupt the development of reproductive organs in infants. Notably, 6:2 Cl-PFESA was significantly associated with AGDAF in the female infants. These findings emphasize the need for further research on the potential effects of emerging PFAS on human reproductive health. However, this study was limited by its sample size and statistical power of the associated reductions. Due to the limited number of blood samples, we were unable to analyze other environmental chemicals. When PFAS are highly correlated with other chemicals, the possibility that they act as substitutes for other contaminants cannot be discounted.
HTML
Competing Interests The authors declare that they have no competing interests.
Ethics This study was approved by the Institutional Review Board of Shanghai Institute of Planned Parenthood Research (Approval No. PJ2018-19), China. All participants provided written informed consent prior to participation.
Authors’ Contributions Writing-original draft, data curation, and formal analysis: Yanmei Wu and Mingzhu Wu. Methodology, conceptualization, and supervision: Hong Liang, Honglei Ji, Hongkai Zhu, and Jingchuan Xue. Funding acquisition, writing, review, and editing: Jianya Xi, and Maohua Miao.
Data Sharing Supplementary materials will be available in www.besjournal.com.
Reference







 Quick Links
Quick Links
 DownLoad:
DownLoad: