-
Central obesity, characterized by excess fat accumulation in the abdomen, remains a major health challenge and a strong predictor of cardiometabolic diseases and mortality compared with general obesity[1-3]. National surveillance surveys (2000–2020) showed that the prevalence of central obesity among adults aged 20–59 years increased to 32.1% by 2020, underscoring its substantial public health burden[4].
Adequate sleep plays a crucial role in metabolic function. Adults aged 18–64 years are recommended to sleep for 7–9 h[5]; however, the prevalence of insufficient sleep in China increased sharply from 11.8% in 2010 to 24.1% in 2016[6]. Several epidemiological studies, such as the Prospective Urban Rural Epidemiology (PURE) study and the China Health and Retirement Longitudinal Study (CHARLS), have investigated the association between sleep duration and central obesity[7-12]. However, these studies reported inconsistent findings owing to several limitations, including relatively small sample sizes, populations drawn from a single province or city, and specific subgroups. Therefore, assessing the association between sleep duration and central obesity in a large Chinese population remains warranted.
Physical activity (PA) reduces waist circumference (WC) and is inversely associated with the risk of cardiovascular disease[13-15]. Changes in moderate-to-vigorous physical activity (MVPA) or sleep duration inevitably alter other behaviors, thereby affecting the risk of central obesity. Epidemiological studies have shown significant associations between substituting sleep duration with PA and reductions in WC[16,17]. However, these studies were primarily conducted in European populations, and evidence from Chinese adults remains limited.
This study aimed to evaluate the relationships of sleep duration and PA with central obesity in the Prediction for Atherosclerotic Cardiovascular Disease Risk in China (China-PAR) project and provide evidence for obesity management in China.
-
All participants were from three subcohorts of the China-PAR project: the International Collaborative Study of Cardiovascular Disease in Asia (InterASIA), the China Multi-Center Collaborative Study of Cardiovascular Epidemiology (ChinaMUCA-1998), the Community Intervention of Metabolic Syndrome in China & Chinese Family Health Study (CIMIC). The InterASIA study enrolled 15,540 participants using a four-stage stratified sampling method. The ChinaMUCA-1998 study included 11,480 participants at baseline using cluster sampling. For these two subcohorts, the first follow-up survey was conducted between 2007 and 2008. Using cluster sampling, the CIMIC project enrolled 86,428 participants who completed surveys in 2007–2008. A subsequent follow-up survey was conducted between 2012 and 2015 in all three subcohorts. Detailed descriptions of these cohorts are available elsewhere[18].
This study included individuals who participated in the 2007–2008 or 2012–2015 surveys and had complete data on sleep duration, PA, and WC. In the 2007–2008 survey (N = 104,957), participants without information on WC (N = 260), sleep duration (N = 245), or PA (N = 73) were excluded, leaving 104,379 individuals for the final analyses. In the 2012–2015 survey (N = 82,289), participants without information on WC (N = 2,279), sleep duration (N = 745), or PA (N = 8,271) were excluded, leaving 70,994 individuals for the final analyses. In total, 175,373 observations from 106,518 individuals were included in the analysis (Figure S1). Excluded individuals were younger and more highly educated, making them more likely to miss the on-site examination due to work. However, comparisons of lifestyle and clinical characteristics showed no meaningful differences between included and excluded participants, suggesting a low risk of sampling bias (Supplementary Table S1).
The study was approved by the Ethics Committee of Fuwai Hospital, Chinese Academy of Medical Sciences (Approval No. 2012-399), and written informed consent was obtained from all participants.
-
Fasting blood specimens were collected to measure blood glucose and lipid levels, including total cholesterol (TC), triglycerides (TG), and high-density lipoprotein cholesterol (HDL-C). Low-density lipoprotein cholesterol (LDL-C) concentrations were calculated for participants with TG levels < 400 mg/dL using the Friedewald equation[19]:
$$ \text{LDL-C(mg/dL)=TC}-\text{HDL-C}-\frac{\text{TG}}{\text{5}} $$ -
During face-to-face interviews, standardized questionnaires were used to collect information on daily activities and the time spent on each. Data on sleep duration and the type and duration of daily activities were collected using a uniform questionnaire, including the average time per day spent on high-, moderate-, and low-intensity PA, sleep, and other sedentary behaviors for each participant over the past year. The duration of each type of daily activity was recorded separately for workdays and non-workdays, and final activity durations were calculated as weighted averages.
Participants were categorized into three groups based on sleep duration: short (< 7 h/day), recommended (7 – < 9 h/day), and long (≥ 9 h/day). Adequate PA was defined as engaging in at least 150 min of MVPA per week, calculated as follows[20]:
$$ {\text{Time}}_{\text{MVPA}}={\text{Time}}_{\text{moderate PA}}+{\text{Time}}_{\text{vigorous PA}}\times \text{2} $$ -
WC was measured twice for each participant, 1 cm above the navel during light breathing, and the two measurements were averaged to reduce potential measurement bias. According to the consensus of Chinese experts, central obesity was defined as WC ≥ 90 cm in males and ≥ 85 cm in females[21].
-
Self-reported data were collected using standardized questionnaires, including age, sex, residence (urban or rural), educational level, smoking status, drinking status, dietary habits, personal medical history, and family history. Residence was classified as urban or rural based on self-reported information. Body weight and height were measured twice for each participant without shoes or heavy clothing, and the averages were used to calculate body mass index.
Participants were classified into two groups according to whether they reported having a high school education or higher. Tobacco smoking was defined as having smoked at least 100 cigarettes over one’s lifetime. Drinking was defined as consuming an alcoholic beverage ≥ 12 times in the past year. Dietary intake was assessed by recording self-reported amounts and frequencies of consumption for each food item. A healthy diet was defined consistently with previous studies[22] as meeting at least two of the following five criteria: intake of vegetables and fruits ≥ 500 g/day, fish ≥ 200 g/week, legumes ≥ 125 g/day, red meat < 75 g/day, and tea consumption ≥ 3 times/week[23]. Diabetes was defined as a fasting glucose level ≥ 126 mg/dL and/or current use of hypoglycemic agents[24]. Cardiovascular disease and malignant tumors were identified based on a prior physician diagnosis.
-
Continuous variables are presented as means ± standard deviations, and categorical variables are expressed as frequencies (percentages). Generalized estimating equations (GEE) were used to assess the associations of adequate PA and sleep duration with central obesity, with results reported as effect sizes or odds ratios (ORs) and 95% confidence intervals (CIs). Three models were constructed as follows: Model 1 was adjusted for age, sex, and cohort source; Model 2 was further adjusted for residence and educational level; and Model 3 was additionally adjusted for healthy diet, smoking status, and drinking status, with mutual adjustment for adequate PA and sleep duration. Because the GEE approach incorporates all available repeated measurements rather than excluding individuals due to missing data at a single wave, the potential risk of selection bias was minimized.
Subgroup analyses stratified by age, sex, residence, and educational level were conducted to assess the robustness of the results. Effect modification was evaluated by including multiplicative interaction terms between sleep duration or PA and each stratification variable in the models, with the corresponding P values reported as P for interaction. In addition, associations of sleep duration with WC and central obesity were examined after stratification by PA, and their combined effects were assessed by incorporating multiplicative interaction terms. Sensitivity analyses were performed after excluding participants with diabetes (N = 14,157), cardiovascular disease (N = 8,097), and malignant tumors (N = 906).
In addition, participants were classified into six groups based on joint categories of sleep duration (three groups) and PA status (adequate vs. inadequate). Participants with recommended sleep duration and adequate PA served as the reference group for the remaining five groups.
The median sleep duration (8 h/day) was used as the cutoff point. Using the isotemporal substitution model (ISM), which keeps total time spent on sleep and MVPA constant, the theoretical effects of substituting one behavior for another were evaluated[25,26]. The formula was as follows:
$$ \text{f(y)}={\text{β}}_{\text{0}}+{\text{β}}_{\text{1}}\times \text{ST}+{\text{β}}_{\text{2}}\times \text{MVPA}+{\text{β}}_{\text{3}}\times \text{TA}+\text{X} $$ Where ST represents sleep duration (30 min/day), MVPA denotes moderate-to-vigorous PA (30 min/day), TA indicates total time spent on sleep and MVPA (30 min/day), and X represents other covariates, f(y) denotes the outcome variable, and β0 represents the intercept. When examining the effects of substituting MVPA time for sleep duration, β1 is set to 0, with β2 indicating the effect after substituting 30 min, and vice versa. β3 serves only to constrain total time and is not interpreted directly.
Statistical analyses were performed using SAS (version 9.4) and R (version 4.2.1). A two-tailed P < 0.05 was considered statistically significant.
-
The characteristics of participants in each survey are presented in Table 1. The mean age was (52.7 ± 12.5) years in the 2007–2008 survey and (59.4 ± 11.5) years in the 2012–2015 survey, with 39.7% and 38.4% male participants, respectively. In the 2007–2008 survey, the mean sleep duration was (8.0 ± 1.3) h/day, and 66.0% of participants met the criteria for adequate PA. In the 2012–2015 survey, the mean sleep duration was (7.9 ± 1.3) h/day, and 63.0% of participants met the adequate PA criteria. The mean WC was (81.5 ± 10.2) cm in 2007–2008 and (82.7 ± 9.8) cm in 2012–2015, and the corresponding prevalence of central obesity was 29.8% and 32.9%, respectively (Table 1). Among participants with central obesity, 60.4% achieved adequate PA, and 64.5% achieved the recommended sleep duration (Table S2).
Variables The 2007–2008 survey
(N=104,379)The 2012–2015 survey
(N=70,994)Age, year 52.7 ± 12.5 59.4 ± 11.5 Male, N (%) 41,476 (39.7) 27,237 (38.4) Urban resident, N (%) 7,820 (7.5) 4,437 (6.2) High school and above, N (%) 14,437 (13.8) 7,915 (11.2) Smoker, N (%) 25,540 (24.5) 15,337 (21.8) Drinker, N (%) 21,638 (20.8) 10,950 (15.6) Adequate physical activity, N (%) 68,852 (66.0) 44,723 (63.0) Healthy diet, N (%) 71,374 (68.4) 48,309 (68.1) Diabetes, N (%) 6,779 (6.9) 7,378 (11.7) Central obesity, N (%) 31,053 (29.8) 23,352 (32.9) BMI, kg/m2 23.9 ± 3.6 24.6 ± 3.6 WC, cm 81.5 ± 10.2 82.7 ± 9.8 SBP, mmHg 130.3 ± 21.6 132.7 ± 18.2 DBP, mmHg 79.4 ± 11.9 79.9 ± 10.3 TC, mg/dL 174.3 ± 35.5 185.1 ± 38.3 TG, mg/dL 144.9 ± 107.0 138.9 ± 96.2 HDL-C, mg/dL 51.3 ± 12.8 54.3 ± 18.6 LDL-C, mg/dL 95.6 ± 28.8 104.0 ± 33.4 Glucose, mg/dL 92.3 ± 27.6 98.8 ± 29.4 Sleep duration, h/day 8.0 ± 1.3 7.9 ± 1.3 Sleep duration, N (%) Short (< 7 h/day), N (%) 11,591 (11.1) 9,517 (13.4) Recommended (7 -< 9 h/day), N (%) 69,660 (66.8) 47,556 (67.2) Long (≥ 9 h/day), N (%) 23,007 (22.1) 13,716 (19.4) Note. Data are presented as mean ± standard deviation or frequency (percentage). BMI, body mass index; DBP, diastolic blood pressure; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; SBP, systolic blood pressure; TC, total cholesterol; TG, triglyceride; WC, waist circumference. Table 1. Basic characteristics for participants of different survey
-
Both short and long sleep durations were significantly associated with increased WC and a higher risk of central obesity (Table 2). In the fully adjusted model (Model 3), compared with participants who had the recommended sleep duration, those with short sleep duration had an increase in WC of 0.15 cm (95% CI: 0.04, 0.27) and a 5% higher risk of central obesity (OR = 1.05, 95% CI: 1.02, 1.08). Participants with long sleep duration showed an increase in WC of 0.27 cm (95% CI: 0.18, 0.35) and a 9% higher risk of central obesity (OR = 1.09, 95% CI: 1.07, 1.12). Adequate PA had beneficial effects in preventing central obesity, with a decrease in WC of 0.39 cm (95% CI: −0.47, −0.32) and a 10% lower risk of central obesity (OR = 0.90, 95% CI: 0.88, 0.91) (Supplementary Table S3).
Outcomes Model 1 Model 2 Model 3 Waist circumference (cm) Recommended (7 –< 9 h/day) Reference Reference Reference Short (< 7 h/day) 0.17 (0.06, 0.28) 0.15 (0.04, 0.27) 0.15 (0.04, 0.27) Long (≥ 9 h/day) 0.25 (0.17, 0.34) 0.27 (0.18, 0.36) 0.27 (0.18, 0.35) Central obesity (odds ratio) Recommended (7 –< 9 h/day) Reference Reference Reference Short (< 7 h/day) 1.05 (1.02, 1.08) 1.05 (1.02, 1.08) 1.05 (1.02, 1.08) Long (≥ 9 h/day) 1.09 (1.07, 1.12) 1.09 (1.07, 1.12) 1.09 (1.07, 1.12) Note. Model 1: adjusted for age, sex, and cohort; Model 2: Model 1 + residence, and educational level; Model 3: Model 2 + healthy diet, smoking status, drinking status, and adequate physical activity. Table 2. Association of sleep duration with waist circumference and central obesity (effect size and 95% confidence interval)
The associations of sleep duration and PA with WC and central obesity stratified by age, sex, residence, and educational level are shown in Supplementary Table S4 and Supplementary Figure S2. Significant associations of suboptimal sleep duration and PA with WC and central obesity were observed in most subgroups. For example, females with long sleep duration had an increase in WC of 0.44 cm (95% CI: 0.33, 0.56) and a 13% higher risk of central obesity (OR = 1.13, 95% CI: 1.10, 1.16), which was greater than that observed in males; in contrast, short sleep duration had a greater adverse effect on WC and central obesity in males than in females (P for interaction < 0.001). Compared with adequate PA, inadequate PA was more strongly associated with increased WC and a higher risk of central obesity among participants aged ≥ 65 years and among males. The effect size of short sleep duration on central obesity was greater among urban residents (urban: OR = 1.09, 95% CI: 1.00, 1.18; rural: OR = 1.04, 95% CI: 1.01, 1.08; P for interaction < 0.001) and among those with higher educational attainment (high school and above: OR = 1.16, 95% CI: 1.07, 1.25; less than high school: OR = 1.03, 95% CI: 1.00, 1.07; P for interaction < 0.001). In addition, the significant associations of sleep duration and PA with WC and central obesity remained consistent after excluding individuals with diabetes, cardiovascular disease, or malignant tumors (Supplementary Table S3).
-
Table 3 shows the association of sleep duration with WC and central obesity stratified by PA. Among individuals with inadequate PA, compared with those with the recommended sleep duration, short sleep duration was associated with an increase in WC of 0.43 cm (95% CI: 0.23, 0.63) and a 10% higher risk of central obesity (OR = 1.10, 95% CI: 1.05, 1.15), while long sleep duration was associated with an 8% higher risk of central obesity (OR = 1.08, 95% CI: 1.04, 1.12). Even among individuals with adequate PA, those with long sleep duration still exhibited a significant increase in WC of 0.56 cm (95% CI: 0.44, 0.67) and a 14% higher risk of central obesity (OR = 1.14, 95% CI: 1.11, 1.18). After incorporating interaction terms between sleep duration groups and PA into the model, evidence of an interaction effect of sleep duration and PA on WC and the risk of central obesity was identified (both P for interaction < 0.001).
Physical activity Recommended sleep duration
(7 –< 9h/day)Short sleep duration
(< 7 h/day)Long sleep duration
(≥ 9 h/day)P for interaction Waist circumference (cm) Adequate Reference 0.00 (−0.16, 0.15) 0.56 (0.44, 0.67) < 0.001 Inadequate Reference 0.43 (0.23, 0.63) 0.14 (−0.03, 0.31) Central obesity (odds ratio) Adequate Reference 1.03 (0.99, 1.07) 1.14 (1.11, 1.18) < 0.001 Inadequate Reference 1.10 (1.05, 1.15) 1.08 (1.04, 1.12) Note. All models were adjusted for age, sex, cohort, residence, educational level, healthy diet, smoking status, and drinking status. Table 3. Associations of sleep duration with waist circumference and central obesity stratified by physical activity (effect size and 95% confidence interval)
We further assessed the joint association of sleep duration (7 –< 9 h/day, < 7 h/day, ≥ 9 h/day) and adequate PA with WC and central obesity (Figure 1). Except for individuals with short sleep duration who achieved adequate PA, individuals in the remaining four groups had increased WC and a higher risk of central obesity compared with those who had adequate PA and the recommended sleep duration. Participants with inadequate PA and short sleep duration exhibited the most pronounced increase in WC (0.88 cm, 95% CI: 0.70, 1.05) and the highest risk of central obesity (OR = 1.23, 95% CI: 1.18, 1.29), corresponding to a 23% higher risk than the reference group. This significant joint association remained consistent after excluding individuals with diabetes, cardiovascular disease, or malignant tumors (Supplementary Table S5).
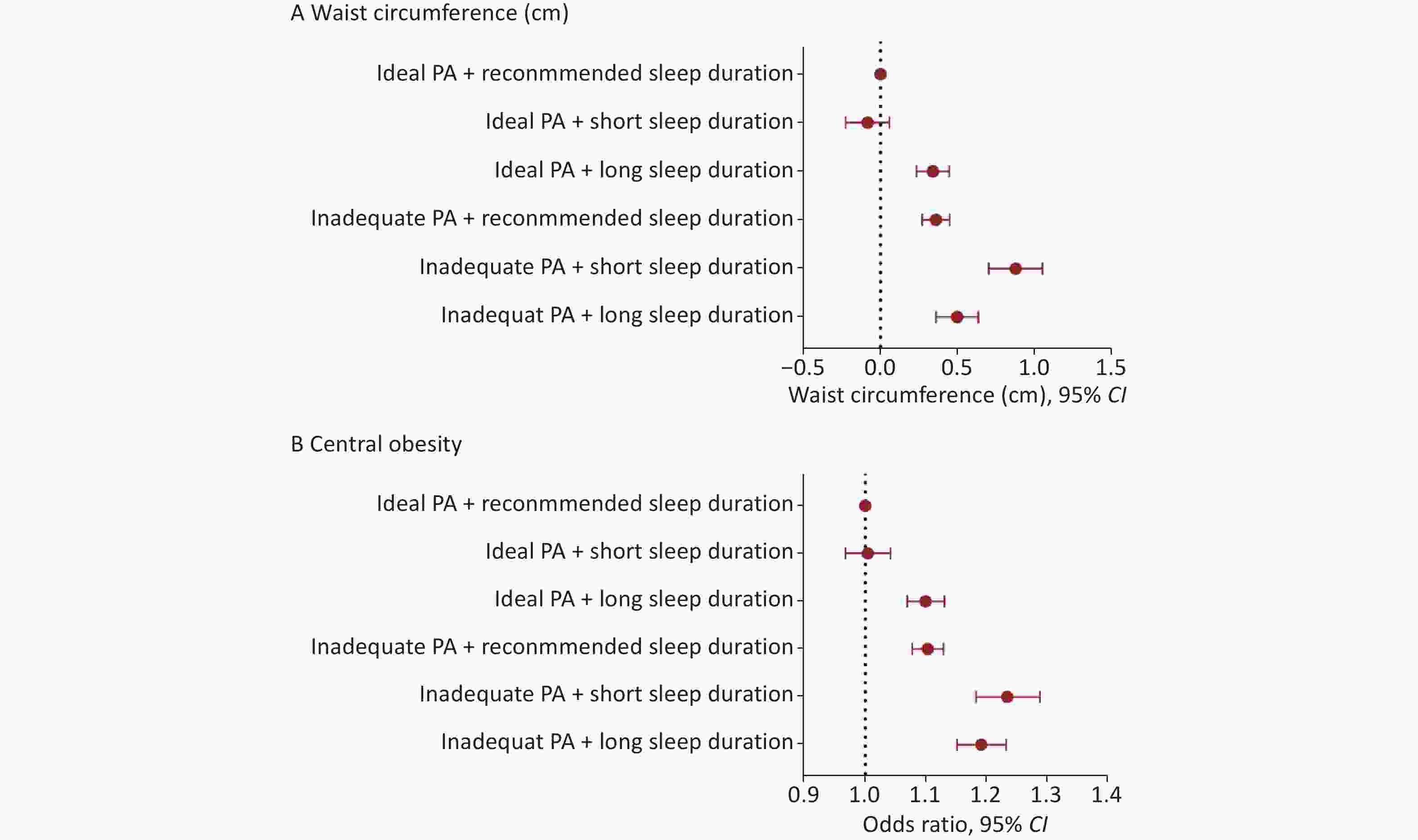
Figure 1. The joint association of sleep duration and physical activity with waist circumference and central obesity.All models were adjusted for age, sex, cohort, residence, educational level, healthy diet, smoking status, drinking status, and with mutual adjustment between adequate PA and sleep duration. CI: confidence interval; PA: physical activity.
-
Replacing 30 min/day of MVPA with sleep showed a beneficial effect on central obesity among individuals sleeping ≤ 8 h/day but had harmful effects on WC and central obesity among those sleeping > 8 h/day, whereas the reverse substitution yielded more pronounced opposite effects (Table 4). Specifically, among individuals sleeping > 8 h/day, replacing 30 min/day of MVPA with sleep was associated with an increase in WC of 0.09 cm (95% CI: 0.04, 0.13) and a 3% higher risk of central obesity (OR = 1.03, 95% CI: 1.02, 1.04). Conversely, substituting 30 min/day of sleep with MVPA was associated with a decrease in WC of 0.09 cm (95% CI: −0.13, −0.04) and a 3% lower risk of central obesity (OR = 0.97, 95% CI: 0.96, 0.98). Among individuals sleeping ≤ 8 h/day, replacing 30 min/day of MVPA with sleep was significantly associated with a reduced risk of central obesity (OR = 0.99, 95% CI: 0.99, 1.00), while replacing sleep with MVPA increased the risk (OR = 1.01, 95% CI: 1.00, 1.01), although changes in WC were relatively small.
Replacing 30 min/day of MVPA with sleep Replacing 30 min/day of sleep with MVPA Waist circumference (cm) Sleep duration ≤ 8 h/day −0.02 (−0.04, 0.01) 0.02 (−0.01, 0.04) Sleep duration > 8 h/day 0.09 (0.04, 0.13)* −0.09 (−0.13, −0.04)* Central obesity (odds ratio) Sleep duration ≤ 8 h/day 0.99 (0.99, 1.00)* 1.01 (1.00, 1.01)* Sleep duration > 8 h/day 1.03 (1.02, 1.04)* 0.97 (0.96, 0.98)* Note. * Represents the statistically significance (P < 0.05). All models were adjusted for age, sex, cohort, residence, educational level, healthy diet, drinking status and smoking status. MVPA: moderate to vigorous physical activity. Table 4. Effects of isotemporal substitution between sleep duration and physical activity on waist circumference and central obesity (effect size and 95% confidence interval)
-
This study demonstrates that suboptimal sleep durations (< 7 h/day and ≥ 9 h/day) are associated with increased WC and a higher risk of central obesity in Chinese adults, whereas adequate PA mitigates these effects. Replacing 30 min/day of sleep with MVPA in individuals with long sleep duration, and replacing 30 min/day of MVPA with sleep in those with short sleep duration, significantly reduced the risk of central obesity. Individuals with short sleep duration and insufficient PA have the greatest risk of central obesity and should consider improving their lifestyle behaviors.
Previous studies have shown that the association between sleep duration and the risk of central obesity remains controversial. Studies based on the China Kadoorie Biobank reported that participants with short sleep duration had a higher risk of central obesity than those with long sleep duration[12]. In contrast, the Swedish Women’s Study demonstrated that both habitual short and long sleepers had a higher prevalence of central obesity than those with optimal sleep duration at follow-up among females aged < 40 years, but not among those aged ≥ 40 years[10]. The China Health and Retirement Longitudinal Study observed an association between long sleep duration and central obesity in older adults and in male subgroups[7]. Studies based on the Dongfeng-Tongji cohort[9] and research from Taiwan[27] found no association between sleep duration and central obesity risk, likely due to limited populations drawn from a single province or city. This study, which included over 100,000 participants from 15 provinces in China, found that both short and long sleep durations were associated with higher WC and an increased risk of central obesity, providing epidemiological evidence for the prevention of central obesity and related metabolic diseases.
Several mechanisms have been proposed to explain the effects of sleep on central obesity. In individuals with suboptimal sleep, levels of various inflammatory markers often change, promoting fat accumulation and contributing to central obesity[28,29]. Studies have shown that alterations in immune cells or serum immune markers in individuals with suboptimal sleep can impair immune function[30]. Immune cells produce inflammatory cytokines that disrupt adipocyte homeostasis and metabolism, thereby promoting obesity[30-33]. Individuals with suboptimal sleep also tend to have lower PA levels and a higher risk of obesity[34]. In addition, short sleep duration is associated with decreased leptin secretion, increased ghrelin levels, reduced satiety, and increased appetite, thereby increasing energy intake and obesity risk[35].
Consistent with previous studies, this study found that adequate PA was significantly associated with reduced WC and a lower risk of central obesity. The Fifth Korean National Health and Nutrition Examination Survey reported that the prevalence of central obesity was significantly lower among individuals engaging in MVPA than among those engaging in low-intensity PA[36]. PA influences the risk of central obesity by directly stimulating skeletal muscles to secrete myokines, altering oxidative stress and inflammatory responses, and improving insulin sensitivity, thereby affecting fat metabolism and accumulation[37-39].
In this study, we evaluated the combined interaction and isotemporal substitution effects of sleep duration and PA on central obesity. Our analysis showed that even among individuals achieving adequate PA, longer sleep duration remained significantly associated with a higher risk of central obesity. In the short sleep duration group, a significant difference in WC was observed between those who met the adequate PA criteria and those who did not; a similar difference was found in the long sleep duration group. The study also revealed that individuals with short sleep duration and inadequate PA had the greatest increase in WC and the highest risk of central obesity. Previous studies on PA substitution have consistently shown that replacing time spent on other activities with MVPA provides significant health benefits. However, data on the relationship between sleep duration substitution and central obesity remain limited.
More recently, the Prospective Physical Activity, Sitting and Sleep consortium (ProPASS) and the Prevention of Diabetes through Lifestyle Intervention and Population Studies in Europe and around the World (PREVIEW) studies reported that reallocating sleep time to MVPA was significantly associated with smaller WC[16,40], consistent with our findings. The results of this study also suggest that the impact of sleep reallocation depends on an individual’s sleep duration. Although associations in the ISM showed small effect sizes and may have limited clinical relevance, the high prevalence of risk factors and supporting physiological mechanisms indicate that these findings warrant consideration for their potential impact at the population level.
Subgroup analyses revealed sex-specific associations: short sleep duration was more detrimental for males, whereas long sleep duration was more detrimental for females, consistent with previous findings[41,42]. Females generally obtain more total sleep but experience poorer sleep quality, characterized by more frequent nocturnal awakenings. In contrast, males are more prone to delayed sleep onset and greater social jetlag[43]. In females, prolonged but fragmented sleep may impair emotional regulation by disrupting prefrontal control over limbic activity[44], potentially promoting obesity through stress-related or emotional eating[45]. Inefficient, extended sleep may also contribute to reduced PA, lower basal metabolic rate, and increased systemic inflammation[46,47]. In males, short sleep duration is often accompanied by late chronotype and circadian misalignment, which activate the sympathetic and hypothalamic–pituitary–adrenal axis, leading to elevated cortisol levels and impaired insulin sensitivity[48-50]. Moreover, short sleep reduces circulating leptin and increases ghrelin concentrations in healthy young men, enhancing hunger and caloric intake, particularly from energy-dense foods[35]. Collectively, these sex-specific neuroendocrine and behavioral changes promote a positive energy balance and increase the risk of obesity in males.
This study has several strengths. First, it leveraged the China-PAR cohort, a large, nationally representative sample of the Chinese population, providing a robust foundation for examining the impact of sleep and PA on central obesity. Second, it demonstrated that both short and long sleep durations are associated with an increased risk of central obesity in a large population. Third, the study evaluated the isotemporal substitution effects of MVPA and sleep, indicating that long sleepers may benefit from replacing some sleep time with MVPA, while short sleepers should prioritize sufficient sleep, highlighting practical implications for public health. Additionally, all physical measurements were conducted under strict quality control, ensuring high accuracy and reliability.
However, the study also has several limitations. First, its cross-sectional design limits causal inference, as exposures and outcomes were assessed simultaneously; nevertheless, the use of repeated measurements, a large sample size, and GEE provides a more robust analytical framework than a purely cross-sectional design, making the study valuable for hypothesis generation and guiding future longitudinal research. Second, exposure assessments were based on self-reported information rather than objective measures, which may inevitably lead to exposure misclassification. Third, the ISM results are theoretical and do not reflect actual behavioral shifts. The model considers only MVPA and sleep, whereas real-life scenarios are more complex, so caution is needed when generalizing these findings. Fourth, the study did not adjust for total caloric intake, potentially introducing bias; however, adjustment for adherence to a healthy diet may partially account for differences in diet quality. Fifth, the surveys were conducted in 2007–2008 and 2012–2015, so the potential influence of more recent behavioral and environmental changes on these associations cannot be entirely ruled out. Future studies using updated datasets are warranted to validate and extend these findings.
-
This study identified suboptimal sleep duration as a significant risk factor for increased WC and central obesity, while adequate PA had protective effects in Chinese adults. Notably, individuals with short sleep duration and inadequate PA showed the greatest increases in WC and central obesity risk, underscoring the importance of lifestyle modifications. Furthermore, the effects of reallocating sleep duration appeared to depend on total sleep time. These findings emphasize the value of promoting both healthy sleep and regular PA as key strategies for preventing obesity at the population level.
HTML
Study Populations
Laboratory Assessment
Exposure Assessment
Outcome Definition
Assessment of Covariates
Statistical Analysis
Basic Characteristics of the Study Population
Association of Sleep Duration and PA with Central Obesity
Joint Associations of Sleep Duration and PA with Central Obesity
Isotemporal Substitution between Sleep and MVPA on Central Obesity
Ethical Approval The study was approved by the Ethics Committee of Fuwai Hospital, Chinese Academy of Medical Sciences (Approval No. 2012-399), and informed consent was obtained from all participants.
Authors’ Contributions Methodology, Data curation, Formal analysis, Visualization, Writing - original draft: Youjing Zhang; Data curation, Formal analysis, Visualization, Writing - original draft: Meiling Hu, and Ziyi Yang; Investigation, Data curation, Writing - review & editing: Jianxin Li; Investigation and Data curation: Jie Cao, Jichun Chen, Fangchao Liu, Keyong Huang, Hongfan Li, Chong Shen, Dongsheng Hu, Xiaoqing Liu, Shujun Gu, and Ling Yu; Supervision, Writing - review & editing: Jianfeng Huang; Resources, Project administration, Supervision, Funding acquisition, Writing - review & editing: Xiangfeng Lu; Resources, Project administration, Supervision, Funding acquisition, Writing - review & editing: Dongfeng Gu; Conceptualization, Project administration, Methodology, Supervision, Funding acquisition, Writing - review & editing: Shufeng Chen; All authors contributed to the interpretation of the study findings, provided critical review of the manuscript, and approved the final version for publication.
Data Sharing The data underlying this article will be shared on reasonable request to the corresponding author.
Disclosure The funding sources of this study had no role in study design, data collection and analysis, decision to publish, or preparation of the article.
&These authors contributed equally to this work.







 Quick Links
Quick Links
 DownLoad:
DownLoad: