-
Decabromodiphenyl ethane (DBDPE) is a brominated flame retardant (BFR) used as an additive of flammable material to decrease the risk of accidental fire. It is used in a broad range of polymers, in products ranging from consumer electronics to wire and cable coatings and insulating foams.
For several decades, BFRs have been a controversial group of chemicals. Flame-retardants protect lives by increasing fire safety, but at the same time, several BFRs are considered as environmental contaminants such as polybrominated diphenyl ethers (PBDEs), tetrabromobisphennol A (TBBPA), which have been extensively used. Due to their persistence, bioaccumulation and biomagnification at food webs, long-range transportation and toxicity, such as endocrine disruption activity, the production and use in European Union was banned, and subsequently was phased out from USA and other countries. Moreover, several BFRs were classified as persistent organic pollutants and listed for global elimination compounds under the Stockholm Convention[1, 2].
Currently DBDPE is one commercially important alternative flame retardant which was predicted to be one of the most widely use flame retardants of the thermoplastics industry[3]. Due to large molecular size, low aqueous solubility and biological availability, DBDPE was believed to be rarely released into the environment and have low toxicity; DBDPE has a similar structure with PBDEs and has been produced and used for more than 20 years. First report on DBDPE was published in 2004[4]. Then it was found in sewage sludge, sediment, indoor air[4], birds[5] and in a benthic food web[6]. Research demonstrated that DBDPE is freely released to the surrounding environment, accumulated in animals and humans[7-10]. But only limited studies on the toxic effects of exposure of DBDPE have been conducted.
Although the in vivo or in vitro studies showed the toxicity of DBDPE was low, the potential endocrine disruptive action of BFRS, especially to PBDEs, has been of concerned recently. Many PBDEs are defined as endocrine-disrupting chemicals (EDCs) due to their structural similarity to endocrine hormones, such as thyroid hormones. A number of in vivo and in vitro studies on human, rodent and other models show that PBDEs have toxic effects on the hypothalamus-pituitary-thyroid axis, thyroid hormone metabolism and energy balance. These effects were typically observed in patients with obesity and metabolic syndromes. Short-term exposure to PBDEs also resulted in decline of thyroid hormones in rats[11, 12].
The liver has been considered the primary target organ of PBDEs[13, 14]. PBDEs could cause the induction in hepatic detoxification enzyme activities including Phase Ⅰ cytochrome P450 monooxygenase (CYP) enzymes and Phase Ⅱ conjugation enzymes [e.g., uridinediphosphate-glucuronosyltransferase (UDPGT), sulfotransferases] which lead to metabolism of these chemicals[15-17]. These results raised people's concern on the possibility of agonizing aryl hydrocarbon receptor (AhR)-mediated CYP1A induction; CYP1A1 induction was mediated primarily by AhR. The AhR was a transcription factor of cytosolic expression that is able to sense a wide range of both endogenous and exogenous ligands. The resulting structural composition of the AhR facilitates the translocation of ligand-AhR complexes into nuclei, where they were associated with the AhR nuclear translocater and bound to specific deoxyribonucleic acid (DNA) recognition sequences, notably dioxin-response elements or xenobiotic-response elements (XREs) in target genes. This increases the transcription of target genes such as gene encoding for CYP1A1. Subsequently, the affected xenobiotic substance is oxygenated to increase its solubility in water and to undergo further phase-Ⅱ conjugation by enzymes such as UDPGT and sulfotransferase. This process either makes the substrate more polar or eliminates it altogether. However, the oxygenation also initiates the production of active intermediates or procarcinogens that form DNA and protein adducts, leading to toxicity. Besides the AhR-mediated CYP1A induction, the pregnant X receptor (PXR) is also a promiscuous nuclear receptor for both xenobiotic chemicals and endogenous metabolites, and has been reported to coordinate hepatic responses with the constitutive androstane receptor (CAR). It also regulates the expression of CYP2 and CYP3 genes which are related with liver injury[18, 19].
Furthermore, while the mechanisms of PBDEs that induced the decline of circulating thyroid hormones concentrations are unclear, induction of metabolising enzymes via AhR, PXR, or CAR can interfere with the homeostasis of thyroid hormones. Their interaction initiates a cellular response, e.g. the transcription of specific genes, among them genes encoding proteins responsible for the metabolism of xenobiotics[20-23]. Research data demonstrated that environmental exposure to PBDEs was positively associated with human diabetes prevalence, which was strengthened by the animal experiment results showing that PBDEs could significantly increase high fasting glucose in rats. The result indicated that PBDEs might induce the disorder of glycometabolism and potentially result in the onset of diabetes[11, 24-26].
In the previous studies, we have found that DBDPE can cause a certain degree of rat liver damage; and the interference in the level of blood glucose in Wistar rats exposed to DBDPE was first observed in this work[20]. Research data on metabolic disturbance activity and endocrine disrupting activity of DBDPE in animals is still lacking. In the present study, Balb/C mice were administrated DBDPE in corn oil by oral gavage for 30 days. Body weight, liver weight and liver-to-body weight ratio were analyzed along with 8 clinical chemistry parameters, levels of insulin and thyroid hormone including thyronine (T3), thyroxine (T4), free triiodothyronine (fT3), and free thyroxine (fT4), Thyroid-stimulating hormone (TSH) were assayed, and blood glucose levels were measured every 5 days. Hepatic enzyme activities, including UDPGT and hepatic cytochrome P450 enzymes were also detected. Histopathological changes of liver and thyroid of mice were observed. The purpose of this study was to help us understand the relationship of hepatotoxicity, metabolic disturbance activity and endocrine disrupting activity of mice treated by DBDPE. This research would also help us understand whether DBDPE can act as an endocrine disrupter.
-
Male and female Balb/C mice, weighting approximately 22-27 g, were obtained from Experimental Animal Central of Academy of Military Medical Science (Beijing, China). They were allowed to acclimate for one week and observed daily for clinical signs of disease prior to the dosing. The laboratory conditions were maintained on a 12 h light/12 h dark cycle at 25 ℃ and (50 ± 10)% relative humidity. The animals were housed by species and sex, 4 mice/cage. The Balb/C mice were given free access to standard commercial rodent feed and drinking water. Animal care and maintenance was conducted in accordance with the applicable portions of the Animal Welfare Act, and all animal procedures were approved by Institutional Animal Care and Use Committee of Academy of Military Medical Science.
-
A powered mixture of DBDPE was obtained from Albemarle Corporation of the United States, which is characteristically 98.5% DBDPE, no more data on impurities were provided by the supplier. The Balb/C mice were separated randomly into treatment groups of 16 animals per dose groups, half male and half female, such that the mean body weights of all groups were statistically comparable at the study initiation. Control animals received corn oil only. DBDPE was dissolved in corn oil as exposure mixture. The doses for the treatment groups were 5, 20, 100, and 200 mg/kg body weight per day [mg/(kg·day)]. The Balb/C mice were treated orally by gavage administration daily for 30 consecutive days. Animals were weighed weekly, and observed daily for signs of toxicity.
Mouse blood glucose was measured every five days by a portable Roche glucose analyzer (Roche ACCU-CHEK® mobile blood glucose monitoring system, Roche Molecular Systems, Inc., Switzerland). Dose administration was based on the most recent individual body weights. After the end of exposure, animals were anesthetized with ether. Blood serum was drawn for biochemical parameter evaluation. Livers and thyroid gland were removed, washed in ice-cold physiological saline, and weighed. Portions of the livers and thyroid glands were collected and immediately frozen in liquid nitrogen, then stored at -80 ℃ until further processing.
-
Lactate dehydrogenase (LDH), γ-glutamyl transferase (GGT), alanine aminotransferase (ALT), aspartate aminotransferase (AST), total bilirubin (TBIL), indirect bilirubin (IBIL), alkaline phosphatase (ALP), and total bilirubin (TBA) of blood serums were measured on an autoanalyser (TBA-120, Toshiba, Japan) using standard kits from Roche Diagnostics.
-
Insulin, T3, T4, fT3, fT4, and TSH of blood serums were determine from a single measure on ELISA using standard kits from R & D systems (Minnesota, USA). All assays were conducted in duplicate.
-
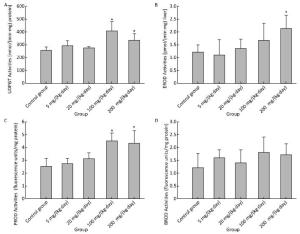
Microsomes were isolated according to the protocol of Melancon et al.[21], and dissolved in 50 mmol/L sodium phosphate buffer, pH 7.4. The microsomal protein concentration was assayed using bovine serum albumin (BSA) as the standard by bicinchoninic acid (BCA) protein assay kit (Biomed Co., Beijing, China). The UDPGT activity were determined by a spectrofluorometric method[22-24], which uses p-nitrophenol (Sigma, USA) as the aglycone and pure ammonium salt of uridine-5-diphosphoglucuronic acid (Sigma, USA) as the glucuronyl donor. Reduced enzyme activities in the liver were considered as an indication of hepatotoxicity. Ethoxyresorufin O-dealkylase (EROD) activity was used as a marker of CYP1A activity and was measured using a method used by Pohl and Fouts[25] as previously described[26]. Pentoxyresorufin O-dealkylase (PROD) activity was determined by a spectrofluorometric method as a marker for CYP2B[27]. Benzyloxyresorufin O-dealkylase (BROD) activity was analysed using the P450-GloTM CYP3A4 assay kit (Promega, Germany) as a marker for CYP3A.
-
Liver and thyroid gland from Balb/C mice in control and treatment groups were fixed in 10% phosphate-buffered formalin (pH 7.4). Tissues were embedded in paraffin and cut into 4 μm-thick slices. Liver and thyroid gland sections were stained with hematoxylin and eosin. The changes in the liver and thyroid gland were evaluated using an Olympus AX80 microscope (Olympus Corp., Tokyo, Japan) in a blinded fashion.
-
All values were expressed as mean ± standard deviation (n = 16). Absolute and relative organ weights were analysed using analysis of variance with the body weight at necropsy as a covariate. Body weight, liver weight, liver-to-body weight ratio, clinical chemistry parameters including blood glucose, AST, ALT, LDH, GGT, TBIL, IBIL, ALP, and TBA, insulin, TSH, T3, T4, free T3, free T4, UDPGT, and cytochrome P450 enzyme activities were compared using one-way analysis of variance (ANOVA) with comparisons among each group followed by Dunnett's multiple comparison tests. If significant heterogeneity of variance was apparent, some data transformations were performed prior to the statistical analysis. The levels of statistical significance were set as P = 0.05.
-
Based on our repeated-measures analysis, there were no obvious signs of toxicity and no significant treatment effect on body weight, or liver-to-body weight ratios between treatment groups. Liver weight significantly increased only at 200 mg/(kg·day) of DBDPE. (Table 1)
Table 1. Body Weight, Liver Weight, and Liver-to-body Weight Ratio
Treatment Group [mg/(kg·day)] Body Weight (g)a Liver Weight (g)a Liver-to-body Weight Ratioa 0 23.08 ± 2.51 1.08 ± 0.18 0.047 ± 0.009 5 23.81 ± 2.31 1.22 ± 0.18 0.052 ± 0.006 20 23.46 ± 2.13 1.22 ± 0.21 0.052 ± 0.003 100 23.90 ± 1.95 1.21 ± 0.16 0.051 ± 0.008 200 25.65 ± 2.36 1.38 ± 0.24b 0.054 ± 0.007 Note. aData are expressed as mean ± standard deviation (n = 16). bSignificantly different from control group (P < 0.05). -
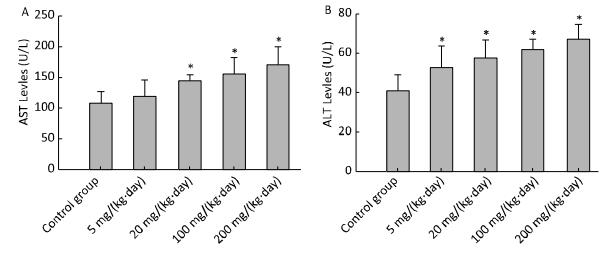
The levels of AST (Figure 1A) and ALT (Figure 1B) of different concentration treatment groups were markedly increased compared to the control group (P < 0.05). Other chemistry parameters including LDH, GGT, TBIL, IBIL, ALP, and TBA did not show significant differences compared to the control group.
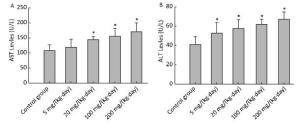
Figure 1. Effects of Balb/C mouse treated by DBDPE on selected chemistry parameters including AST (A) and ALT (B). Parameters was measured by an autoanalyser. Data represent means ± standard deviation. *Statistically significant differences between treatment group and control group were determined by a one-way ANOVA (n = 16, P < 0.05).
-
In comparision with control group, there was a significant increase in blood glucose levels in the treatment groups of 200 mg/(kg·day) starting from the 15th day, and 100 mg/(kg·day) from the 20th day, and 20 mg from the 30th day (Figure 2).
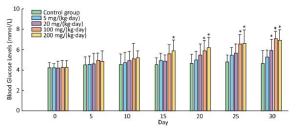
Figure 2. Effects of Balb/C mouse treated by DBDPE on blood glucose. Glucose was measured by a portable glucose analyzer. Data represent means ± standard deviation. *Statistically significant differences between treatment group and control group were determined by a one-way ANOVA (n = 16, P < 0.05).
-
There was a weak induction in TSH, only statistically significant in the treatment group of 200 mg/(kg·day) (Figure 3A). Serum total T3 was found to have decreased significantly for 200 mg/(kg·day) (Figure 3B), serum fT3 for 100 mg/(kg·day) and 200 mg/(kg·day) (Figure 3C). There was a declining but not significant change in serum total T4 (Figure 3D), fT4 (Figure 3E) and insulin (Figure 3F) in any treatment groups.
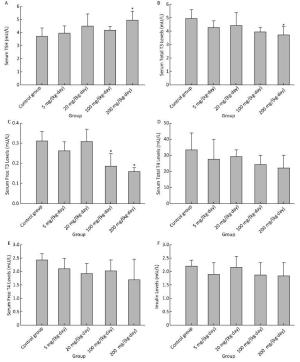
Figure 3. Effects of Balb/C mouse treated by DBDPE on TSH (A), T3 (B), fT3 (C), T4 (D), fT4 (E), and insulin (F). TSH, T3, fT3, T4, fT4, and insulin were measured by standard kits. Data represent means ± standard deviation. *Statistically significant differences between treatment group and control group were determined by a one-way ANOVA (n = 16, P < 0.05).
-
Effects on major xenobiotic metabolizing enzymes for phase Ⅰ metabolism were examined. UDPGT and PROD activities were found to have been increased significantly for 100 mg/(kg·day) and 200 mg/(kg·day) (Figure 4A and 4C), and EROD activity for 200 mg/(kg·day) (Figure 4B). Compared to the control group, no significant changes were observed in BROD activity in all treatment groups (Figure 4D).
-
Histopathologic liver changes were characterized by hepatocyte hypertrophy and cytoplasmic vacuolization in mice treated in high exposure dose (Figure 5B). No histopathological change of thyroid was observed in mice (Figure 5A).
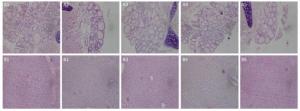
Figure 5. Thyroid gland from a mouse of the exposure group of 5 mg/(kg·day) (A2), 20 mg/(kg·day) (A3), 100 mg/(kg·day) (A4), and 200 mg/(kg·day) (A5) shown no significant change compared with the control group (A1). Liver from a mouse of the exposure group of 5 mg/(kg·day) (B2), 20 mg/(kg·day) (B3), and 100 mg/(kg·day) (B4) shown no significant change compared with the control group (B1). Liver from a mouse of the exposure group of 200 mg/(kg·day) (B5) show histopathologic liver changes characterized by hepatocyte hypertrophy and cytoplasmic vacuolization.
-
Many research studies suggest that the liver is the target organ for exposure to flame retardants[14, 28-30]. In our previous research, we have found no significant changes were observed in liver and relative liver weights of Wistar rats following exposure to DBDPE, indicating that DBDPE at the dose of 50-1, 000 mg/(kg·day) does not result in overt toxicity. But Glu, TBA, ALT, and AST levels following exposure to various doses of DBDPE were markedly higher in DBDPE treatment groups than in control group, indicating that DBDPE could cause liver function injury in Wistar rats[20]. In the present study, there was also no significant difference in body weight, liver weight, or relative liver weight of Balb/C mice in addition to liver weight for 200 mg/ (kg·day) treatment group, which indicated that these dose levels did not cause overt treatment-related toxicity effects after 30 days. In this research, significant alterations including AST and ALT levels were also observed in Balb/C mice by the biochemical parameters assay. Histopathologic liver changes were characterized by hepatocyte hypertrophy and cytoplasmic vacuolization in mice. All changes indicated that DBDPE could also cause liver damage in Balb/C mice, which may be due to possible oxidative stress induced by the accumulation of DBDPE or its metabolites even though there is limited information regarding the in vivo metabolism of DBDPE[10, 31, 32]. Our present research data suggested that the liver is also the target organ of DBDPE and it could cause liver insufficiency, which is consistent with the PBDEs.
Our previous research hinted that DBDPE had an elevation in blood glucose levels in Wistar rats. In this study, we also found that blood glucose concentrations in treated mice were dose-dependently higher than the control mice; measured every 5 days. Blood glucose data suggested that the glucose metabolic disorder might be caused by DBDPE dosing. We also found that there was a declining but not siginificant change of serum insulin in the treatment groups compared to control group. The mechanism of this disorder was not known at this time and needs further research. However, the potential of xenobiotics to disrupt glucose metabolism in mammals is a well-developed theory in toxicology. Indeed, many early toxicity responses in animal studies with a range of pollutants note glucosuria, increased gluconeogenesis[33, 34]. For example there was a significant decrease in insulin-stimulated glucose oxidation, compared with adipocytes from coin oil-treated controls, after gavaging Balb/C mouse with pentabrominated diphenyl ether (penta-BDEs) for 4 weeks[35].
Thyroid hormones are important for normal growth and development, playing an important role in the metabolic balance of animals, such as glucose metabolism and lipid metabolism. PBDEs may take a key role in destruction of thyroid homeostasis[36-38]. In a study involving exposure of rats to PBDEs and polychlorinated biphenyls, 14 consecutive days of treatment was sufficient to produce observable changes in thyroid histopathology[39]. In the present study, although DBDPE and PBDEs have similar structure, histopathological examination of thyroid sections revealed no abnormalities. However, our research results on thyroid hormones showed that serum TSH, total T3 and fT3 were affected, especially fT3, except serum fT4 and total T4, which were declining but no significant changes in any of the treatment groups, which represent a physiologic pattern in the direction of hypothyroidism. Furthermore, although we did not observe any inverse association with fT4, which would also be expected in the case of hypothyroidism, our suggestive finding of increased fT3 and total T3 may be physiologically consistent. In the hypothyroid state, the thyroid may attempt to compensate and convert more thyroid hormone to the most biologically active form, T3[40, 41]. Although we found that DBDPE was associated with TSH and T3, it is possible that these associations were not independent due to the thyroid's negative feedback loop, which results in functional relationships between hormones. Our finding of a tendency toward lower total T4 was explained through experimental research in animals treated by PBDEs. Research about rats and mice has revealed that PBDEs induce decreases in total T4; and one study additionally noted increases in TSH, which is similar with our research results[42, 43]. PBDEs, decreases in total T4 have been certified to be related with decreases in T4 binding to transthyretin, one of the main thyroid transport protein in rats[44, 45]. PBDEs also could combine with transport proteins competitively and replace T4[46]. Declining tendency in total T4 was also related with induction of hepatic microsomal enzymes such as CYPs and UDPGT, which increased metabolism and elimination of thyroid hormones. In addition, mechanism of DBDPE-induced thyroid disruption might be related with deiodinase activity[37]. Deiodinase are enzymes that activate or deactivate thyroid hormones by removing an iodine atom from different locations on thyroid hormones, which changes the bioavailability of target tissues. PBDEs, acting as a deiodinase inhibitor, could prevent conversion of T4 to T3[37]. Our present finding suggests that DBDPE, or its possible metabolites may disrupt thyroid function in mice in a manner similar to that of PBDEs. Furthermore, our studies have shown that there is a relationship between thyroid function and blood glucose. Thyroid hormone disorder may be associated with the change of blood glucose, which has been found in the present research of Balb/C mice and previous research of Wistar rats treated by DBDPE[20, 31]. Therefore, these findings support our contention for the endocrine-disrupting activity of DBDPE.
There are only limited studies on the effects of exposure to DBDPE on hepatic enzyme activities. Many researchers have found that effects of PBDEs in the liver were associated with induction of hepatic CYP450 enzymes and UDPGT, from mainly three assayed systems, CYP1A, CYP2B, and CYP3A[11, 12, 47-53]. CYP1A belongs to a hepatic phase Ⅰ metabolizing enzyme family and adds an endogenous or xenogenous hydroxyl group to the substrate, which leads to either detoxification or activation to more toxic metabolites. As the induction of CYP1A is a highly sensitive marker of AhR activation, measuring CYP1A offers a valuable means of quantifying AhR activation in the same way that CYP2B induction mediated CAR and CYP3A induction mediated by the PXR[52]. The UDPGT, belongs to a phase Ⅱ metabolizing enzyme family (isoenzymes) encoded by multigenes, that play a key role in the excretion of both endogenous and xenobiotic compounds, by adding a glucuronic acid to the substrate[54]. Additionally, phase Ⅰ CYPs and phase Ⅱ conjugates, such as UDPGT, are involved in the metabolism of both xenobiotics and key endogenous compounds, including various hormones such as steroids and thyroid hormones. In particular, induction of hepatic CYP450 enzymes and UDPGT via AhR, PXR, or CAR can interfere with the homeostasis of thyroid hormones[12, 55, 56]. The current study provides convincing evidence that there was a significant effect of DBDPE treatment of mice on hepatic CYP1A, CYP2B, and UDPGT enzyme activities compared to control group. No significant activity change of CYP3A was observed in any treatment groups. Then our present research results provide the evidence that DBDPE could induce the enzyme activities of CYP1A and CYP2B, which also may be an activator for xenobiotic nuclear receptors, namely AhR and CAR. AhR is a transcription factor, whose activity is modulated by hormones including glucocorticoids and estrogens[57]. The activation of the AhR receptor could be involved in the disruption of thyroid hormone homeostasis during liver insufficiency. Research data indicated that decreased thyroid hormone levels including total T4 and total T3 in parallel with increased TSH secretion suggested the stimulation of thyroid hormone glucuronidation as a result of AhR receptor activation and UDPGT up-regulation. The stimulation of glucuronidation may lead to reduced thyroid hormone levels and induced TSH secretion[58], which are fully consistent with our present research results. Generally, CAR has been implicated in regulation of circulating thyroid hormone concentrations. TBBPA (a BFR) treatment induced thyroid hormone conjugation pathways in a CAR-dependent manner resulting in diminished serum thyroid hormone levels, especially to T4[33]. However, we did not find a significant reduction in total T4 or free T4 in this study only a declining tendency. However, we did find that total T3 and fT3 decreased significantly. Apparently, interactions of DBDPE with the thyroid hormone system and mechanisms of feedback and adaptation by AhR and CAR signaling pathway can lead to the thyroid hormone's changes. Comparing DBDPE with other BFRs including DE-71 and TBBPA, the diversity of induced thyroid hormones may be due to the difference of chemical structure, expose time, animal species, and so on. In rodents, UDPGT is an important hepatic enzyme that metabolizes thyroid hormone, helping to characterize possible biochemical mechanisms for thyroid hormone[59]. In the present study, serum TSH, total T3 and fT3 were significantly affected. A declining trend, but non-significant reduction, in total T4 and fT4 were observed in the treatment groups, which showed significant induction of UDPGT activity. Whether this phenomenon hints at a relationship between serum thyroid hormone levels changes and induction of the UDPGT activity needs to be studied further. Hence, we believe that DBDPE could obstruct the thyroid function mediated by AhR and CAR.
In conclusion, DBDPE can cause a certain degree of mouse liver damage and insufficiency. We found that DBDPE demonstrated the activity of endocrine disruptors in Bal/C mice, which may induce drug-metabolizing enzymes including CYPs and UDPGT, and interfere with thyroid hormone levels mediated by AhR and CAR signaling pathways. Endocrine disrupting activity of DBDPE could also affect the glucose metabolism homeostasis.
-
SUN Ru Bao and SHANG Shuai carried out the majority of the experiments. ZHANG Wei and LIN Ben Cheng carried out the Clinical chemistry parameters assay experiment. WANG Qiang and SHI Yun participated in the experiments regarding Hepatic UDPGT and Cytochrome P450 enzyme activities assay. XI Zhu Ge designed this work and drafted the manuscript.
doi: 10.3967/bes2018.002
Endocrine Disruption Activity of 30-day Dietary Exposure to Decabromodiphenyl Ethane in Balb/C Mouse
-
Abstract:
Objective This study aimed to evaluate the hepatotoxicity, metabolic disturbance activity and endocrine disrupting activity of mice treated by Decabromodiphenyl ethane (DBDPE). Methods In this study, Balb/C mice were treated orally by gavage with various doses of DBDPE. After 30 days of treatment, mice were sacrificed; blood, livers and thyroid glands were obtained, and hepatic microsomes were isolated. Biochemical parameters including 8 clinical chemistry parameters, blood glucose and hormone levels including insulin and thyroid hormone were assayed. The effects of DBDPE on hepatic cytochrome P450 (CYP) levels and activities and uridinediphosphate-glucuronosyltransferase (UDPGT) activities were investigated. Liver and thyroid glands were observed. Results There were no obvious signs of toxicity and no significant treatment effect on body weight, or liver-to-body weight ratios between treatment groups. The levels of ALT and AST of higher dose treatment groups were markedly increased. Blood glucose levels of treatment groups were higher than those of control group. There was also an induction in TSH, T3, and fT3. UDPGT, PROD, and EROD activities were found to have been increased significantly in the high dose group. Histopathologic liver changes were characterized by hepatocyte hypertrophy and cytoplasmic vacuolization. Our findings suggest that DBDPE can cause a certain degree of mouse liver damage and insufficiency. Conclusion DBDPE has the activity of endocrine disruptors in Bal/C mice, which may induce drug-metabolizing enzymes including CYPs and UDPGT, and interfere with thyroid hormone levels mediated by AhR and CAR signaling pathways. Endocrine disrupting activity of DBDPE could also affect the glucose metabolism homeostasis. -
Figure 1. Effects of Balb/C mouse treated by DBDPE on selected chemistry parameters including AST (A) and ALT (B). Parameters was measured by an autoanalyser. Data represent means ± standard deviation. *Statistically significant differences between treatment group and control group were determined by a one-way ANOVA (n = 16, P < 0.05).
Figure 2. Effects of Balb/C mouse treated by DBDPE on blood glucose. Glucose was measured by a portable glucose analyzer. Data represent means ± standard deviation. *Statistically significant differences between treatment group and control group were determined by a one-way ANOVA (n = 16, P < 0.05).
Figure 3. Effects of Balb/C mouse treated by DBDPE on TSH (A), T3 (B), fT3 (C), T4 (D), fT4 (E), and insulin (F). TSH, T3, fT3, T4, fT4, and insulin were measured by standard kits. Data represent means ± standard deviation. *Statistically significant differences between treatment group and control group were determined by a one-way ANOVA (n = 16, P < 0.05).
Figure 5. Thyroid gland from a mouse of the exposure group of 5 mg/(kg·day) (A2), 20 mg/(kg·day) (A3), 100 mg/(kg·day) (A4), and 200 mg/(kg·day) (A5) shown no significant change compared with the control group (A1). Liver from a mouse of the exposure group of 5 mg/(kg·day) (B2), 20 mg/(kg·day) (B3), and 100 mg/(kg·day) (B4) shown no significant change compared with the control group (B1). Liver from a mouse of the exposure group of 200 mg/(kg·day) (B5) show histopathologic liver changes characterized by hepatocyte hypertrophy and cytoplasmic vacuolization.
Table 1. Body Weight, Liver Weight, and Liver-to-body Weight Ratio
Treatment Group [mg/(kg·day)] Body Weight (g)a Liver Weight (g)a Liver-to-body Weight Ratioa 0 23.08 ± 2.51 1.08 ± 0.18 0.047 ± 0.009 5 23.81 ± 2.31 1.22 ± 0.18 0.052 ± 0.006 20 23.46 ± 2.13 1.22 ± 0.21 0.052 ± 0.003 100 23.90 ± 1.95 1.21 ± 0.16 0.051 ± 0.008 200 25.65 ± 2.36 1.38 ± 0.24b 0.054 ± 0.007 Note. aData are expressed as mean ± standard deviation (n = 16). bSignificantly different from control group (P < 0.05). -
[1] Ricklund N, Kierkegaard A, McLachlan MS. An international survey of decabromodiphenyl ethane (deBDethane) and decabromodiphenyl ether (decaBDE) in sewage sludge samples. Chemosphere, 2008; 73, 1799-804. doi: 10.1016/j.chemosphere.2008.08.047 [2] Ricklund N, Kierkegaard A, McLachlan MS, et al. Mass balance of decabromodiphenyl ethane and decabromodiphenyl ether in a WWTP. Chemosphere, 2009; 74, 389-94. doi: 10.1016/j.chemosphere.2008.09.054 [3] Konstantinov A, Arsenault G, Chittim B, et al. Characterization of mass-labeled[13C14]-decabromodiphenylethane and its use as a surrogate standard in the analysis of sewage sludge samples. Chemosphere, 2006; 64, 245-9. doi: 10.1016/j.chemosphere.2005.12.009 [4] Kierkegaard A, Bjorklund J, Friden U. Identification of the flame retardant decabromodiphenyl ethane in the environment. Environ Sci Technol, 2004; 38, 3247-53. doi: 10.1021/es049867d [5] Gauthier LT, Potter D, Hebert CE, et al. Temporal trends and spatial distribution of non-polybrominated diphenyl ether flame retardants in the eggs of colonial populations of Great Lakes herring gulls. Environ Sci Technol, 2009; 43, 312-7. doi: 10.1021/es801687d [6] Law K, Halldorson T, Danell R, et al. Bioaccumulation and trophic transfer of some brominated flame retardants in a Lake Winnipeg (Canada) food web. Environ Toxicol Chem, 2006; 25, 2177-86. doi: 10.1897/05-500R.1 [7] Zhang J, Li Y, Wang Y, et al. Spatial distribution and ecological risk of polychlorinated biphenyls in sediments from Qinzhou Bay, Beibu Gulf of South China. Mar Pollut Bull, 2009; 80, 338-43. https://www.sciencedirect.com/science/article/pii/S0025326X13007613 [8] Iqbal M, Syed JH, Breivik K, et al. E-Waste Driven Pollution in Pakistan:The First Evidence of Environmental and Human Exposure to Flame Retardants (FRs) in Karachi City. Environ Sci Technol, 2017; 51, 13895-905. doi: 10.1021/acs.est.7b03159 [9] Cristale J, Aragao Bele TG, Lacorte S, et al. Occurrence and human exposure to brominated and organophosphorus flame retardants via indoor dust in a Brazilian city. Environ Pollut, 2017. https://www.sciencedirect.com/science/article/pii/S0269749117322376 [10] Wang G, Chen H, Du Z, et al. In vivo metabolism of organophosphate flame retardants and distribution of their main metabolites in adult zebrafish. Sci Total Environ, 2017; 590-591, 50-59. https://www.sciencedirect.com/science/article/pii/S0048969717305430 [11] Dunnick JK, Nyska A. Characterization of liver toxicity in F344/N rats and B6C3F1 mice after exposure to a flame retardant containing lower molecular weight polybrominated diphenyl ethers. Exp Toxicol Pathol, 2009; 61, 1-12. doi: 10.1016/j.etp.2008.06.008 [12] Tseng LH, Li MH, Tsai SS, et al. Developmental exposure to decabromodiphenyl ether (PBDE 209):effects on thyroid hormone and hepatic enzyme activity in male mouse offspring. Chemosphere, 2008; 70, 640-7. doi: 10.1016/j.chemosphere.2007.06.078 [13] Noyes PD, Hinton DE, Stapleton HM. Accumulation and Debromination of Decabromodiphenyl Ether (BDE-209) in Juvenile Fathead Minnows (Pimephales promelas) Induces Thyroid Disruption and Liver Alterations. Toxicol Sci, 2011; 122, 265-74. doi: 10.1093/toxsci/kfr105 [14] Curran IH, Liston V, Nunnikhoven A, et al. Toxicologic effects of 28-day dietary exposure to the flame retardant 1, 2-dibromo-4-(1, 2-dibromoethyl)-cyclohexane (TBECH) in F344 rats. Toxicology, 2017; 377, 1-13. doi: 10.1016/j.tox.2016.12.001 [15] Hakk H, Letcher RJ. Metabolism in the toxicokinetics and fate of brominated flame retardants——a review. Environ Int, 2003; 29, 801-28. doi: 10.1016/S0160-4120(03)00109-0 [16] Conley A, Mapes S, Corbin CJ, et al. Structural determinants of aromatase cytochrome p450 inhibition in substrate recognition site-1. Mol Endocrinol, 2002; 16, 1456-68. doi: 10.1210/mend.16.7.0876 [17] Nelson DR, Koymans L, Kamataki T, et al. P450 superfamily:update on new sequences, gene mapping, accession numbers and nomenclature. Pharmacogenetics, 1996; 6, 1-42. doi: 10.1097/00008571-199602000-00002 [18] Gramec Skledar D, Tomasic T, Carino A, et al. New brominated flame retardants and their metabolites as activators of the pregnane X receptor. Toxicol Lett, 2016; 259, 116-23. doi: 10.1016/j.toxlet.2016.08.005 [19] Richardson VM, Staskal DF, Ross DG, et al. Possible mechanisms of thyroid hormone disruption in mice by BDE 47, a major polybrominated diphenyl ether congener. Toxicol Appl Pharmacol, 2008; 226, 244-50. doi: 10.1016/j.taap.2007.09.015 [20] Sun RB, Xi ZG, Zhang HS, et al. Subacute effect of decabromodiphenyl ethane on hepatotoxicity and hepatic enzyme activity in rats. Biomed Environ Sci, 2014; 27, 122-5. https://www.researchgate.net/publication/260808723_Subacute_Effect_of_Decabromodiphenyl_Ethane_on_Hepatotoxicity_and_Hepatic_Enzyme_Activity_in_Rats [21] Melancon MJ, Williams DE, Buhler DR, et al. Metabolism of 2-methylnaphthalene by rat and rainbow trout hepatic microsomes and purified cytochromes P-450. Drug Metab Dispos, 1985; 13, 542-7. http://dmd.aspetjournals.org/content/13/5/542 [22] Castren M, Oikari A. Optimal assay conditions for liver UDP-glucuronosyltransferase from the rainbow trout, Salmo gairdneri. Comp Biochem Physiol C, 1983; 76, 365-9. doi: 10.1016/0742-8413(83)90091-9 [23] Winsnes A. Studies on the activation in vitro of glucuronyltransferase. Biochim Biophys Acta, 1969; 191, 279-91. doi: 10.1016/0005-2744(69)90247-2 [24] Nanbo T. Developmental changes in hepatic microsomal interactions between UDP-glucuronyltransferase and phospholipid in the rat fetus. Reprod Toxicol, 1998; 12, 449-56. doi: 10.1016/S0890-6238(98)00022-7 [25] Pohl RJ, Fouts JR. A rapid method for assaying the metabolism of 7-ethoxyresorufin by microsomal subcellular fractions. Anal Biochem, 1980; 107, 150-5. doi: 10.1016/0003-2697(80)90505-9 [26] Standeven AM, Goldsworthy TL. Identification of hepatic mitogenic and cytochrome P-450-inducing fractions of unleaded gasoline in B6C3F1 mice. J Toxicol Environ Health, 1994; 43, 213-24. doi: 10.1080/15287399409531916 [27] DeVito MJ, Ma X, Babish JG, et al. Dose-response relationships in mice following subchronic exposure to 2, 3, 7, 8-tetrachlorodibenzo-p-dioxin:CYP1A1, CYP1A2, estrogen receptor, and protein tyrosine phosphorylation. Toxicol Appl Pharmacol, 1994; 124, 82-90. doi: 10.1006/taap.1994.1011 [28] Giraudo M, Douville M, Letcher RJ, et al. Effects of food-borne exposure of juvenile rainbow trout (Oncorhynchus mykiss) to emerging brominated flame retardants 1, 2-bis(2, 4, 6-tribromophenoxy) ethane and 2-ethylhexyl-2, 3, 4, 5-tetrabromo-benzoate. Aquat Toxicol, 2017; 186, 40-9. doi: 10.1016/j.aquatox.2017.02.023 [29] Dunnick JK, Morgan DL, Elmore SA, et al. Tetrabromobisphenol A activates the hepatic interferon pathway in rats. Toxicol Lett, 2017; 266, 32-41. doi: 10.1016/j.toxlet.2016.11.019 [30] Yu L, Jia Y, Su G, et al. Parental transfer of tris(1, 3-dichloro-2-propyl) phosphate and transgenerational inhibition of growth of zebrafish exposed to environmentally relevant concentrations. Environ Pollut, 2017; 220, 196-203. doi: 10.1016/j.envpol.2016.09.039 [31] Sun RB, Xi ZG, Yan J, et al. Cytotoxicity and apoptosis induction in human HepG2 hepatoma cells by decabromodiphenyl ethane. Biomed Environ Sci, 2012; 25, 495-501. https://www.sciencedirect.com/science/article/pii/S0895398812600841 [32] Monte MJ, Marin JJ, Antelo A, et al. Bile acids:chemistry, physiology, and pathophysiology. World J Gastroenterol, 2009; 15, 804-16. doi: 10.3748/wjg.15.804 [33] Choi JS, Lee YJ, Kim TH, et al. Molecular Mechanism of Tetrabromobisphenol A (TBBPA)-induced Target Organ Toxicity in Sprague-Dawley Male Rats. Toxicol Res, 2011; 27, 61-70. doi: 10.5487/TR.2011.27.2.061 [34] Jones OA, Maguire ML, Griffin JL. Environmental pollution and diabetes:a neglected association. Lancet, 2008; 371, 287-8. doi: 10.1016/S0140-6736(08)60147-6 [35] Hoppe AA, Carey GB. Polybrominated diphenyl ethers as endocrine disruptors of adipocyte metabolism. Obesity (Silver Spring), 2007; 15, 2942-50. doi: 10.1038/oby.2007.351 [36] Eggesbo M, Thomsen C, Jorgensen JV, et al. Associations between brominated flame retardants in human milk and thyroid-stimulating hormone (TSH) in neonates. Environ Res, 2011; 111, 737-43. doi: 10.1016/j.envres.2011.05.004 [37] Szabo DT, Richardson VM, Ross DG, et al. Effects of perinatal PBDE exposure on hepatic phase Ⅰ, phase Ⅱ, phase Ⅲ, and deiodinase 1 gene expression involved in thyroid hormone metabolism in male rat pups. Toxicol Sci, 2009; 107, 27-39. doi: 10.1093/toxsci/kfn230 [38] Boas M, Main KM, Feldt-Rasmussen U. Environmental chemicals and thyroid function:an update. Curr Opin Endocrinol Diabetes Obes, 2009; 16, 385-91. doi: 10.1097/MED.0b013e3283305af7 [39] Bansal R, Zoeller RT. Polychlorinated biphenyls (Aroclor 1254) do not uniformly produce agonist actions on thyroid hormone responses in the developing rat brain. Endocrinology, 2008; 149, 4001-8. doi: 10.1210/en.2007-1774 [40] Nishio N, Katsura T, Ashida K, et al. Modulation of P-glycoprotein expression in hyperthyroid rat tissues. Drug Metab Dispos, 2005; 33, 1584-7. doi: 10.1124/dmd.105.004770 [41] Mitchell AM, Tom M, Mortimer RH. Thyroid hormone export from cells:contribution of P-glycoprotein. J Endocrinol, 2005; 185, 93-8. doi: 10.1677/joe.1.06096 [42] Hallgren S, Sinjari T, Hakansson H, et al. Effects of polybrominated diphenyl ethers (PBDEs) and polychlorinated biphenyls (PCBs) on thyroid hormone and vitamin A levels in rats and mice. Arch Toxicol, 2001; 75, 200-8. doi: 10.1007/s002040000208 [43] Hallgren S, Darnerud PO. Polybrominated diphenyl ethers (PBDEs), polychlorinated biphenyls (PCBs) and chlorinated paraffins (CPs) in rats-testing interactions and mechanisms for thyroid hormone effects. Toxicology, 2002; 177, 227-43. doi: 10.1016/S0300-483X(02)00222-6 [44] Auyeung DJ, Kessler FK, Ritter JK. Mechanism of rat UDP-glucuronosyltransferase 1A6 induction by oltipraz:evidence for a contribution of the Aryl hydrocarbon receptor pathway. Mol Pharmacol, 2003; 63, 119-27. doi: 10.1124/mol.63.1.119 [45] Nishimura N, Yonemoto J, Miyabara Y, et al. Altered thyroxin and retinoid metabolic response to 2, 3, 7, 8-tetrachlorodibenzo-p-dioxin in aryl hydrocarbon receptor-null mice. Arch Toxicol, 2005; 79, 260-7. doi: 10.1007/s00204-004-0626-4 [46] Peeters RP, Friesema EC, Docter R, et al. Effects of thyroid state on the expression of hepatic thyroid hormone transporters in rats. Am J Physiol Endocrinol Metab, 2002; 283, E1232-8. doi: 10.1152/ajpendo.00214.2002 [47] Boon JP, van Zanden JJ, Lewis WE, et al. The expression of CYP1A, vitellogenin and zona radiata proteins in Atlantic salmon (Salmo salar) after oral dosing with two commercial PBDE flame retardant mixtures:absence of short-term responses. Mar Environ Res, 2002; 54, 719-24. doi: 10.1016/S0141-1136(02)00127-7 [48] Fery Y, Buschauer I, Salzig C, et al. Technical pentabromodiphenyl ether and hexabromocyclododecane as activators of the pregnane-X-receptor (PXR). Toxicology, 2009; 264, 45-51. doi: 10.1016/j.tox.2009.07.009 [49] Fowles JR, Fairbrother A, Baecher-Steppan L, et al. Immunologic and endocrine effects of the flame-retardant pentabromodiphenyl ether (DE-71) in C57BL/6J mice. Toxicology, 1994; 86, 49-61. doi: 10.1016/0300-483X(94)90052-3 [50] Jonsson H, Schiedek D, Goksoyr A, et al. Expression of cytoskeletal proteins, cross-reacting with anti-CYP1A, in Mytilus sp. exposed to organic contaminants. Aquat Toxicol, 2006; 78 Suppl 1, S42-8. https://www.sciencedirect.com/science/article/pii/S0166445X0600066X [51] Olsvik PA, Lie KK, Sturve J, et al. Transcriptional effects of nonylphenol, bisphenol A and PBDE-47 in liver of juvenile Atlantic cod (Gadus morhua). Chemosphere, 2009; 75, 360-7. doi: 10.1016/j.chemosphere.2008.12.039 [52] Van der Ven LT, van de Kuil T, Leonards PE, et al. A 28-day oral dose toxicity study in Wistar rats enhanced to detect endocrine effects of decabromodiphenyl ether (decaBDE). Toxicol Lett, 2008; 179, 6-14. doi: 10.1016/j.toxlet.2008.03.003 [53] Zacharewski T, Harris M, Safe S, et al. Applications of the in vitro aryl hydrocarbon hydroxylase induction assay for determining " 2, 3, 7, 8-tetrachlorodibenzo-p-dioxin equivalents" :pyrolyzed brominated flame retardants. Toxicology, 1988; 51, 177-89. doi: 10.1016/0300-483X(88)90148-5 [54] Richardson TA, Klaassen CD. Role of UDP-glucuronosyltransferase (UGT) 2B2 in metabolism of triiodothyronine:effect of microsomal enzyme inducers in Sprague Dawley and UGT2B2-deficient Fischer 344 rats. Toxicol Sci, 2010; 116, 413-21. doi: 10.1093/toxsci/kfq125 [55] Sueyoshi T, Li L, Wang H, et al. Flame retardant BDE-47 effectively activates nuclear receptor CAR in human primary hepatocytes. Toxicol Sci, 2013; 137, 292-302. https://tools.niehs.nih.gov/.../portfolio/publicationDetail/id/2087679 [56] Xu T, Wang Q, Shi Q, et al. Bioconcentration, metabolism and alterations of thyroid hormones of Tris(1, 3-dichloro-2-propyl) phosphate (TDCPP) in Zebrafish. Environ Toxicol Pharmacol, 2015; 40, 581-6. doi: 10.1016/j.etap.2015.08.020 [57] Ma Z, Yu Y, Tang S, et al. Differential modulation of expression of nuclear receptor mediated genes by tris(2-butoxyethyl) phosphate (TBOEP) on early life stages of zebrafish (Danio rerio). Aquat Toxicol, 2015; 169, 196-203. doi: 10.1016/j.aquatox.2015.10.017 [58] Wu SY, Green WL, Huang WS, et al. Alternate pathways of thyroid hormone metabolism. Thyroid, 2005; 15, 943-58. doi: 10.1089/thy.2005.15.943 [59] Zhang L, Wang J, Zhu GN. Pubertal exposure to saisentong:effects on thyroid and hepatic enzyme activity in juvenile female rats. Exp Toxicol Pathol, 2010; 62, 127-32. doi: 10.1016/j.etp.2009.03.001 -




 下载:
下载:



 Quick Links
Quick Links