-
Perfluorooctane sulfonate (PFOS) is a class of stable organic compounds with wide industrial, commercial, and consumer applications, such as in textiles, paper, pesticides, and shampoos[1]. It is readily absorbed, but poorly eliminated, with the elimination half-life of approximately 5 years[2]. Hence, there have been concerns regarding its potential damage to human health. Some studies have indicated that the exposure to PFOS might have adverse effects on human gestation. In particular, PFOS can be freely transferred to the placenta, inducing developmental toxicity and causing fetal growth retardation[3].
Placenta is responsible for nutrient and oxygen exchange and blood flow, which is necessary for fetal growth. Placental dysfunction might cause several pregnancy-related complications. Angiogenesis is crucial for the normal development of placenta in humans. Adequate angiogenesis supports sufficient blood flow to the fetus, which is essential for fetal growth and development. Aberrant vascular formation is known to correlate with impaired fetal and placental development, resulting in lower birth weight.
So far, several studies have discussed the effects of long non-coding RNAs (lncRNAs) and microRNAs (miRNAs) on placental angiogenesis[4]. Several interactions exist between different noncoding RNAs, in which the activity of a specific lncRNA can alter the effect of a miRNA and vice versa. According to the competing endogenous RNA (ceRNA) hypothesis, the lncRNAs can compete with miRNA, resulting in miRNA-mediated gene silencing. The involvement of the ceRNA network has been confirmed in several diseases, but it has not been explored in a PFOS-exposed mouse model.
In this study, two adult CD-1 female mice were housed overnight with one male, starting at 18:00 h. The females were checked at 8:00 h the next morning, and the presence of a vaginal plug was defined as gestational day 0 (GD0). Adult pregnant dams were randomly divided into control and three treatment groups (n = 8 per group): 0.5, 2.5, and 12.5 mg PFOS/kg body weight (BW/day). The doses were selected based on a previous study on developmental toxicity of PFOS in mice[5]. Perfluorooctane sulfonate was orally administered daily by gavage from GD1 to GD17. The control group mice were administered an equivalent volume of corn oil (10 mL/kg). All the dams were euthanized on GD18 and caesarean section was performed. The placenta and fetus were removed from the uterus and weighed. The placental diameter and crown-rump length were measured.
The placenta tissue was fixed in 10% neutral-buffered formalin, embedded in paraffin, cut into sections of approximately 4 μm thickness, and stained with hematoxylin and eosin (H & E). For immunohistochemistry, 4-μm-thick horizontal sections of placenta were blocked with 3% Bovine Serum Albumin (BSA) for 30 min, and then incubated overnight with the primary antibodies for laminin (1:200; Biosynthesis, Beijing, China) and Ki67 (1:500; Servicebio, Wuhan, China). HRP-labeled secondary antibodies were used at a dilution of 1:200. The slides were washed in PBS and immunoreactivity was visualized with a DAB developer (Servicebio, G1211). After washing with tap water, the sections were counterstained with hematoxylin, dehydrated, and mounted with CITOTEST® new (Nanjing, China).
The expression level of angiogenesis-related mRNA, miRNA, and lncRNA in the placenta was determined by quantitative reverse transcription-polymerase chain reaction (qRT-PCR). U6 and Glyceraldehyde 3-phosphate dehydrogenase (Gapdh) were used as internal controls for miRNA and mRNA. The primer sequences are listed in Supplementary Table S1, available in www.besjournal.com. Western blotting was performed to evaluate the expression level of VEGF-A, MMP2, and MMP9. The protein in each sample was estimated using the BCA Protein Assay Kit (Beyotime Biotechnology, Shanghai, China). The membranes were incubated with primary antibodies (anti-VEGF-A, 1:1, 000; anti-MMP2, 1:1, 500; anti-MMP9, 1:1, 000; and anti-GAPDH, 1:1, 000) at 4 ℃ overnight. The membranes were then washed four times with TBS containing 0.1% Tween and incubated with goat anti-rabbit secondary antibodies (1:10, 000) for 90 min. The immunoreactivity bands were visualized using immobile Western Chemiluminescent HRP Substrate (Millipore, Billerica, MA, USA). The protein bands were scanned, and then the integrated light density values were calculated using Image Lab Software (Bio-Rad, California, USA) and normalized with those of GAPDH.
Gene Forward (5'-3') Reverse (5'-3') Hotair TTAACCATGAAACTACCAAAG GAAACCAAGAACGACCTAATA Xist TTTGTTCAGAGTAGCGAGGAC AAGGATTGCCTGGATTTAGAG Neat1 AGGTAAGGCTGTAGAGGACGG CAATGAAAGGTTTGAATGGCG miR-429 CGCGCGTAATACTGTCTGGTAA AGTGCAGGGTCCGAGGTATT Vegf-a ATGGATGTCTACCAGCGAAGC GGTTTGATCCGCATGATCTGCA Mmp9 GGACCCGAAGCGGACATTG CGTCGTCGAAATGGGCATCT Mmp2 ACCTGAACACTTTCTATGGCTG CTTCCGCATGGTCTCGATG U6 CTCGCTTCGGCAGCACA AACGCTTCACGAATTTGCGT Gapdh AGGTCGGTGTGAACGGATTTG GGGGTCGTTGATGGCAACA Table Supplementary Table S1. Primers for QPCR Amplification of MiRNA and MRNA
The data are expressed as mean ± standard error of the mean (SEM) and analyzed by Student's t-test, one-way analysis of variance (ANOVA), and Dunnett's method. The results with P-value < 0.05 were considered significant. The statistical analyses were performed using SPSS software for Windows (IBM SPSS Statistics 22.0, IBM Corporation, Somers, NY, USA).
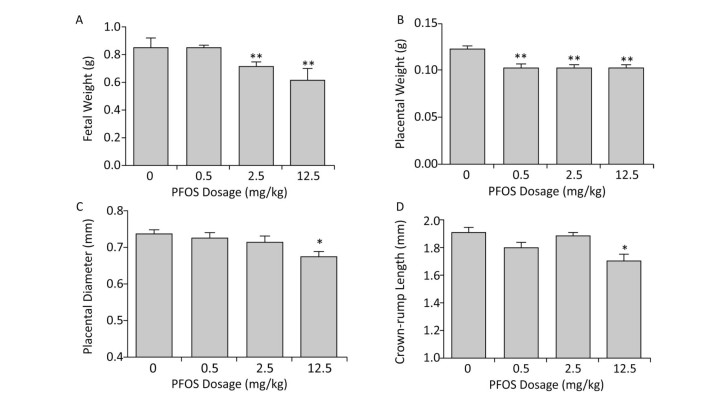
The results clearly demonstrated that prenatal exposure to PFOS resulted in developmental toxicity. Fetal weight was reduced in the 2.5 and 12.5 mg/kg PFOS-treated groups, which corresponded to the findings of an earlier study[6] (Figure 1A). In addition, the placental weight, diameter, and crown-rump length were significantly reduced after PFOS treatment (Figure 1B-D). Therefore, we speculate that PFOS exposure might cause low birth weight by affecting the development of placenta. The placenta is a highly complex tissue that is located between the fetal and prenatal compartments, allowing the exchange of oxygen and nutrients during gestation. Several studies have revealed that placental angiogenesis plays a crucial role in fetal growth[7]. Although placental angiogenesis has been extensively researched, its role in placental development has not been elucidated clearly.
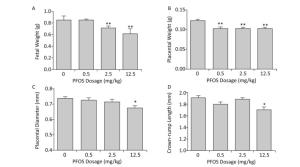
Figure 1. Effects of Perfluorooctane sulfonate (PFOS) on fetal and placental development. PFOS was orally administered daily by gavage from gestational day (GD)1 to GD17 at concentrations of 0.5, 2.5, and 12.5 mg/(kg·day); the controls received an equivalent volume of corn oil (10 mL/kg). All dams were euthanized and caesarean section was performed on GD18. The (A) fetal weight, (B) placental weight, (C) placental diameter, and (D) crown-rump length were measured. The values are expressed as mean ± SEM. (n = 88-97). *P < 0.05 compared with the control group and **P < 0.01 compared with the control group.
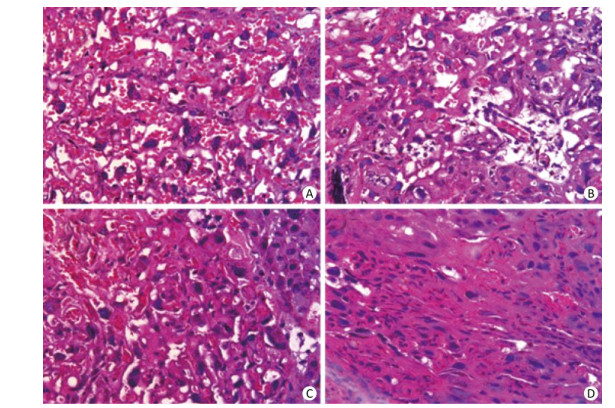
To further clarify the morphological characteristics of the placenta, both longitudinal and horizontal sections were prepared and stained with H & E. Histopathological examination showed that in the control group, the placental vessel and interstitial villi were normal, blood cells were filled, trophoblasts were orderly arranged, and blood vessel branches were clear (Supplementary Figure S1A, available in www.besjournal.com). However, branching of blood vessels was reduced significantly in the labyrinth layer of mice from the PFOS-treated group (Supplementary Figure S1B and S1C), especially from the 12.5 mg/kg PFOS-treated group (Supplementary Figure S1D). In this group, the blood vessels penetrated the labyrinth layer without branching and developing capillaries (exchange vessels). This abnormal vessel formation enabled PFOS to permeate the vessel and enter the blood circulation easily. In addition, the constriction of blood sinuses in the labyrinthine zone was observed in the horizontal sections of the placenta from the PFOS-treated dams (Supplementary Figure S1B-S1D).
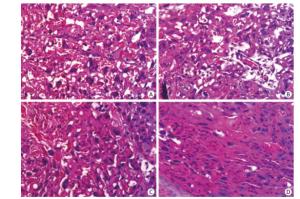
Figure Supplementary Figure S1. Exposure to PFOS results in histopathological alterations in the placental vascularization. Placenta samples (15 sections per group) were fixed with 4% paraformaldehyde; 10-μm-thick sections were cut using a cryostat, and counter-stained with hematoxylin and eosin. Bright-field microscopy was used to shoot the microphotographs. (A) Normal placental vessel. (B) Placental vascular changes in low dose (0.5 mg/kg) PFOS-treated group. (C) Placental vascular changes in medium dose (2.5 mg/kg) PFOS-treated group. (D) Abnormal placental vascularity in 12.5 mg/kg PFOS-treated group. (Original magnification 200×).
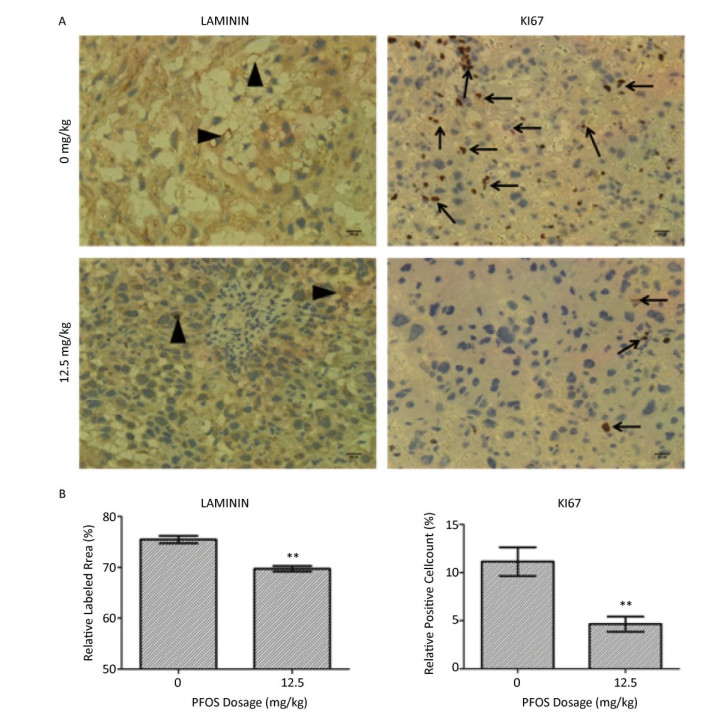
In this study, we investigated the development of labyrinth vascular by laminin staining. Our results suggested that PFOS exposure leads to placental vascular collapse, atresia, and basement membrane breakage, as well as significantly decreases laminin expression (Supplementary Figure S2A and S2B, available in www.besjournal.com). Moreover, the vascular region coinciding with the proliferating vascular cells was evaluated by Ki67 staining. As expected, we found that the relative number of Ki67 positive cells was reduced in the high dose PFOS-treated group (Supplementary Figure S2A and S2C). These results suggest that PFOS might affect placental angiogenesis by disrupting the integrity of basement membrane and by inhibiting the proliferation of vascular cells.
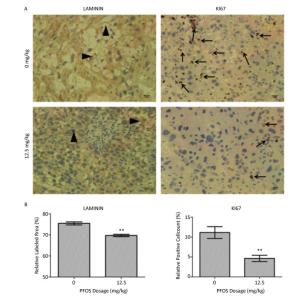
Figure Supplementary Figure S2. Immunohistochemistry analysis of the placenta. (A) Immunohistochemically staining for Laminin and Ki67 in the placenta (Laminin - positive cell; arrow head, Ki67 - positive cell; arrow). (B) Quantification of Laminin staining. The data represent the average of 10 photos (ten placentas from more than five dams in per group) of 0 mg/kg and 12.5 mg/kg PFOS-treated group. (C) Percentage of Ki 67 - positive cells. IHC, DAB chromogen, Hematoxylin counterstain, original magnification 400×. All data was expressed as means ± SEM. Statistical evaluation was performed with Student's t-test (**P < 0.01). Scale bars: 50 µm.
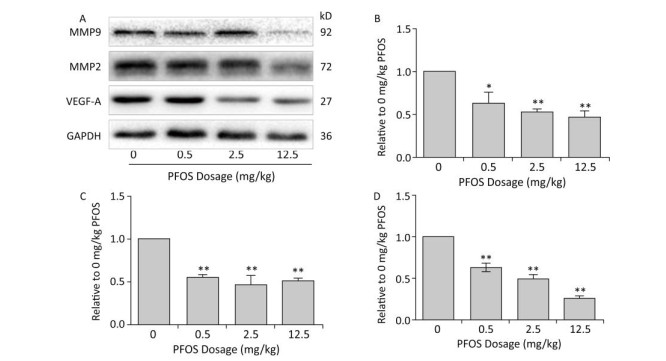
VEGF-A, one of the first angiogenic factors recognized, is widely believed as the most significant regulator of both normal and pathological angiogenesis[8]. During pregnancy, VEGF-A is involved in the migration, proliferation, and metabolic activity of trophoblasts. In the present study, the VEGF-A mRNA and protein levels in the placenta decreased after PFOS treatment (Figure 2A, 2B, and Supplementary Figure S3A, available in www.besjournal.com). In addition, several studies have clarified that MMP2 and MMP9 play crucial roles in placental angiogenesis. The down-regulation of MMP9 expression is associated with poor fetal growth and abnormal angiogenesis, which are most likely induced by poor transformation of maternal spiral arteries and limited trophoblastic invasion. On the contrary, the expression of MMP2 has been associated not only with angiogenesis, but also with vascular maturation and pericyte recruitment via multiple effectors. Our results demonstrated that the mRNA and protein expression levels of MMP2 and MMP9 in the placenta were reduced in the 12.5 mg/kg PFOS-treated group (Figure 2A, 2C, 2D and Supplementary Figure S3B, S3C). Overall, PFOS treatment decreased the VEGF-A, MMP2, and MMP9 mRNA and protein levels, suggesting that prenatal exposure to PFOS can induce aberrant angiogenesis of the placenta.
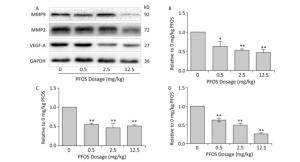
Figure 2. Perfluorooctane sulfonate (PFOS)-regulated proteins involved in the angiogenesis of placenta. All pregnant mice except controls were administered PFOS (0.5, 2.5, and 12.5 mg/kg) daily from gestational day (GD) 1 to GD17. The placenta from mice was collected on GD18. Three dams were chosen from each PFOS treatment group. One or two placenta sample(s) from each dam (n = 4) were randomly selected for western blotting. VEGF-A, MMP2, and MMP9 were subjected to western blotting. (A) A representative gel for VEGF-A, MMP2, and MMP9 is shown. Quantitative densitometry evaluation of (B) VEGF-A, (C) MMP9, and (D) MMP2 in the placenta. The data are presented as mean ± SEM of four samples from each group. *P < 0.05 and **P < 0.01.

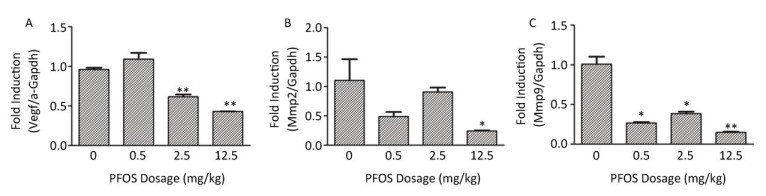
Figure Supplementary Figure S3. Effects of maternal exposure to PFOS on Vegf-a, Mmp2, and Mmp9 mRNA levels in the placenta on GD18. Three dams were chosen from each PFOS dose group, and one or two placentas (n = 4) were randomly selected for qRT-PCR analysis from each dam. (A) Vegf-a in placenta. (B) Mmp2 in placenta. (C) Mmp9 in placenta. The mRNA expression levels of the Vegf-a, Mmp2, and Mmp9 in the placentas were normalized to the ratio of the corresponding Gapdh mRNA expression levels. Data were expressed as mean ± SEM for the four samples from each group. (*P < 0.05, **P < 0.01).
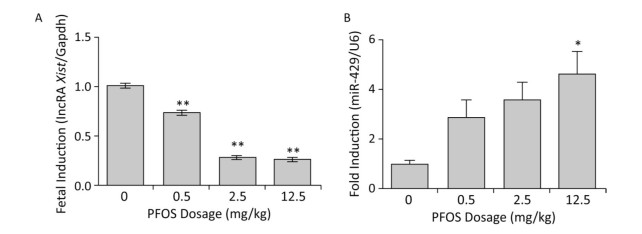
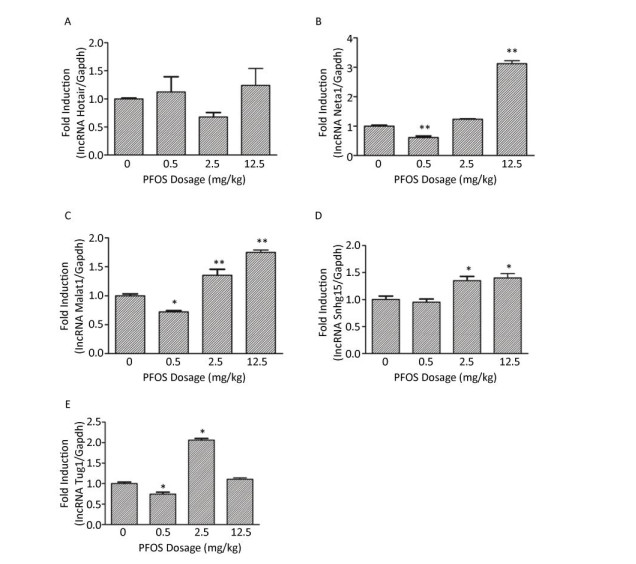
It is well known that the non-coding RNAs are pivotal regulators of both normal placental angiogenesis and pathological conditions. Unlike miRNA, the lncRNAs can fold into intricate secondary and higher order structures to enhance both target and protein recognition. The lncRNAs are emerging as new contributors to angiogenesis. Here, we chose several lncRNAs, which have been reported to be associated with angiogenesis, such as Hotair, Neat1, Xist, Malat1, Snhg15, and Tug1[9]. The present study results confirmed that the level of lncRNA Xist in the placenta decreased in a dose-dependent manner (Figure 3A), whereas the level of other lncRNAs was inconspicuous (Supplementary Figure S4, available in www.besjournal.com). Therefore, we presumed that lncRNA Xist might play a pathological role in placental angiogenesis.
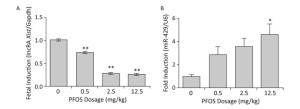
Figure 3. Effects of prenatal exposure to Perfluorooctane sulfonate (PFOS) on angiogenesis-related lncRNA Xist and miR-429 mRNA levels in the placenta on gestational day (GD) 18. Three dams were chosen from each PFOS dose group, and one or two placenta samples from each dam (n = 4) were randomly selected for the qRT-PCR analysis. (A) lncRNA Xist expression in the placenta. (B) miR-429 expression in the placenta. The expression level of lncRNA Xist and miR-429 in the placenta was normalized to that of Gapdh and U6 mRNA, respectively. The data are expressed as mean ± SEM of four samples from each group. *P < 0.05 and **P < 0.01.
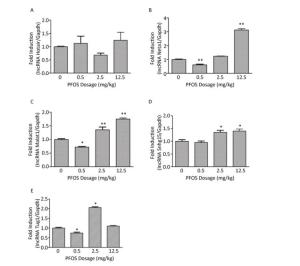
Figure Supplementary Figure S4. Effects of maternal exposure to PFOS on angiogenesis-related lncRNA mRNA levels in the placenta on GD18. (A) lncRNA Hotair in placenta. (B) lncRNA Neat1 in placenta. (C) lncRNA Malat1 in placenta. (D) lncRNA Snhg15 in placenta. (E) lncRNA Tug1 in placenta. Three dams were chosen form each PFOS dose group, and one or two placentas (n = 4) were randomly selected for qRT-PCR analysis from each dam. The mRNA expression levels of the angiogenesis-related lncRNA in the placentas were normalized to the ratio of the corresponding Gapdh mRNA expression levels. Data were expressed as mean ± SEM for the four samples from each group. (*P < 0.05, **P < 0.01).
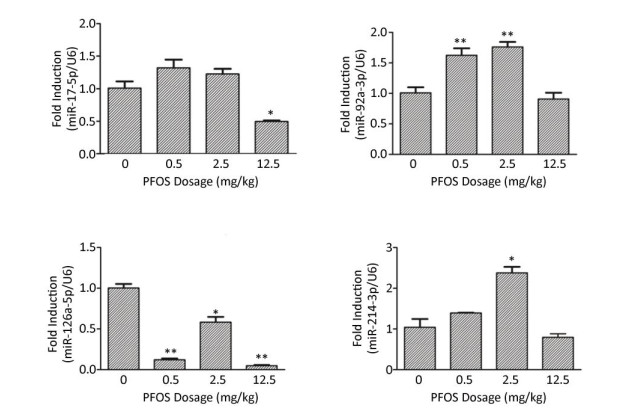
Recent studies have elucidated a complex interaction among multiple RNA species, including the coding and non-coding RNAs. These RNAs, including mRNA, miRNA, and lncRNA, co-regulate and interact with each other via the ceRNA mechanism. The mRNA and lncRNA function as ceRNAs to regulate and communicate with each other by competitively binding to the shared miRNA. During recent years, several studies have investigated the relationship between lncRNA and miRNA expression in placental angiogenesis. However, several lncRNAs or miRNAs playing crucial roles in placental angiogenesis remain to be determined. Besides, the lncRNA-miRNA-mRNA regulation networks involved in placental angiogenesis, as well as the roles of ceRNAs in the development of placental blood vessels have not been clarified. In this study, we detected the expression of lncRNA Xist-related miRNAs, such as miR-429, miR-17-5p, miR-92a-3p, miR-126a-5p, and miR-214-3p. The expression of miR-429 in the placenta was induced after PFOS treatment, showing a trend opposite to that of lncRNA Xist, whereas the other miRNAs were not induced (Figure 3B and Supplementary Figure S5, available in www.besjournal.com). Moreover, it has been reported that the overexpression of miR-429 contributed to lower birth weight[10]. LncRNA Xist is considered to directly regulate miR-429 expression by acting as a molecular sponge[11]. In addition, miR-429 decreases the 3'UTR luciferase activity of VEGF-A, and cancer cells transfected with miR-429 mimic manifested decreased expression of VEGF-A[12]. Therefore, we speculate that the lncRNA Xist-miR-429-VEGF-A regulation network plays a pivotal role in placental angiogenesis. Mechanistically, prenatal exposure of PFOS changed the expression of lncRNA Xist, which might act as a ceRNA by sponging miR-429 and suppressing VEGF-A, leading to abnormal placental angiogenesis.
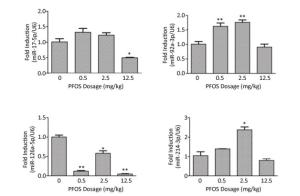
Figure Supplementary Figure S5. Effects of maternal exposure to PFOS on angiogenesis-related miRNA mRNA levels in the placenta on GD18. (A) miR-17-5p in placenta. (B) miR-92a-3p in placenta. (C) miR-126a-5p in placenta. (D) miR-214-3p in placenta. Three dams were chosen form each PFOS dose group, and one or two placentas (n = 4) were randomly selected for qRT-PCR analysis from each dam. The mRNA expression levels of the angiogenesis-related miRNA in the placentas were normalized to the ratio of the corresponding U6 mRNA expression levels. Data were expressed as mean ± SEM for the four samples from each group. (*P < 0.05, **P < 0.01)
In summary, prenatal exposure to PFOS impaired normal placental angiogenesis by disrupting the lncRNA Xist-miR-429-VEGF-A pathway, inducing adverse fetal development. Our study provides valuable information on the mechanism of developmental toxicity of PFOS.
There were no competing interests.
HTML
the Education Department of Jiangsu Province 16KJB330010
awards from National Natural Science Foundation of China 81703260
the Science and Technology Department of Jiangsu Province BK20160227
the China Postdoctoral Science Foundation funded project 2016M601892







 Quick Links
Quick Links
 DownLoad:
DownLoad: