-
Persistent high-risk human papillomavirus (hrHPV) infection is a necessary factor for precancerous cervical intraepithelial neoplasia and invasive cervical cancer. HPV infection and persistent infection are associated with poor body immunity. Human immunodeficiency virus (HIV)-positive women are susceptible to HPV infection due to the immunosuppression caused by HIV infection[1]. Hence, women infected with HIV face a heightened risk of developing precancerous lesions and cervical cancers.
It was estimated that China is home to 158,600 HIV-positive women over 15 years old[2]. Since free antiretroviral treatment (ART) was made available in the country in 2002, the life expectancy of cases living with HIV has been prolonged. Cervical cancer is one of the most common tumors found among HIV-positive women in China. A planned, systematic, quality-controlled cervical cancer screening program can significantly reduce the incidence and mortality of cervical cancer in HIV-positive and -negative women[1]. Unfortunately, the national cervical cancer screening project targets only rural women aged 35–64 years, and its coverage is limited[3]. Thus, the need for cervical cancer screening and prevention services for HIV-positive women in China has not been satisfied.
No specific recommendations on cervical cancer screening and prevention program for HIV-positive women is currently available in China due to the shortage of adequate information. The issue of the prevalence of and risk factors for hrHPV among this population could have potential implications for informing future cervical cancer prevention and control programs for Chinese HIV-positive women. Thus, we investigated the prevalence of and associated risk factors for hrHPV infection among HIV-positive women in high-burden HIV areas in China.
The study population was HIV-positive women from five selected counties in Yunnan Province, Guangxi Zhuang Autonomous Region, and Xinjiang Uygur Autonomous Region. We calculated the sample size by using the formula for a cross-sectional observational study. Given an allowable deviation of 15%, design effect of 2, and hrHPV infection rate of 34%[4,5], the approximate sample size was determined to be 677 cases. Considering a refusal rate of 10%, we planned to recruit about 150 women in every county. Random cluster sampling was performed, and 28 townships were randomly selected from five counties (71 townships in total). Finally, all HIV-positive women from the chosen townships who met the inclusion criteria were enrolled in this study. The inclusion criteria were the following: 1) confirmed HIV status by a laboratory test and 2) aged 18–49 years. HIV-positive women were excluded when they: 1) were ill at the time the study was conducted, 2) could not be contacted following meeting the recruitment requirements of the study protocol, 3) had no sexual intercourse experience, and 4) had a history of uterectomy.
The study was conducted between May and August 2015. A face-to-face structured questionnaire survey was performed to collect sociodemographic characteristics and information on sexual practices. The medical records of the HIV-positive women were reviewed to extract data on ART and laboratory results. Trained clinicians collected serum samples, and two exfoliative cervical cell specimens from each woman were obtained through gynecological examinations.
The first cervical cell specimen was sent to Beijing Di’an Laboratory for cytological examination and HPV DNA testing via the Cobas 4800 HPV test (Roche Molecular Diagnostics [Shanghai] Co. Ltd., Shanghai, China) to obtain results on HPV16 and HPV18 and pooled results on 12 other hrHPV types (31, 33, 35, 39, 45, 51, 52, 56, 58, 59, 66, and 68b). The second cervical cell specimen was sent to the designated laboratories for hrHPV genotyping. Samples from Yunnan were tested by high-throughput HPV genotyping assay based on next-generation sequencing and multiplex polymerase chain reaction in Clinical Laboratory Center of the Beijing Genomics Institute, Shenzhen. Samples from Xinjiang were tested by using a DNA-chip technology kit (Yaneng Bioscience [Shenzhen] Co. Ltd, Shenzhen, China) in Yili Prefectural MCH Hospital, Xinjiang. Samples from Guangxi were tested by using the 21 HPV GenoArray Diagnostic Kit (HBGA-21PKG; Hybribio Co. Ltd., Hongkong, China) in Hezhou Prefectural MCH Hospital.
HIV-positive women with cytological abnormality or hrHPV infection received colposcopy. The women with abnormal findings of colposcopy underwent biopsies following the national technical protocol. HIV-positive women with high-grade squamous intraepithelial lesions (HSIL) were referred to appropriate treatment.
The study team separated serum samples at the county-level MCH hospital laboratories and tested them for syphilis using the treponema pallidum particle agglutination (Zhuhai Livzon Diagnostics Inc., Zhuhai, China) and toluidine red unheated serum tests (Beijing Kinghawk Pharmaceutical Co., Ltd., Beijing, China) and for hepatitis B surface antigen using enzyme-linked immunosorbent assay (Shanghai Kehua Bio-engineering Co., Ltd., Shanghai, China). The results of CD4 lymphocyte count and HIV viral load tests within 6 months of the fieldwork were obtained from clinical records. The serum samples of participants who had not undergone these tests were sent to the local prefecture CDC laboratories for appropriate testing. The study team carried out strict quality control in all aspects of laboratory specimen collection, storage, transportation, and testing.
We defined hrHPV infection as a positive result in the Cobas 4,800 HPV test for HPV16, HPV18, or 12 other hrHPV types. HPV16 and HPV18 infection were defined as a positive result in the Cobas 4,800 HPV test for HPV16 and HPV18, respectively. Infection with any of the 12 other hrHPV types was defined as a positive result in the HPV genotyping test. We defined HIV viral load as undetectable (< 50 copies/mL) or detectable (≥ 50 copies/mL) and CD4 lymphocyte count as high (≥ 500 cells/mm3) or low (< 500 cells/mm3). We defined sexual debut as early (before 18 years old) or late (after 18 years old). We defined the degree of cervical lesions as normal, low-grade squamous intraepithelial lesion (LSIL), or HSIL. The HSIL group included women with cervical intraepithelial neoplasia grades two and worse (CIN2+), while the LSIL group included women with CIN1.
We double-entered data and used logical checks during data entry. We then analyzed the data using SPSS software (version 23.0, IBM Corp, Armonk, NY, USA). We presented categorized variables as frequencies and proportions. We used Pearson’s χ2 test to assess the association of categorical variables with hrHPV infection categories. We entered factors with a P-value of < 0.1 into a binary logistic regression to build the final model. Moreover, we defined a P-value of < 0.05 as statistically significant and calculated odds ratios (OR) and 95% confidence intervals (95% CI).
This study was reviewed and approved by the Institutional Review Board of the National Center for Women and Children’s Health, China CDC (No. FY2015–014). All subjects provided informed consent prior to participating in this study.
Among 947 HIV-positive women identified in the CDC database from the selected townships, 250 (26.5%) could not be contacted or refused to participate in this study and 2 (0.2%) were excluded because of a history of uterectomy. Finally, the data of 695 women were analyzed. The sociodemographic characteristics, biological indicators of HIV infection, and STI coinfection of these 695 HIV-positive women are presented in Table 1.
Characteristics N Percentage (%) Total 695 100.0 Age (years) 18–24 22 3.2 25–29 98 14.1 30–34 192 27.6 35–39 207 29.8 40–49 176 25.3 Province Guangxi 168 24.1 Yunan 266 38.3 Xinjiang 261 37.6 Ethnic group Han and others 247 35.5 Uighur 237 34.1 Dai/Jingpo 211 30.4 Education level Junior high and below 597 85.9 Senior high and above 98 14.1 Current employed Yes 457 65.8 No 238 34.2 Marital status Married/cohabited 511 73.5 Single/divorced/widow 184 26.5 Residence registration Urban 188 27.1 Rural 507 72.9 Annual income (RMB per capita) ≥ 5,000 185 26.6 < 5,000 510 73.4 CD4 lymphocyte count (/mm3) High (≥ 500) 354 50.9 Low (< 500) 341 49.1 HIV viral load (copies/mL) Undetectable (< 50) 552 79.4 Detectable (≥ 50) 143 20.6 Duration of ART (years) ≥ 2 591 85.0 < 2 79 11.4 No ART 25 3.6 Syphilis Yes 43 6.2 No 652 93.8 HBsAg positive Yes 44 6.3 No 651 93.7 Note. hrHPV, high-risk human papillomavirus; HIV, human immunodeficiency virus; ART, antiretroviral therapy; HBsAg, hepatitis B surface antigen. Table 1. Socio-demographic characteristics, biological indicators of HIV infection, and STIs coinfection among HIV-positive women in high HIV-burdens of China, 2015
We found an hrHPV prevalence of 25.9% (95% CI 22.6–29.2), with 180 women testing positive for at least one of 14 hrHPV types. Out of these 180 hrHPV-infected women, 125 (69.4%) were infected with a single hrHPV type, while 55 (30.6%) were infected with multiple hrHPV types. The three most commonly detected hrHPV types in the cervical cytological specimens of the 180 hrHPV-positive women were HPV16 (47, 26.1%), HPV58 (38, 21.1%), HPV52 (35, 19.4%). The hrHPV prevalence among HIV-positive women in HIV high-burden areas of China in our study is similar to a meta-analysis that revealed an hrHPV prevalence of 31% among HIV-positive women in Asia[6] but lower than that reported in previous hospital-based studies conducted in Yunnan and Hubei Provinces of China[4,5]. Such variation may be due to the characteristics of the study populations. The hrHPV prevalence among HIV-positive women in our study is also relatively higher than that of general women in China, which is consistent with the results of previous studies on hrHPV infection among HIV-positive women[5]. Thus, HIV-positive women are more vulnerable to hrHPV infection than HIV-negative women.
In our study, among 617 HIV-positive women who underwent the full cervical cancer screening procedure, the majority (574, 93.0%) were normal, 2.9% (18) were with LSIL, and 4.1% (25) were with HSIL. The detection rate of precancerous lesions (HSIL) among HIV-positive women in our study is higher than that of general women aged 35–64 years in rural China in 2012 (1.1‰) and 2013 (1.2‰)[3]. Our findings thus confirm that the issue of cervical cancer in HIV-positive women in China warrants urgent attention and that cervical cancer prevention and screening services must be provided to HIV-positive women.
In our study, the proportion of women who were screened to be positive for hrHPV rose with increasing severity of histological abnormalities from 17.6% (101/574) in normal women to 88.9% (16/18) in women with LSIL to 92.0% (23/25) in those with HSIL (χ2 = 111.70, P < 0.001). Among these 140 hrHPV-positive women, the three most commonly detected hrHPV types were HPV16 (23.6%), HPV58 (22.1%), and HPV52 (20.0%) (Figure 1). HPV16 (64.0%), HPV52 (20.0%), and HPV58 (16.0%) were the most common hrHPV types among the 25 women with HSIL. The total proportion of HPV16 and HPV18 infections accounted for 72% of all women with HSIL. In line with another study[7], our findings demonstrate the association between hrHPV infection and HSIL among HIV-positive women. HPV-based cervical screening has high sensitivity for a high degree of cervical lesions. Hence, we suggest developing an HPV-based cervical cancer screening project for HIV-positive women in China. If conditions permit, type-specific HPV testing is recommended. HIV-positive women infected with hrHPV should be provided with frequent follow-up and screening. The dominance of HPV16 and HPV18 positivity among HIV-positive women with a high degree of histological abnormality implies that HIV-positive women would benefit from currently available 2-valent and 4-valent HPV vaccines in China.
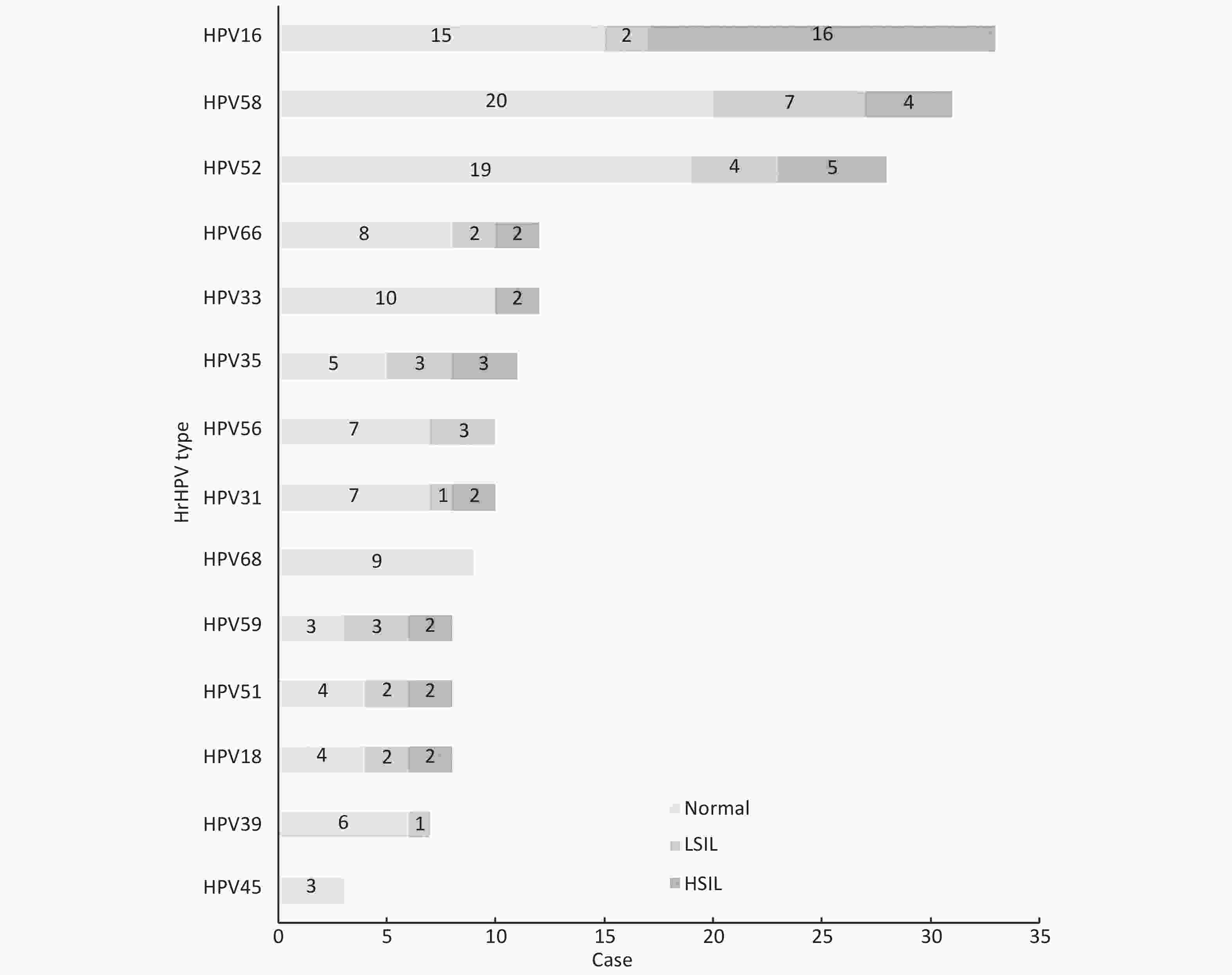
Figure 1. HrHPV type distribution in cervical histological status among HIV-positive women in high HIV-burden areas of China, 2015. Data was not available for 78 women who refused to undergo a colposcopy. LSIL, low-grade squamous intraepithelial lesion; HSIL, high-grade squamous intraepithelial lesion
Our findings reveal that HPV16 is the dominant hrHPV type in HIV-positive women, especially those with HSIL, which agrees with a meta-analysis on the hrHPV distribution among HIV-positive women[7]and previous studies in China[5]. However, previous scholars reported that HPV52 is the most common hrHPV type in general women and that HPV16 is the most common hrHPV type in general women with cervical lesions in China[8]. This difference in dominant hrHPV type may be due to the rapid progression of some HPV types, such as HPV16 and HPV18, compared with others[8]. Our results imply that, compared with general women, HIV-positive women in China are more likely to be persistently infected with HPV16. This finding must be investigated further through a comparative cohort study.
The results of univariate analysis and multivariate logistic regression of factors associated with hrHPV infection among HIV-positive women are shown in Table 2. We found that the adjusted odds of hrHPV infection for women with a low CD4 lymphocyte count is 1.61 times that of women with a high CD4 lymphocyte count [adjusted OR (aOR) 1.61, 95% CI 1.11–2.32, P = 0.01]. HPV infection induces an immune response in the body that can clear HPV-infected cells. The cellular immune response plays an important role in clearing HPV-infected and tumor cells. CD4 lymphocytes are mainly involved in the cellular immune response. After HIV infection, CD4 lymphocytes are consistently reduced, and the risk of initial and persistent infection of HPV increases. In general, the CD4 lymphocyte count level of HIV-positive individuals on ART treatment is higher than that of HIV-positive individuals without ART. A meta-analysis showed that HIV-positive women on ART have low hrHPV prevalence[9]. However, our findings did not find such correlation between ART and hrHPV infection, likely because the ART coverage in our sample was high and most of the participants had received ART for over 2 years. Hence, our results imply that CD4 lymphocyte count level is a possible marker for hrHPV infection risk. This finding is crucial for cervical cancer screening and control projects for HIV-positive women.
Characteristics No. of observations N hrHPV positive
n (%)Univariate analysis Multivariable analysis OR (95% CI) P-value aOR (95% CI) P-value Age (years) 18–24 22 7 (31.8) 1 0.24 25–29 98 24 (24.5) 0.70 (0.25–1.91) 30–34 192 59 (30.7) 0.95 (0.37–2.45) 35–39 207 54 (26.1) 0.76 (0.29–1.95) 40–49 176 36 (20.5) 0.55 (0.21–1.45) Province Guangxi 168 53 (31.5) 2.39 (1.51–3.79) < 0.001 2.17 (1.03–4.56) 0.04 Yunan 266 43 (16.2) 1 1 Xinjiang 261 84 (32.2) 2.46 (1.62–3.74) 1.67 (0.55–5.08) 0.37 Ethnic group Han and others 247 72 (29.1) 1 < 0.001 1 Uighur 237 77 (32.5) 1.17 (0.80–1.72) 0.99 (0.37–2.64) 0.98 Dai/Jingpo 211 31 (14.7) 0.42 (0.26–0.67) 0.76 (0.36–1.61) 0.47 Education level Junior high and below 597 148 (24.8) 0.68 (0.43–1.08) 0.10 Senior high and above 98 32 (32.7) 1 Current employed Yes 457 108 (23.6) 0.71 (0.50–1.01) 0.06 1.25 (0.81–1.94) 0.31 No 238 72 (30.3) 1 1 Marital status Married/cohabited 511 123 (24.1) 0.71 (0.49–1.03) 0.07 0.94 (0.61–1.47) 0.80 Single/divorced/widow 184 57 (31.0) 1 1 Residence registration Urban 188 63 (33.5) 1.68 (1.16–2.42) 0.01 1.51 (0.99–2.30) 0.06 Rural 507 117 (23.1) 1 1 Annual income (RMB per capita) ≥ 5,000 185 47 (25.4) 1 0.86 < 5,000 510 133 (26.1) 1.04 (0.70–1.52) Age at sexual debut* (years) Early (< 18) 95 33 (34.7) 1.64 (1.03–2.60) 0.03 1.68 (1.03–2.73) 0.04 Late (≥ 18) 592 145 (24.5) 1 1 Currently have a sexual partner Yes 590 147 (24.9) 0.72 (0.46–1.14) 0.16 No 105 33 (31.4) 1 Lifetime sexual partners 1–2 sexual partners 570 141 (24.7) 0.73 (0.48–1.11) 0.14 ≥ 3 sexual partners 125 39 (31.2) 1 Used condom in the last year Yes 407 94 (23.1) 1 0.05 1 No 288 86 (29.9) 1.42 (1.01–2.00) 1.36 (0.89–2.06) 0.15 Currently smoking Yes 25 9 (36.0) 1.64 (0.71–3.78) 0.24 No 670 171 (25.5) 1 Number of children delivered 0–2 581 145 (25.0) 1 0.20 3–6 114 35 (30.7) 1.33 (0.86–2.07) CD4 lymphocyte count (/mm3) High (≥ 500) 354 76 (21.5) 1 1 Low (< 500) 341 104 (30.5) 1.61 (1.14–2.26) 0.01 1.61 (1.11–2.32) 0.01 HIV viral load (copies/mL) Undetectable (< 50) 552 125 (22.6) 0.47 (0.32–0.69) < 0.001 0.70 (0.44–1.13) 0.15 Detectable (≥ 50) 143 55 (38.5) 1 1 Duration of ART (years) ≥ 2 591 144 (24.4) 1 0.07 1 < 2 79 26 (32.9) 1.52 (0.92–2.52) 1.28 (0.75–2.20) 0.37 No ART 25 10 (40.0) 2.07 (0.91–4.71) 1.37 (0.55–3.40) 0.50 Syphilis Yes 43 17 (39.5) 1.96 (1.04–3.71) 0.04 1.39 (0.70–2.74) 0.35 No 652 163 (25.0) 1 1 HBsAg positive Yes 44 13 (29.5) 1.22 (0.62–2.38) 0.58 No 651 167 (25.7) 1 Note. hrHPV, high-risk human papillomavirus; HIV, human immunodeficiency virus; ART, antiretroviral therapy; HBsAg, hepatitis B surface antigen; OR, odds ratio; aOR, adjusted odds ratio. *: 687 women reported age at sexual debut; factors significant at P < 0.1 in univariate analysis entered into the regression model. Table 2. Univariate and multivariate of factors associated with hrHPV infection among HIV-positive women in high HIV-burden areas of China, 2015
HPV infection, similar to all sexually transmitted diseases, is associated with a number of sexual behaviors, such as multiple sexual partners, early sexual debut age, and marriage[10]. We found that the hrHPV infection rate of women with early sexual debut is greater than that of women with late sexual debut (aOR 1.68, 95% CI 1.03–2.73, P = 0.04). Thus, early sexual debut may be associated with a higher frequency of sexual intercourse and number of sexual partners. We also found differences in the hrHPV positivity of HIV-positive women from different provinces, which may be attributed to differences in social habits and lifestyles in these areas.
The study has several limitations. First, our results are generated from Southern and Western China and may not be generalizable to the national level. Second, our findings may reflect selection bias for areas where free ART is widely available. Third, we applied different HPV genotyping tests due to a lack of research resources. The Cobas 4,800 HPV DNA test was applied to diagnose hrHPV infection in this study and minimize potential errors occurring in different tests.
Despite these limitations, the findings of our research yield important information on the prevalence and type distribution of hrHPV among HIV-positive women in HIV high-burden areas of China. Interestingly, we found an association between CD4 lymphocyte count level and hrHPV infection in this population. These findings show the necessity of a cervical cancer screening program for HIV-positive women in China and indicate the importance of maintaining high CD4 lymphocyte count levels to prevent hrHPV infection. Further cohort studies are needed to understand the characteristics of initial and persistent infection of hrHPV among HIV-positive women and its associated factors.
No conflict of interest to declare.
We would like to express our thanks to XU Wen Qing from the UNICEF China, Dr. SU Sui Qing from the National Center for Women and Children’s Health, Chinese Center for Disease Control and Prevention, Dr. LIANG Wen from the Guangxi Maternal and Child Health Hospital, the staff at the study sites who contributed to the fieldwork, and all participants in this study.
QYP, WLH, and WAL conceived and designed the study. QYP, WAL, and FLW conducted data collection and analysis. QYP, WAL, WLH, FLW, and KH interpreted the results and wrote the first and subsequent drafts of the paper. All authors read and approved the final manuscript.
High-risk Human Papillomavirus Infection and Associated Factors among HIV-positive Women in High HIV-burden Areas of China
doi: 10.3967/bes2020.029
- Received Date: 2019-07-29
- Accepted Date: 2020-02-05
| Citation: | QIAO Ya Ping, WANG Ai Ling, FANG Li Wen, Katrina HANN, WANG Lin Hong. High-risk Human Papillomavirus Infection and Associated Factors among HIV-positive Women in High HIV-burden Areas of China[J]. Biomedical and Environmental Sciences, 2020, 33(3): 206-212. doi: 10.3967/bes2020.029 |







 Quick Links
Quick Links
 DownLoad:
DownLoad: