-
Gastrointestinal stromal tumors (GISTs) are derived from the interstitial cells of Cajal, and are the most common mesenchymal tumors of the gastrointestinal tract[1]. KIT proto-oncogene, receptor tyrosine kinase (KIT), and platelet-derived growth factor receptor alpha (PDGFRA) gene mutations are found in approximately 85%–90% cases of GIST, and are thought to play a role in its development[2-3]. The most common KIT mutation is localized in exon 11 (approximately 70% of cases), which inhibits the autoinhibitory function of the KIT juxtamembrane domain, resulting in constitutive activity. Further, exon 11 mutation is reportedly involved in the development of liver metastasis and might be associated with a worse clinical outcome. The other KIT mutations are localized in several gene regions including exons 9, 13, and 17, which are found in 5%–10%, 1%–3%, and < 1%–3% cases of primary GISTs respectively. Among patients with acquired resistance to imatinib (IM), secondary KIT mutations in exons 13, 14, 17, and 18 are common[4].
Accordingly, in this study, we used immunohistochemistry and next-generation sequencing (NGS) to identify rare KIT mutation-types, excluding exon 11 mutation, in 95 patients with GIST, enrolled at the People’s Hospital of Zhengzhou University, from July 2010 to August 2017. In addition, we examined the clinicopathological features of GISTs associated with rare mutations, and determined whether different mutations vary with respect to the prediction of the clinical outcome. The inclusion criteria were: (1) pathologically confirmed GIST with at least one measurable lesion and the pathological parameters examined included[1-2]: tumor size, morphology (spindle, epithelioid, or mixed), mitotic index [5/50, > 5/50 high-power fields (HPF)], risk (National Institutes of Health); (2) did not receive IM treatment before operation; and (3) had complete and accurate immunohistochemistry [i.e. CD117, CD34, DOG-1, succinate dehydrogenase subunit B (SDHB)[5], Ki-67] and genetic sequencing results.
All 95 patients enrolled in the study provided written informed consent for NGS testing and research (Geneseeq Technology Inc., Nanjing, Jiangsu, China). Briefly, 5 to 10 tumor sections (5 μm) were placed in Eppendorf tubes. Genomic DNA was extracted from tumor tissues using DNeasy Tissue Kits (Qiagen Inc., Germantown, MD, USA). The KAPA Hyper Prep kit (Kapa Biosystems, Wilmington, MA, USA) was used for DNA library preparation as it is a versatile reagent kit adapted to the Illumina platform. Customized xGen lockdown probes were used (Integrated DNA Technologies Inc., Coralville, IA, USA) for hybridization enrichment. The probe panel was designed to target 39 tumor-specific genes. Hybrid Capture Selection was performed using a NimbleGen SeqCap EZ Hybridization & Wash Kit (Roche Inc., Indianapolis, IN, USA) and Dynabeads M-270 Streptavidin (Life Technologies, Waltham, MA, USA). All procedures were performed according to the manufacturer’s instructions. High quality reads were mapped to the human genome (hg19, GRCh37 Genome Reference Consortium Human Reference 37) using modified BWA aligner 0.7.12 with BWA-MEM algorithm and default parameters to create SAM files[6].
Overall, 80 of 95 cases were screened for KIT mutations (84.2%). Most had mutations in exon 11, only 16 of 80 mutations were in other KIT exons (20%, 16/80). Nine cases had exon 9 mutations (11.2%, 9/80), four cases had exon 13 mutations (5%, 4/80), and three cases had exon 17 mutations (3.8%, 3/80). The 16 patients belonged to the age range of 28 to 78 years (median, 64 years), with no difference in sex ratio (male: female, 1:1). Pain, dyspepsia, abdominal mass, and bleeding were the most common symptoms reported by the patients at presentation. The tumors were located in the small intestine (81%, 13/16) and antrum of stomach (19%, 3/16). One case with GIST that originated in the stomach had synchronous adenocarcinoma of the colon. The mean size of the primary GIST was 7.3 cm (range, 1.6–15 cm). Synchronous colon cancer showed a moderately differentiated tubular adenocarcinoma with a maximum diameter of 3 cm. One of sixteen cases had peritoneal metastasis at the time of diagnosis. Histologically, 15 of the 16 tumors were spindle cell type, and 3 of the 16 tumors showed hemorrhage, neoplastic necrosis, and cystic degeneration. Mitotic activity was < 5 per 50 HPF in 14 of 16 cases (87.5%) and 6–10 per 50 HPF in 2 cases (12.5%). The distribution of risk was very low in 2 of 16 cases (12.5%), intermediate in 2 cases (12.5%), and high in 12 cases (75%). All GISTs were strongly and diffusely positive for CD117, DOG-1, SDHB, and CD34 (75%), indicating that they did not belong to the SDH-deficient GIST type. The Ki-67 index was < 5% (Table 1).
Case No. Site Size, cm Histology Risk Codon Mutation Amino acid 1 Ileum 5.8 Spindle cell High KIT exon 9 Duplication 502-503 2 Duodenum 6.0 Spindle cell High KIT exon 9 Duplication 502-503 3 Ileum 9.0 Spindle cell High KIT exon 9 Duplication 502-503 4 Ileum 8.5 Spindle cell High KIT exon 9 Duplication 502-503 5 Ileum 7.5 Spindle cell High KIT exon 9 Duplication 502-503 6 Duodenum 5.6 Spindle cell High KIT exon 9 Deletion insertion 501 7 Ileum 9.0 Spindle cell High KIT exon 9 deletion insertion 501 8 Jejunum 15.0 Spindle cell High KIT exon 9 deletion insertion 501 9 Jejunum 6.5 Mixed cell High KIT exon 9, CCNYL1-BRAF Substitution, Fusion 486 10 Jejunum 7.0 Spindle cell High KIT exon 13 Substitution 642 11 Jejunum 6.8 Spindle cell High KIT exon 13 Substitution 642 12 Jejunum 8.0 Spindle cell High KIT exon 13 Substitution 642 13 Duodenum 1.8 Spindle cell Very low KIT exon 13 Substitution 642 14 Stomach 1.6 Spindle cell Very low KIT exon 17 Substitution 822 15 Stomach 10.0 Spindle cell Intermediate KIT exon 17 Substitution 822 16 Stomach 8.0 Spindle cell Intermediate KIT exon 17 Substitution 822 Table 1. Clinicopathologic features and genetic mutations of GIST
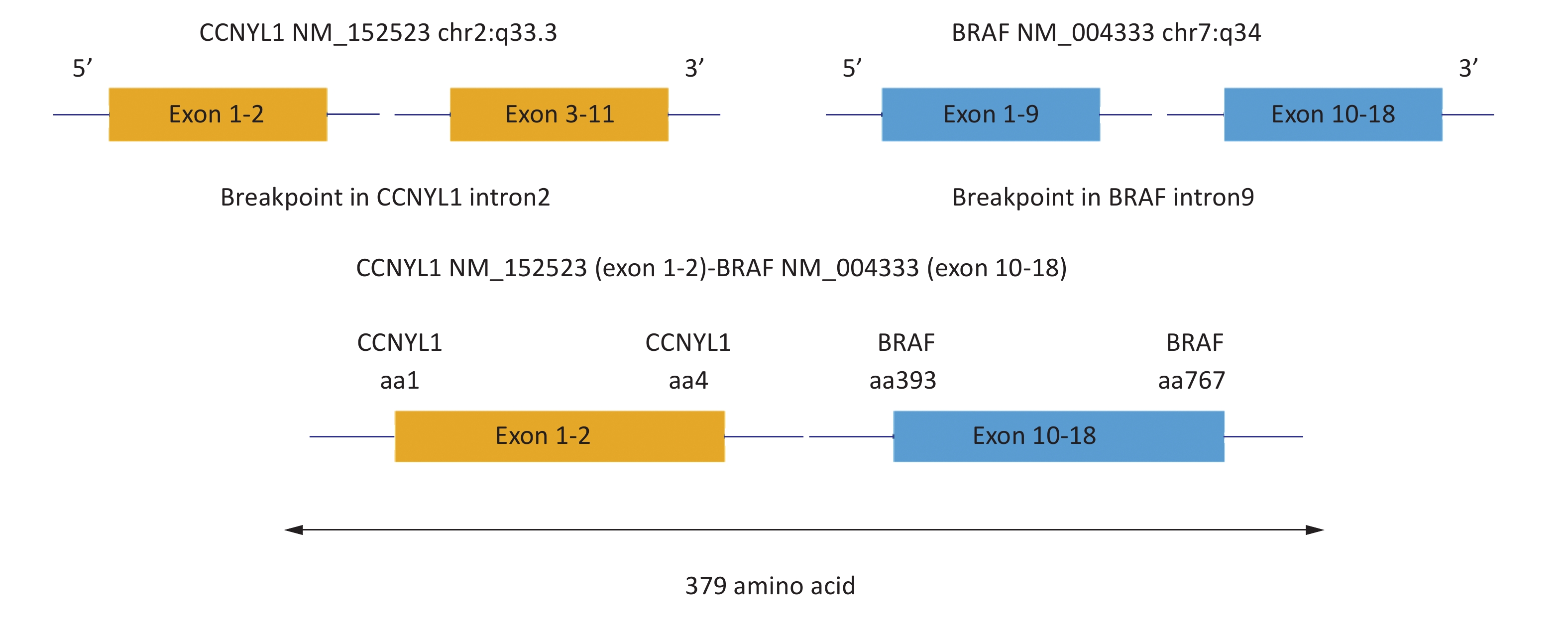
In GISTs, 95% of exon 9 mutations involve a duplication mutation of codon 502 and 503, which is located in the extracellular domain of the KIT protein[5]. This mutation can induce the binding of the receptor and the ligand, thereby, mimicking the binding of the stem cell factor and thus, promoting the dimerization and continuous activation of the protein. Interestingly, most GISTs associated with this type of mutation more often occur in the small or large bowel, and less often in the stomach. Studies have also shown that in non-resectable GISTs, exon 9 mutation is an adverse factor for overall survival and disease-free survival of tumors[1-2]. In this study, we identified nine cases with exon 9 mutations (9/16), five had the common codon duplication mutation at amino acids 502_503 (5/9, p.502-503dup AY, c.1504-1509dupGCCTAT), while three had an uncommon insertion mutation at amino acid 501 (3/9, p.S501delinsSAY, c.1502_1503insTGCCTA), with an allele mutation frequency of 15.15%–21.55%. Only one case had an uncommon T > A substitution mutation in exon 9 at amino acid 486 (1/9, p.N486K, c.T1458A, position chr4:55592134), and the allele mutation frequency was 1.30%. Interestingly, a concomitant novel fusion mutation of the BRAF and cyclin Y like 1 (CCNYL1) genes was also observed in this case, generated by the fusion of exon 1–2 of CCNYL1 on chr2:q33.3 to exon 10–18 of BRAF on chr7:q34, with an allele mutation frequency of 7.30%. The predicted CCNYL1–BRAF protein product contained 379 amino acids comprising the N-terminal amino acids 1–4 of CCNYL1 and C-terminal amino acids 393–767 of BRAF (Figure 1). To our knowledge, this fusion mutation has not been reported previously and was identified here for the first time. Thus, this is the first report of a novel BRAF fusion variant. Specifically, the BRAF gene point mutation has previously been reported in a wide type GIST. CCNYL1 and cyclin Y (CCNY) have overlapping functions[7]. The CCNY family can enhance Wnt/β-catenin signaling in mitosis. CCNYL1 and CCNY are crucial for mouse embryonic development and mammary stem/progenitor cell function, but their physiological roles in mammalian development are yet unknown. Mutation of the CCNYL1 gene has not been reported in GIST, especially the fusion mutation with the BRAF gene.
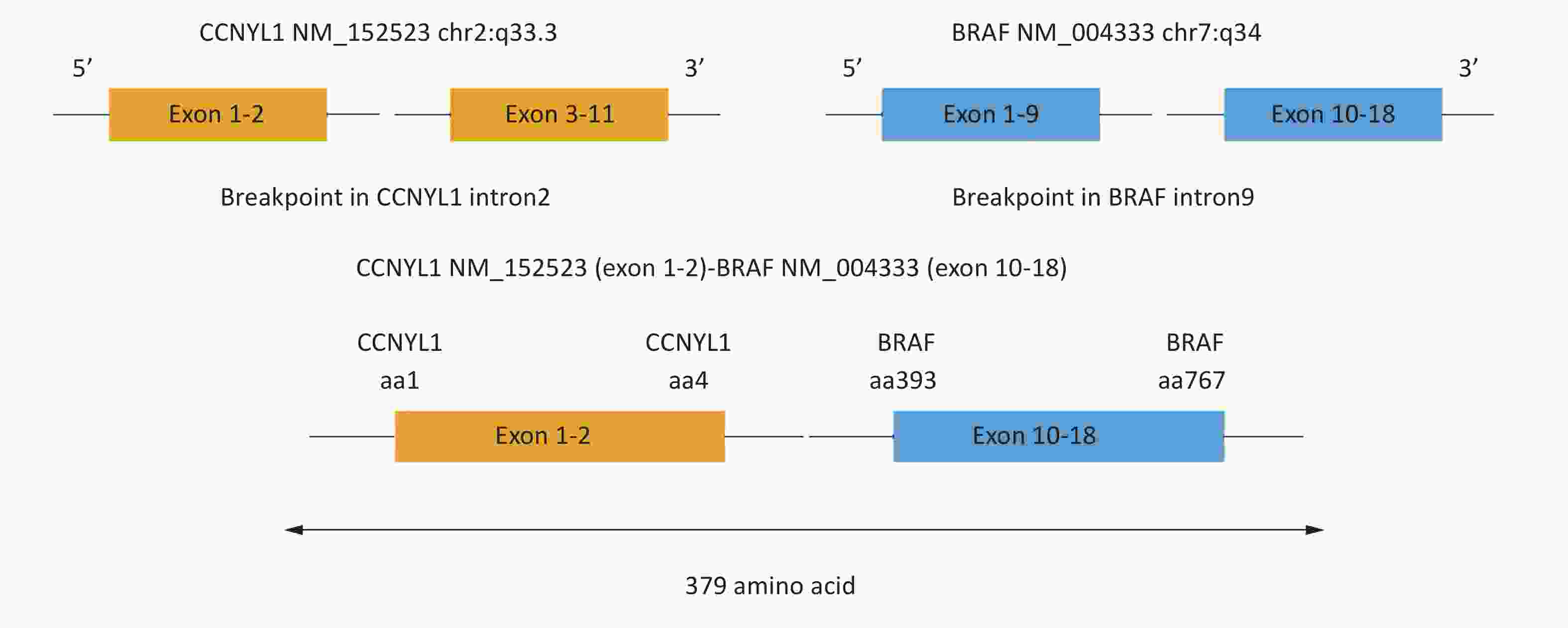
Figure 1. CCNYL1–BRAF rearrangement of GIST. Integrative Genomics Viewer snapshot of CCNYL1–BRAF. Breakpoints were localized at 2q33.3 and 7q34 respectively. Soft-clipped bases match one another in reverse complementarity. Schematic representation of the CCNYL1–BRAF fusion protein domain structure. Orange, CCNYL1; blue, BRAF. The fusion protein is 379 amino acids in length.
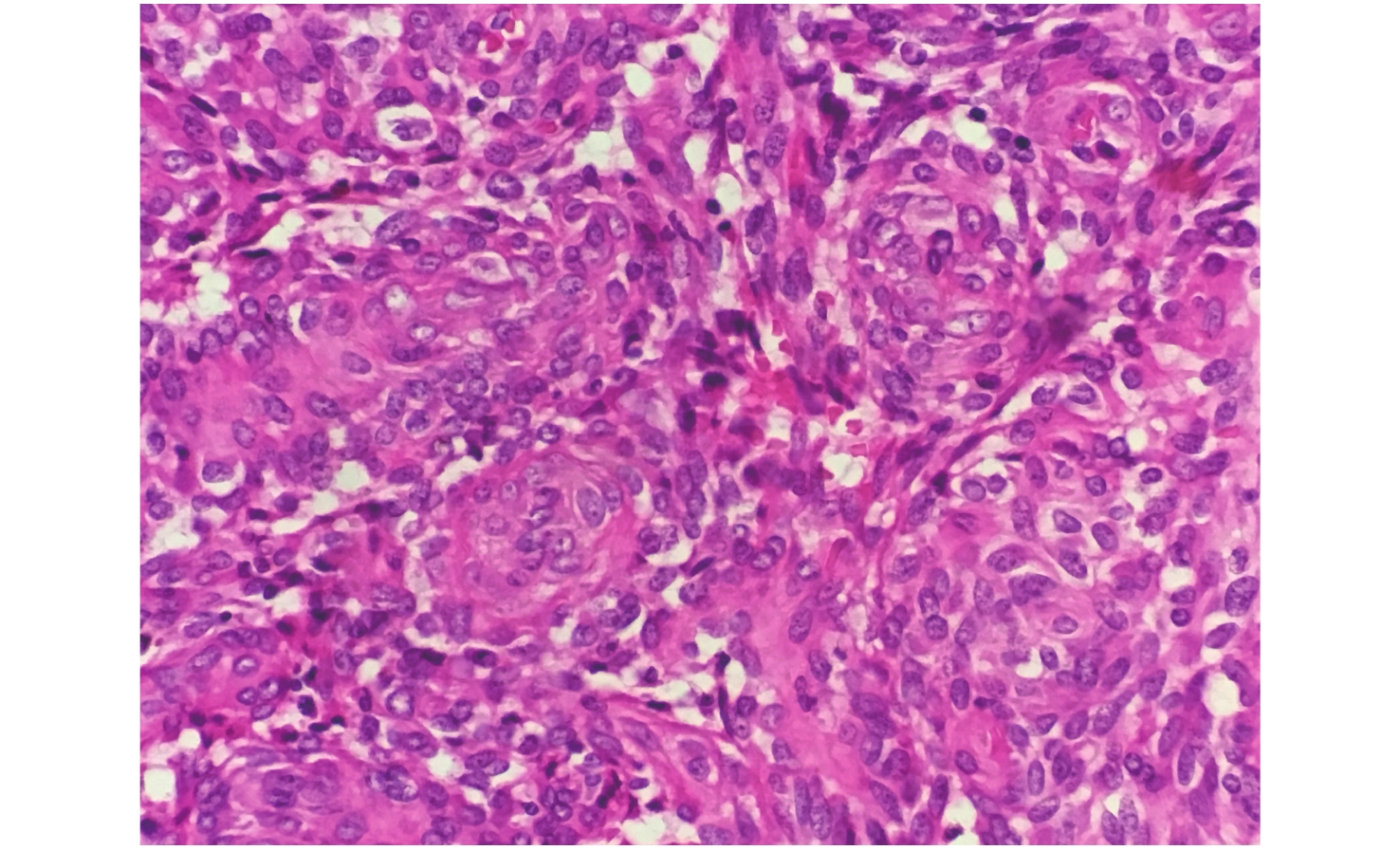
GIST with exon 9 mutation was formed in the intestinal tract, and the tumor cells were mainly spindle cells (8/9), which is consistent with previous reports. The case with a substitution mutation of codon 486 and concomitant fusion mutation of the CCNYL1–BRAF genes presented with a mixed cell morphology (1/9), which was different from the other tumors. Accordingly, in this case, some tumor cells presented with a spindle morphology and were distributed in bands, while other cells presented as characteristic small nests or swirled epithelioid cell clusters, with abundant eosinophilic cytoplasm, round or oval nuclei, a clear nuclear membrane, slightly coarse nuclear chromatin, and small nucleoli, which was similar to a meningioma-like vortex structure. This looked epithelioid in morphology and showed an alternant structure with spindle shaped tumor cells (Figure 2). Existing research shows that GIST with BRAF substitution mutation usually originates in the small intestine and exhibits a spindle cell morphology[8-9]. However, in this case, whether BRAF fusion mutation is the cause of the unique vortex-like tumor morphology as well as the morphology used to predict the type of genetic mutation, remains to be determined by the analysis of more cases. Nonetheless, despite the differing histomorphological manifestations and mutation types of the patients in this group, the biological behavior of the tumors all showed high risk, which is consistent with previous reports.
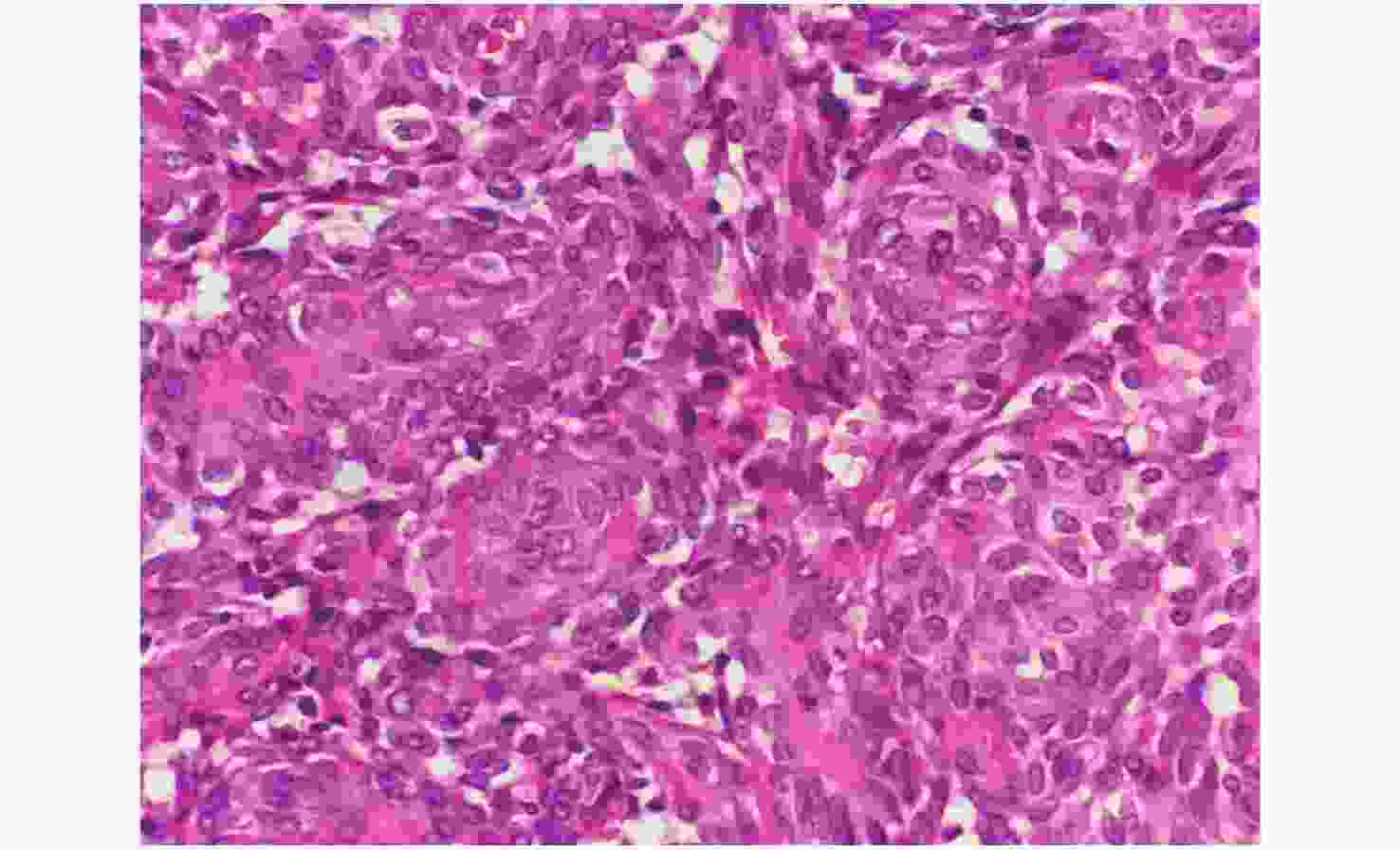
Figure 2. Histopathology of GIST with synchronous CCNYL1–BRAF rearrangement and N486K mutation; the tumor cells with mixed cell type, a portion of the tumor cells with a circinate or small nest growth pattern, abundant eosinophilic cytoplasm, round or oval nuclei, small nucleoli, unique meningioma-like vortex structure (hematoxylin and eosin, 20x).
The most common exon 13 mutation type is A > G substitution of codon 642 (K642E)[3]. This mutation mainly occurs in the ATP-binding pocket of KIT. Further, this tumor often occurs in the stomach and shows a spindle cell histology[2]. The tumor biological behavior is not yet clear. In this study, four cases with exon 13 mutations (4/16, p.K642E, c.A1924G, position chr4:55594221) were consistent with the previous reports, and the allele mutation frequency was 28.05%–39.91%. The tumor cells showed a spindle form, and occurred in the intestine, mainly in the small intestine, which was inconsistent with previous reports. In this group, three cases were associated with a high risk of biological behavior, and one case was very low risk. This suggests that these tumors could mainly be high risk; however, this needs to be confirmed with observations with more cases (Table 1).
The most common exon 17 mutation type is the substitution mutation of codon 822 (N822K)[3]. Most of these tumors mainly occur in the intestinal tract, with a few also being found in the stomach, and they all share a spindle cell morphology[2]. Consistent with previous reports, exon 17 mutations were detected in three cases with spindle cell morphology in our study, with the T > G or T > A substitution mutation at amino acid 822 (p.N822K, c.T2466G or c.T2466A, position chr4:55599340), and the allele mutation frequency was 19.02%–27.53%. Nonetheless, our results show that these tumors appear to arise mainly in the stomach rather than the intestinal tract, which is contradictory to previous reports. In addition, one case presented with synchronous primary intestinal adenocarcinoma. Two cases showed a medium risk of biological behavior, and one case showed a very low risk. Altogether, this suggests that the biological behavior of this type of tumor is still uncertain (Table 1).
According to the guidelines of the European Society for Medical Oncology (ESMO) and National Comprehensive Cancer Network (NCCN)[2], IM treatment should be administered for GIST patients at medium and high risk of biological behavior. According to past studies, GISTs with exon 9 mutations usually show primary resistance to IM, which may be because the exon 9 mutation reduces tumor cell sensitivity to IM. Indeed, a therapeutic effect can be achieved by increasing the dose. However, the functional consequence of a GIST with an exon 13 mutation remains unclear. Consequently, there is no definite consensus on the sensitivity of this type of tumor to IM at present; however, some studies have demonstrated that it is sensitive to IM. Tumors with exon 17 mutations usually present primary drug resistance to IM, similar to tumors with exon 9 mutations. Although resistant to IM, this type of tumor appears to respond to regorafenib[2-4]. Of the 16 tumor cases in this group, 11 showed survival for 20–82 months after diagnosis, with no recurrence, and 9 postoperative patients were administered IM treatment, suggesting that for primary GIST with KIT gene mutation, excluding exon 11, the overall prognosis is good.
In summary, in primary resectable localized GIST, upon excluding the exon 11 KIT mutation, other KIT mutations are mainly found occur in exons 9, 13, and 17 of the KIT gene. Most tumors with these mutations show a spindle cell morphology, mainly occur in the stomach and small intestine, and all exhibit similar immune phenotypes. The most common types of gene mutations are duplication mutations of exon 9 and substitution mutations of exon 13 and exon 17. Exon 9 is associated with rare insertion and point mutations, as well as concomitant CCNYL1–BRAF fusion gene mutation. To our knowledge, this is the first report of this gene mutation, which is associated with a distinct meningioma-like morphology. These rare characteristics identified in this study are worthy of clinical and pathological attention. Generally, tumors with these mutations are more responsive to IM therapy and have a better prognosis.
The authors declare no conflict of interest.
This study does not involve any ethical violations. All tumor tissues are archived samples from the Department of Pathology after surgical resection. Written informed consent was obtained from all the 95 patients for NGS testing and research.
Mutation of the KIT Gene, excluding Exon 11, in Gastrointestinal Stromal Tumors
doi: 10.3967/bes2020.050
- Received Date: 2019-09-07
- Accepted Date: 2020-03-30
| Citation: | LIU Qiu Yu, KONG Ling Fei, XU Zi Gung, LI Zhen, XUE Huan Zhou. Mutation of the KIT Gene, excluding Exon 11, in Gastrointestinal Stromal Tumors[J]. Biomedical and Environmental Sciences, 2020, 33(5): 369-373. doi: 10.3967/bes2020.050 |







 Quick Links
Quick Links
 DownLoad:
DownLoad: