-
With the in creasing use of microwave technology in wireless communication, medical research, and other fields, the risk of energy leakage and excessive human exposure requires more attention. Exposure to a certain frequency of microwave has been found to have harmful effects on multiple organs, including the heart[1-3], which is an important organ for blood circulation and any injury to which can cause a series of adverse consequences.
Studies have shown that microwaves can induce injuries to heart structures and affect heartfunctions[4-5]. However, these studies focused on the biological effects of a single frequency of microwave radiation on the heart. Frequency-dependent and cumulative effects have been rarely reported. Currently, no study has been reported on the dose-dependent, frequency-dependent, and cumulative effects of 2.856 GHz and 1.5 GHz microwaves on cardiomyocyte damage. In the daily working environment of microwaves, multiple frequencies or bands coexist. Therefore, it is important to study the cumulative effects of exposure to multi-band microwaves on the heart.
Autophagy plays an important role in heart disease and injury. Beclin-1 and LC3 are important factors in autophagy[6-7]. We found that 5 mW/cm2 and 10 mW/cm2 microwave radiation could cause damage to heart function, tissue structure, and ultrastructure in rats. These changes were positively correlated with radiation dose[8]. However, whether autophagy is involved in microwave-induced myocardial injuries remains unknown. Thus, we investigated the relationship between autophagy and the biological effects of combined and single microwave exposure.
We used 175 adult male Wistar rats (average weight: 200 ± 20 g) obtained from the Laboratory Animal Center (Beijing, China). The ambient temperature and relative humidity were maintained at (22 ± 1) °C and 60%, respectively. Five rats were housed per cage and were given access to food and water ad libitum. They were kept on a 12 h light-dark cycle (lights on at 7 AM.). All animal procedures complied with the guidelines of the Laboratory Animal Center of Beijing Institute of Radiation Medicine, following approval of the Animal Welfare and Ethics Committee of the Beijing Institute of Radiation Medicine (Beijing, China). All surgeries were performed under sodium pentobarbital anesthesia, and efforts were made to minimize animal suffering.
Here, we used the character “S” to represent the 2.856 GHz microwave exposure group because 2.856 GHz belongs to the S-band microwave frequency range. Likewise, we used the character “L” to represent the 1.5 GHz microwave exposure group.
The grouping and microwave exposure system were based on a previous study (Tan et al., 2017). The rats were randomly divided into seven groups (n = 25 per group) and the Specific Absorption Ratio (SAR) value for each group was also calculated: (1) control group (C group, 0 W/kg); (2) 5 mW/cm2 microwave (2.856 GHz) exposure group (S5 group,1.7 W/kg); (3) 5 mW/cm2 microwave (1.5 GHz) exposure group (L5 group, 1.8 W/kg); (4) 10 mW/cm2 microwave (2.856 GHz) exposure group (S10 group, 1.7 W/kg for the first 6 min and 1.8 W/kg for the last 6 min); (5) 10 mW/cm2 microwave (1.5 GHz) exposure group (L10 group, 3.3 W/kg); (6) 5 mW/cm2 cumulativemicrowave exposure group (SL5 group, 3.7 W/kg) and (7) 10 mW/cm2 cumulative microwave exposure group (SL10 group, 3.3 W/kg for the first 6 min and 3.7 W/kg for the last 6 min).
The rats were fixed in a polypropylene cage, and the radiation table was rotated to ensure whole-body radiation exposure. Rats from the exposure groups received microwave exposure for 6 min. The microwave generator, a klystron amplifier model JD 2000 (Vacuum Electronics Research Institute, Beijing, China), was able to generate pulsed microwaves at 2.856 GHz and 1.5 GHz.
The indicators tested in our study were the activities of aspartate transaminase (AST), lactate dehydrogenase (LDH), and creatine kinase (CK). A multichannel physiologic recorder and supporting analysis system were used for electrocardiography. Histological andultrastructural abnormalities and autophagic activities in the heart were observed after microwave radiation exposure. SPSS version 19.0 (IBM SPSS Statistics, China) was used for statistical analyses.
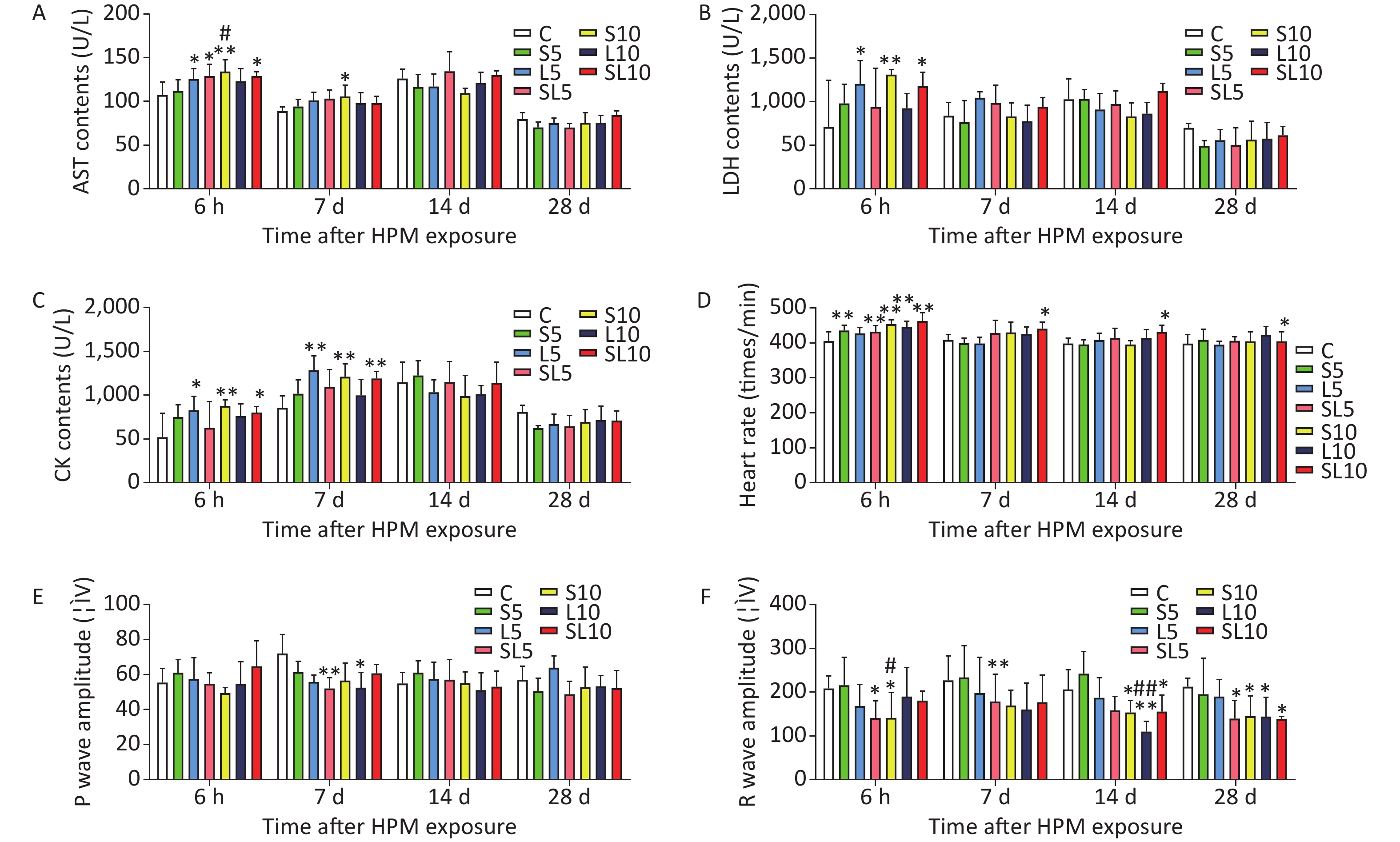
The accepted level of significance for all the tests was P < 0.05. According to the comparison target and definitions, significance markers were classified as follows: significant exposure effects, *P < 0.05, **P < 0.01, (vs. C); significant frequency-dependent effects, △P < 0.05, △△P < 0.01, (L10 vs. S10 and L5 vs. S5); significant dose-dependent effects, #P < 0.05, ##P < 0.01, (S10 vs. S5 and L10 vs. L5) and significant interaction effects, ▲P < 0.05, ▲▲P < 0.01.
We found that after microwave radiation, them yocardial enzymes content in rat sera increased. The mean AST, LDH, and CK contents (U/L) in rat serum (n = 5) were measuredat 6 h, 7 d, 14 d, and 28 d after microwave exposure (Figure 1A–C). Abnormalities were observed in the rat ECG after microwave radiation. The heart rate, P waves, and R waves of the rats were determined at 6 h, 7 d, 14 d, and 28 d after exposure (Figure 1D–F).
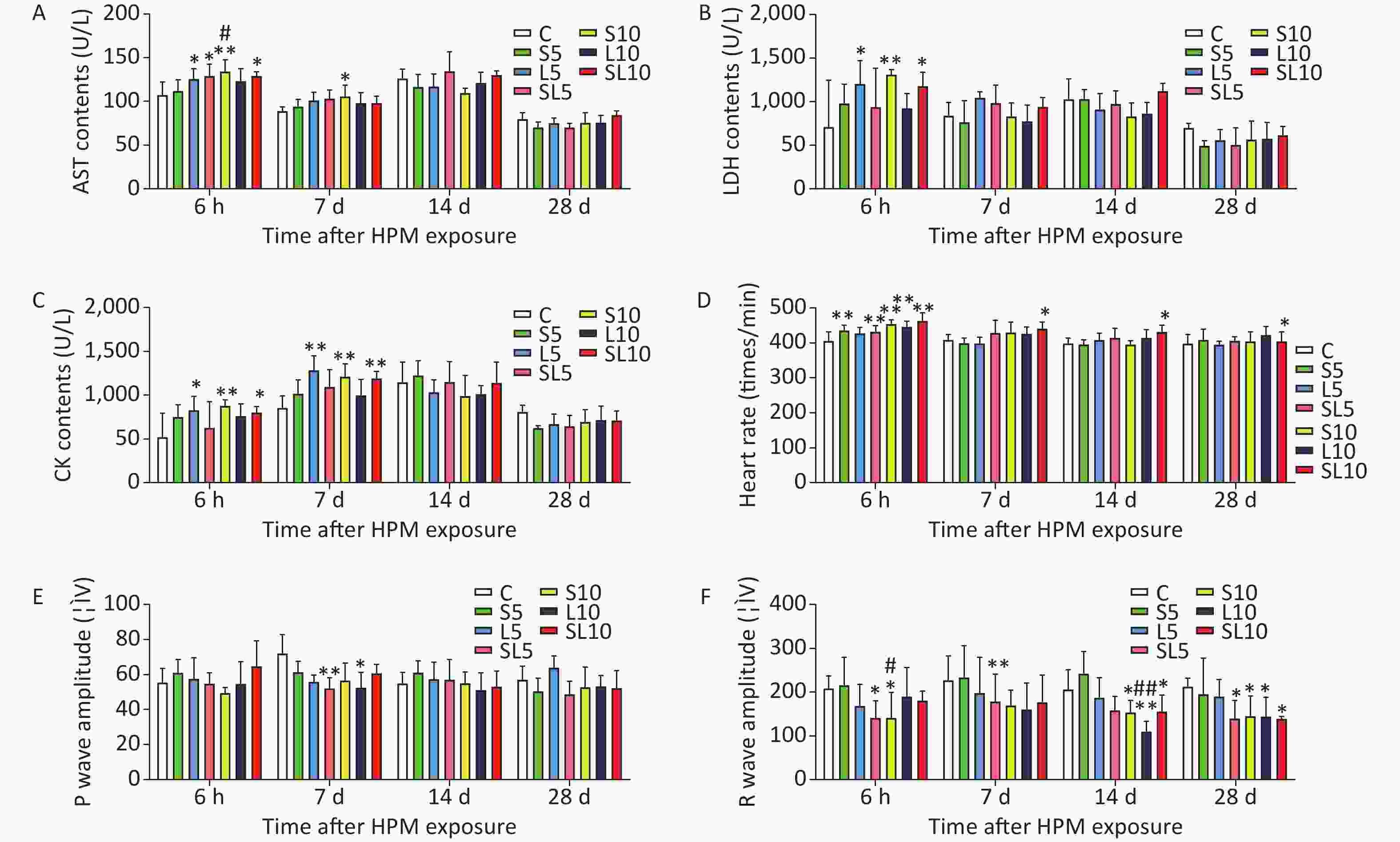
Figure 1. Changes incardiac function of therats. (A) Ratserum aspartate transaminase (AST) content changes. (B) Rat serum lactate dehydrogenase (LDH) content changes. (C) Rat serum creatine kinase (CK) content changes. (D) Rat heart rate changes. (E) Rat P waves changes. (F) Rat R waves changes. Compared with the control group (C), *P < 0.05 and **P < 0.01; significant dose-dependent effect (S10 vs. S5 and L10 vs. L5), #P < 0.05 and ##P < 0.01
Results of the ECG and myocardium enzymes content showed that the changes in the 10 mW/cm2 microwave groups were more serious than those in the 5 mW/cm2 microwave groups. The differences between the 2.856 GHz groups and the 1.5 GHz groups were not significant. Cardiomyocyte injury induced by microwave was mainly related to the power density rather than to the frequency of the microwave. Results of the ECG and myocardium enzymes content indicated that the changes in some of the cumulative microwave exposure groups were to some extent more significant than in those of the single microwave exposure groups. However, there were no interactions between the different frequencies, which were also confirmed by the morphological observations. There might be acumulative effect in cardiomyocyte injury induced by microwave exposure.
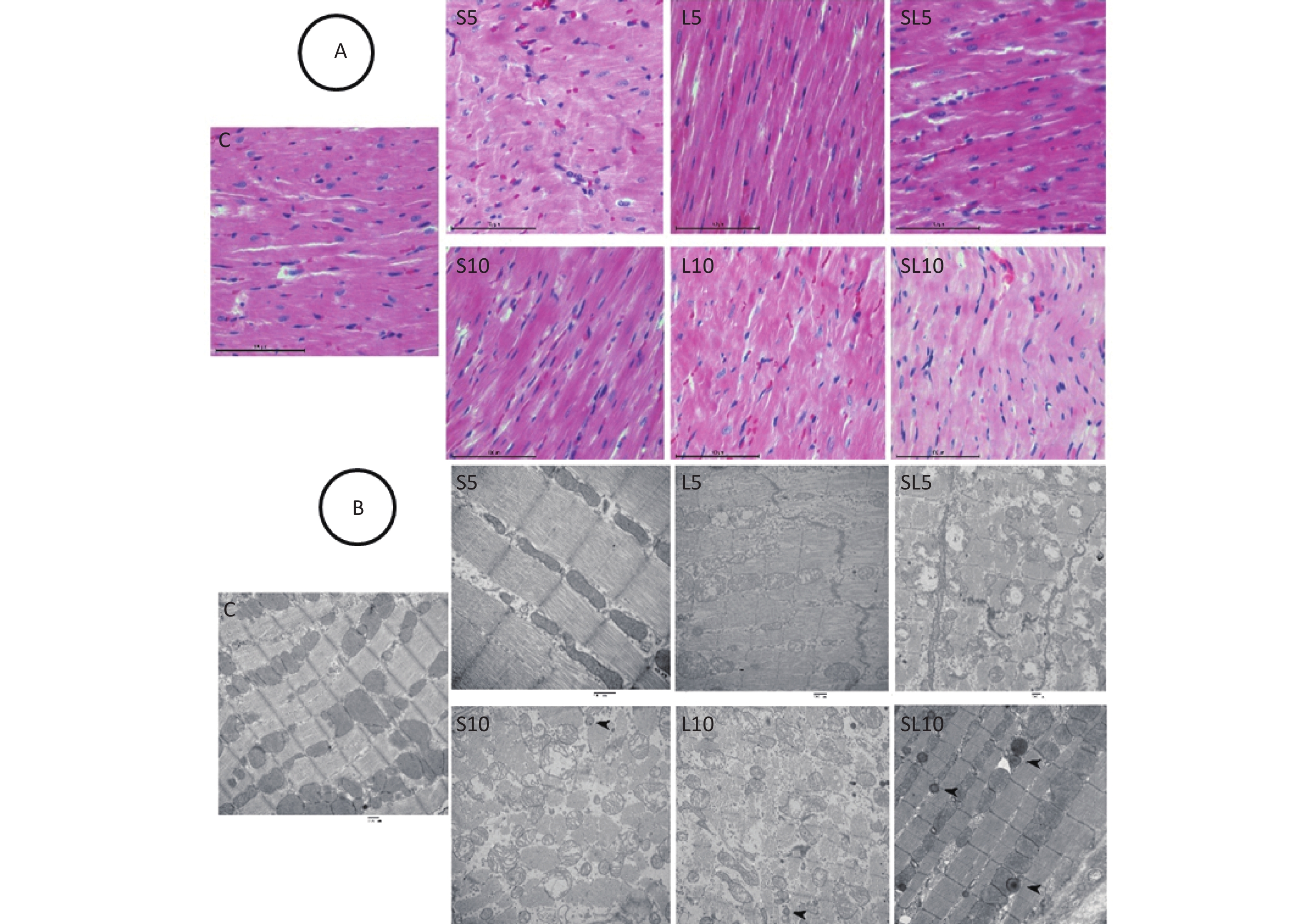
Histological and ultrastructural abnormalities in the heart were observed after microwave exposure. We selected seven days after radiation exposure as the morphological time point of view in myocardial cell injury because we found that the injury effects were most obvious seven days after microwave exposure. Histological observation of the rat hearts showed that in the control group, the arrangement of the myocardium fibers was neat, the cytoplasm was abundant, and the nuclei were oval and centrally located. Seven days after microwave exposure, the myocardium fibers in the L5 and S5 groups still had a regular arrangement and few myocytes displayed swelling. In the L10 and S10 groups, the myocardium fibers were in a wavy arrangement and some myocytes displayed nuclei pyknosis with dark staining. In the SL5 group, the muscle fibers were disorderly and some nuclei pyknosis was observed. In the SL10 group, a large number of myocardium fibers were in an apparentwavy arrangement and several myocytes displayed nuclear condensation with dark staining (Figure 2A).
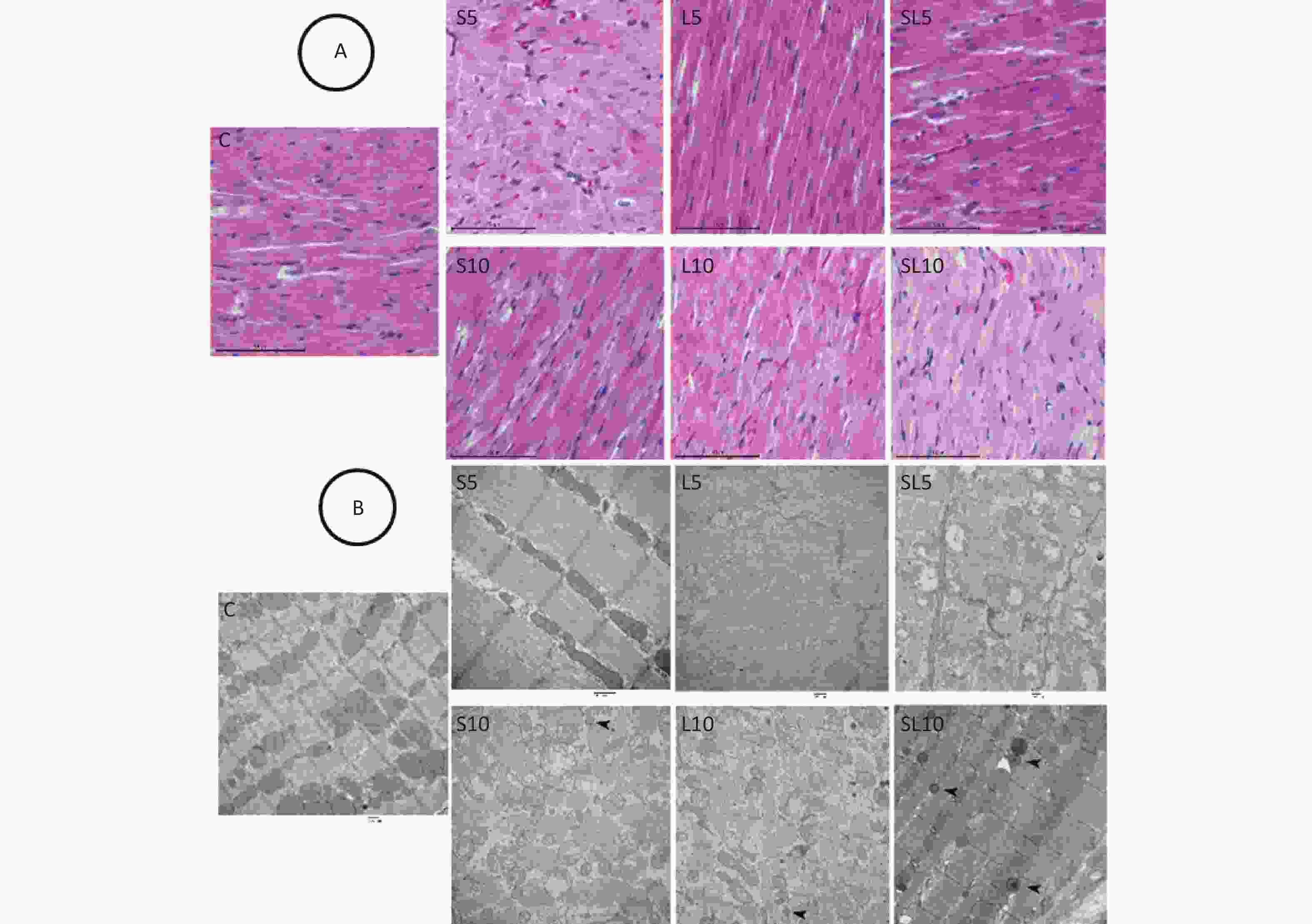
Figure 2. Histological and ultrastructural changes in the heart (H&E staining, scale bar = 100 μm; TEM, scale bar = 500 nm). (A) Seven days after exposure of the L5 and S5 groups, the myocardium fibers were still orderly and few myocytes displayed swelling. In the L10 and S10 groups, the myocardium fibers were in a wavy arrangement and some myocytes displayed nuclei pyknosis with dark staining. In the SL5 group, the muscle fibers were disorderly and some nuclei pyknosis was observed. In the SL10 group, a large number of myocardium fibers were inan apparentwavy arrangement and several myocytes hadnuclear condensation with dark staining. (B) In the L5 and S5 groups, the myofilaments were in a regular arrangement and few mitochondria were swollen and vacuolated. In the SL5 group, few autophagosomes were seen occasionally. In the S10 and L10 groups, the myofilaments were disorderlyand broken and the mitochondria were swollen and vacuolated. In the SL10 group, many autophagosomes were detected. Arrows indicate autophagosomes.
Pathological changes in the myocyte ultrastructure were observed by TEM. In the control group, the myofilaments were neatly arranged and mitochondria had complete structures. Seven days after microwave exposure, the myofilaments in the L5 and S5 groups had a regular arrangement and a few mitochondria were swollen and vacuolated. In the SL5 group, few autophagosomes were seen. In the S10 and L10 groups, the myofilaments were disorderly and broken, the mitochondria were swollen and vacuolated and some autophagosomes were detected. In the SL10 group, many autophagosomes were detected (Figure 2B).
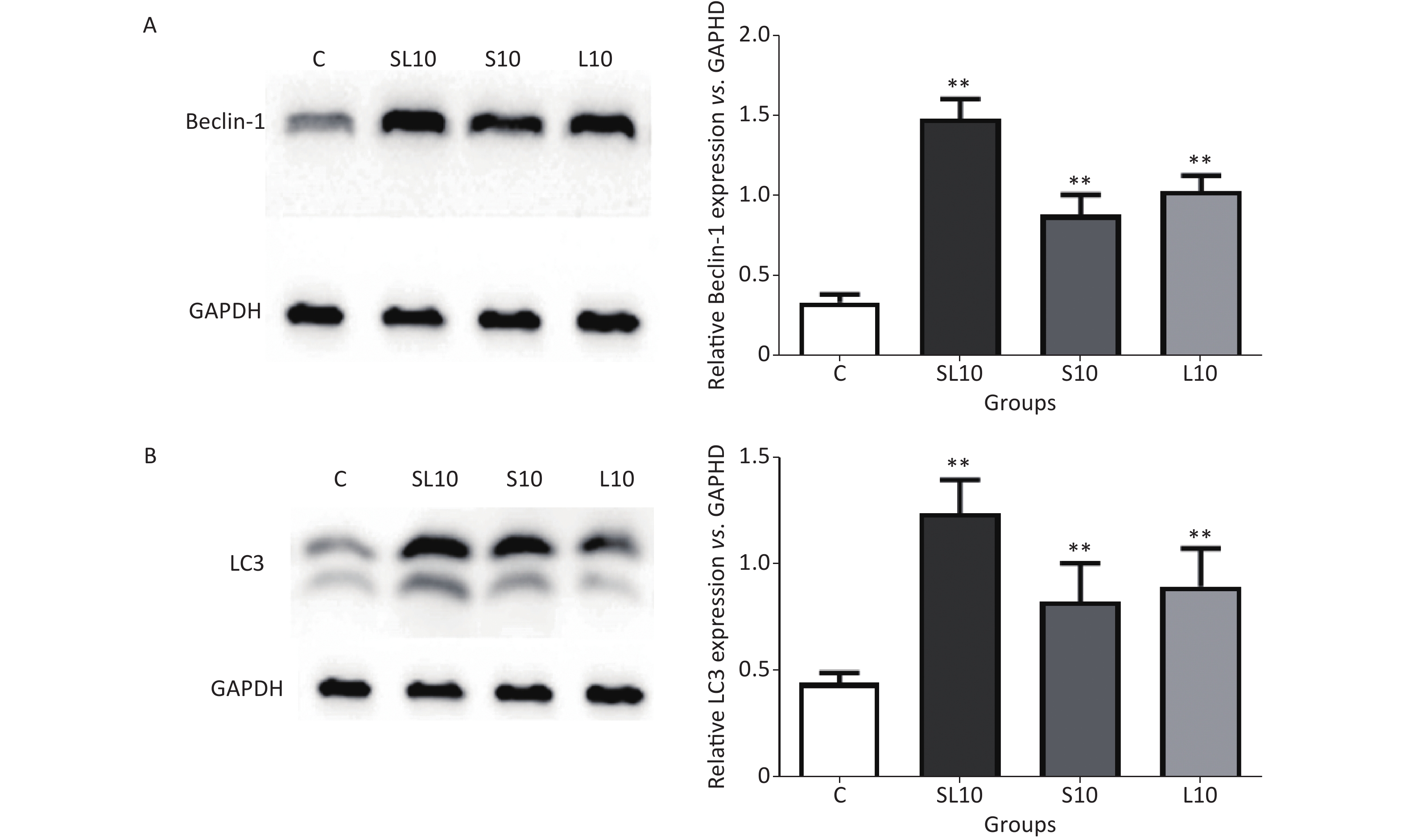
To observe the frequency-dependent and cumulative effects, we selected the 10 mW/cm2 groups (S10, L10 and SL10) for autophagic activity detection at seven days after microwave exposure because of their very apparentlesions. Western blot (WB) was performed to semi-quantify Beclin1 and LC3. An enhancement of the autophagic activities in the heart tissue was observed after microwave radiation.
Beclin-1 and LC3 are very important autophagy markers[9]. Studies have shown that autophagy leads to cell death during myocardial ischemia-reperfusion. Blocking Beclin-l using RNAi or protein could cause autophagy inhibition and cell death reduction[10]. After AMPK/mTOR pathway activation in the acute myocardial infarction model, autophagy was enhanced and LC3 protein expression increased[11]. Results of the WB analysis of Beclin-1 and LC3 in rat cardiomyocytes after radiation exposure showed that Beclin-1 and LC3 protein content in the S10, L10 and SL10 groups were significantly increased seven days after radiation exposure, especially in the SL10 group. These results suggest that Beclin-1 and LC3-mediated autophagy may be an important cause of microwave-induced cardiomyocyte injury. Further studies on the Beclin-1 and LC3-related signal pathways are needed to understand the mechanism of microwave-induced cardiomyocyte injury and to develop prevention and treatment measures (Figure 3).
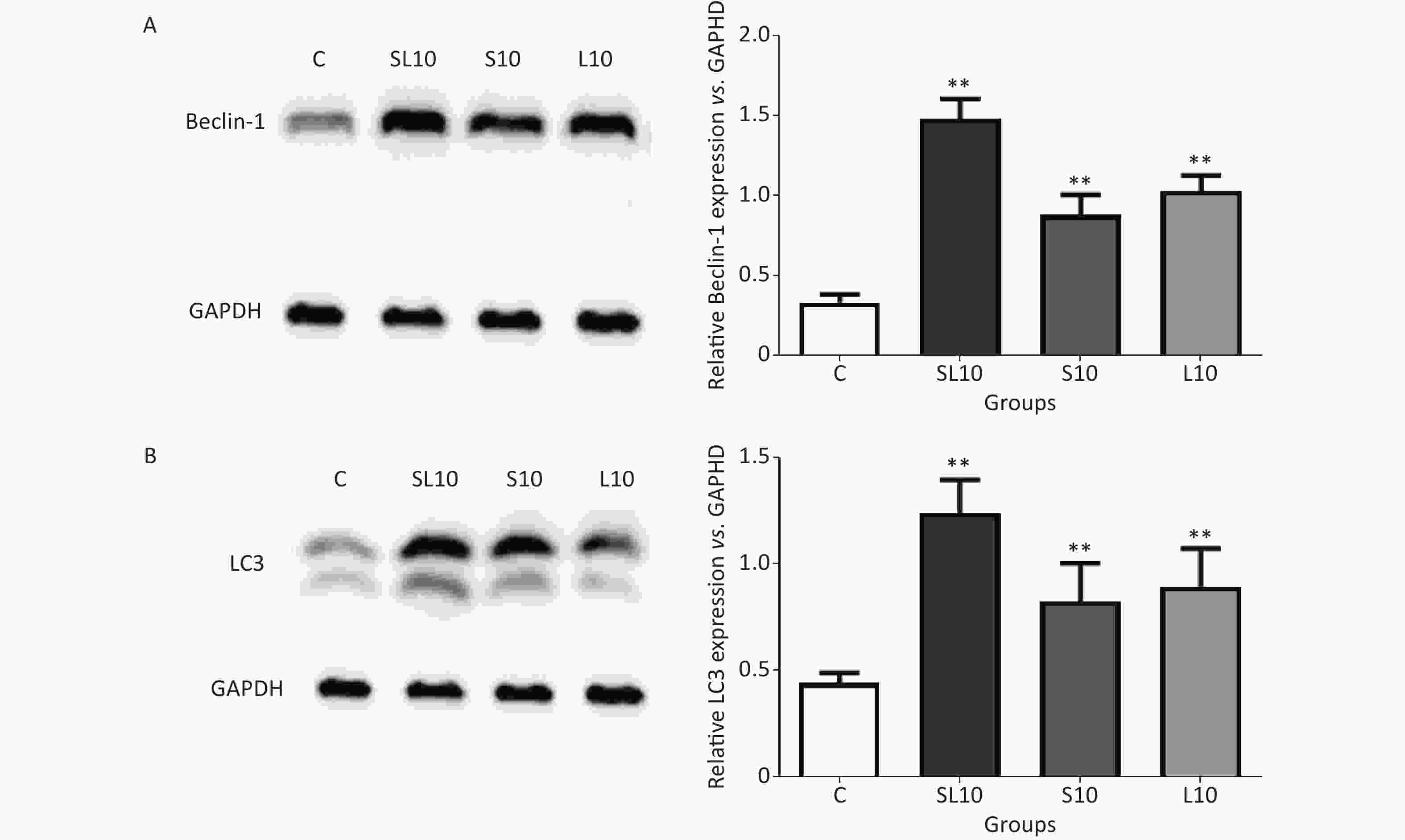
Figure 3. The expression levels of (A) Beclin-1 and (B) LC3 in rats treated with radiation. **P < 0.01 (vs. C group).
In conclusion, cardiomyocyte injury induced by microwave radiation was mainly related to the power density rather than the frequency of the microwave. Cumulative effects might exist in the injury induced by 2.856 GHz and 1.5 GHz microwave exposure. Beclin-1 and LC3-mediated autophagy may be an important cause of the process.
Ethical Approval The experiments were approved by the Ethical Committee of the Academy of Military Medical Science (authorization number: IACUC-AMMS-2010-007).
Authors Contributions ZHANG Bo, ZHANG Jing, and PENG Rui Yun conceived the experiments and designed the study. WANG Hui and YAO Bin Wei conducted the microwave radiation. XU Xin Ping conducted the ECG and image analysis. ZHAO Li and TAN Sheng Zhi analyzed the blood biochemistry and ECG data. DONG Ji and WANG Hao Yu marked the pathological section of HE staining and Western blotting data. ZHANG Bo and ZHANG Jing wrote the manuscript. All the authors reviewed the manuscript.
HTML
Reference







 Quick Links
Quick Links
 DownLoad:
DownLoad: