-
Zika virus (ZIKV) is a mosquito-borne virus of the Spondweni group that belongs to the Flaviviridae family and genus Flavivirus. Microcephaly (small head size, decreased brain size, and poor neurocognitive development) was linked to ZIKV infection in Brazil in 2015. Outbreaks and indications of transmission have spread quickly over the Americas, Africa, and other parts of the globe. Over 80 nations and territories have documented symptoms of Zika infection spread by mosquitoes, including microcephaly and Guillain–Barré syndrome (GBS)[1].
Vaccines are the most cost-effective method for preventing infectious diseases. The latest ZIKV outbreak has warranted the development of a safe and effective Zika vaccine. To protect against ZIKV, the WHO has defined an emergency response, which involves targeted mass vaccination during an ongoing epidemic or an imminent outbreak of ZIKV to prevent ZIKV-associated disease in women of childbearing age and mitigate congenital Zika syndrome (CZS). Until recently, 45 potential Zika vaccines have been developed, including inactivated, live-attenuated, DNA, virus-like particles (VLPs), peptide, chimeric, and mRNA platforms[2]. However, no approved ZIKV vaccine has been made available to the entire population in impacted nations.
When it comes to clinical symptoms, transmission routes, and epidemic areas, ZIKV shares many similarities with other flaviviruses, including Japanese encephalitis virus (JEV), Dengue virus (DENV), Yellow fever virus (YFV), West Nile virus (WNV), Tick-borne encephalitis virus (TBV), and others. Previous studies have demonstrated antigen cross-reactivity between flaviviruses [3-5]. Because there is no ZIKV vaccine, this study aimed to investigate the cross-protection of several licensed flavivirus vaccines against ZIKV and the viability of mass immunization with the current flavivirus vaccine during an active ZIKV outbreak.
According to growing data, virus-specific memory T cell populations have significant protective functions during flavivirus infections in humans and animals. Memory T cells that are cross-reactive with a heterologous virus can provide partial protective immunity and, in certain experimental settings, can determine the difference between life and death for an infected person [6]. The degree of cross-reactivity of memory T cells with ZIKV and their contribution to protective immunity were explored in this study. Cross-reactive cellular immune responses to ZIKV after immunization with JEV, DENV, YFV, and TBV vaccines were observed using enzyme linked immunosorbent spot (ELIspot) and flow cytometry when individuals were stimulated with inactivated complete ZIKV particles. The cross-reactivity of different flavivirus vaccines to ZIKV varied. In the DENV group, both interferon (IFN) γ-spot forming cells (SFCs) and interleukin (IL)-2-SFCs were all significantly increased compared to the control group at all time-points. The reactivity peaked at 569.2 ± 59.9 [mean ± standard error of the mean (SEM), IFNγ-SFCs] and 599.4 ± 51.6 (IL-2-SFCs) in week four, with P(IFNγ) = 0.0002 and P(IL-2) < 0.0001. The proportions of CD4+ IFNγ+ (0.215% ± 0.045%) and CD4+ IL-2+ cells (0.263% ± 0.042%) were significantly higher in the DENV group than that in the control group. However, there was only a slight statistical difference in the cross-reactive CD8+ cell response between the DENV and control groups. In the TBV group, IL-2-SFCs showed a significant statistical difference at all time points, peaking at 219.3 ± 33.9 in week four, with P < 0.0001. No statistical differences were observed in the SFCs for IFNγ between the TBV group and the control. CD4+ IL-2+ showed a significant statistical difference except in week two between the TBV group and the control. Additionally, the statistical difference in CD4+ IFNγ+ was observed from week three to five between the TBV group and the control (Supplementary Figure S1 available in www.besjournal.com). CD8+ IL-2+ in the TBV group was significantly different from the control group in weeks three and four, and there were minor statistical differences in CD8+ IFNγ+ from weeks three to five (Supplementary Figure S1). The levels of IFNγ-SFCs were similar to those of IL-2-SFCs, and there was no obvious gap between the cross-reactive CD4+ and CD8+ T-cell responses in the JEV and YFV groups. The peak value of IFNγ-SFCs were 54.3 ± 15.2 and 28.7 ± 7.8 for the JEV and YFV group, respectively. The IL-2-SFCs of the JEV and YFV group were 122.5 ± 19.1 and 80.0 ± 5.2 at week four, respectively. Cross-reactive CD4+ IFNγ+ was observed from week two to week five in both the JEV and YFV groups, with peaks of 0.123% and 0.137% in week three, respectively (Supplementary Figure S1). Statistical differences in CD4+ IL-2+ levels were only observed in week four, P = 0.0019 in JEV and P = 0.0002 in YFV compared to the control group. CD8+ IL-2+ and CD8+ IFNγ+ in the JEV group showed a statistical difference from the control group from week three to five, whereas in the YFV group, there was a temporary difference only at week three (Supplementary Figure S1). Generally, IL-2-SFCs were more abundant than IFNγ SFCs. Furthermore, CD4+ T-cell responses were higher than CD8+ T-cell responses in all vaccine groups. DENV vaccination elicited the greatest T-cell cross-reactivity with ZIKV, although JEV, YFV, and TBV vaccination all elicited a substantial amount of T-cell cross-reactivity with ZIKV. Flow cytometry revealed that cross-reactive CD4+ T cells were more common than CD8+ T cells in terms of their frequency. Although cross-reactive flavivirus-specific CD8+ T cells may provide universal protection against ZIKV, the complementary involvement of CD4+ T cells in cross-protection has also been described [7,8]. The protective functions of CD4+ T cells in this study imply that CD4+ T cell-mediated responses against ZIKV from flavivirus vaccinations may comprise an optimum cross-reactive T cell response.
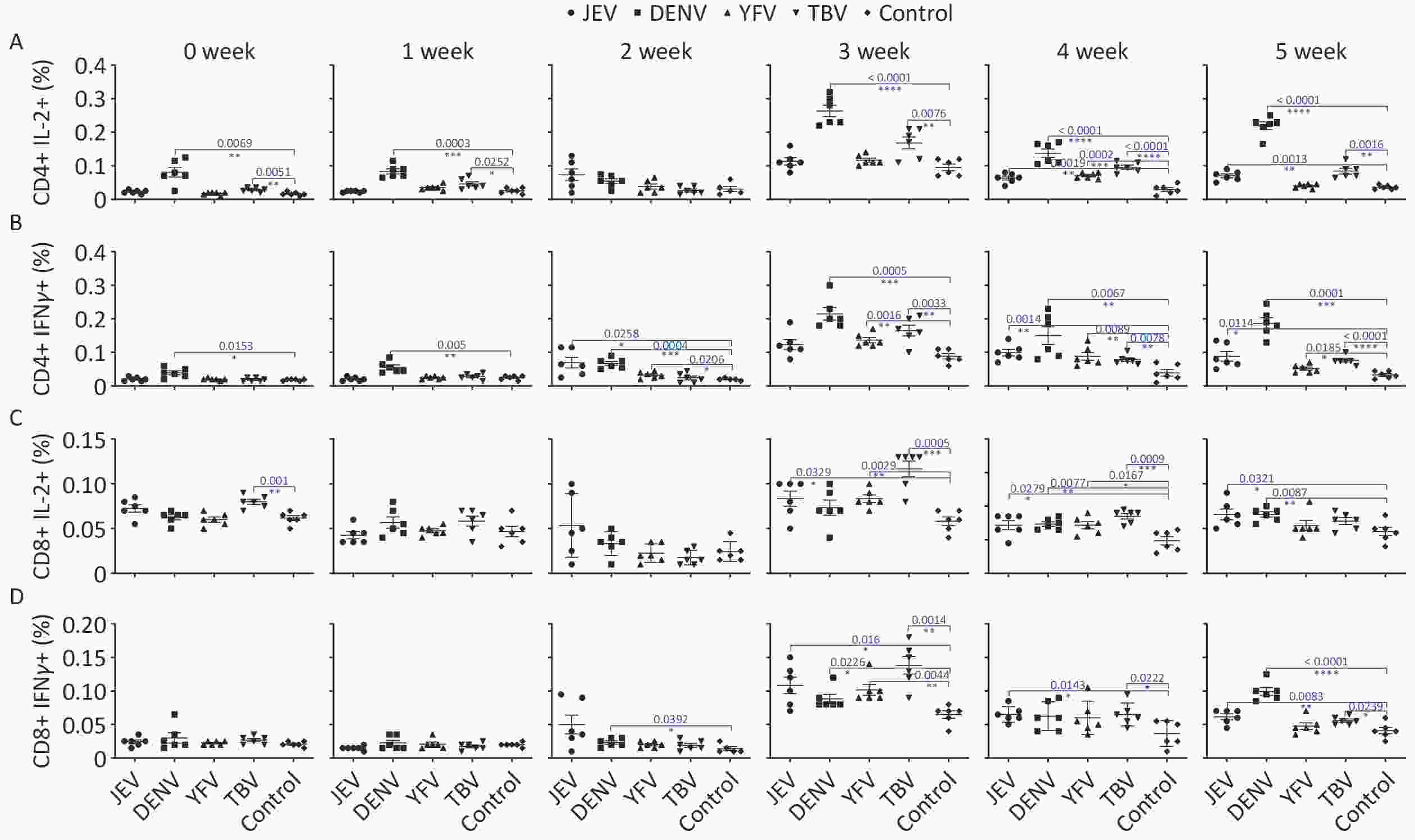
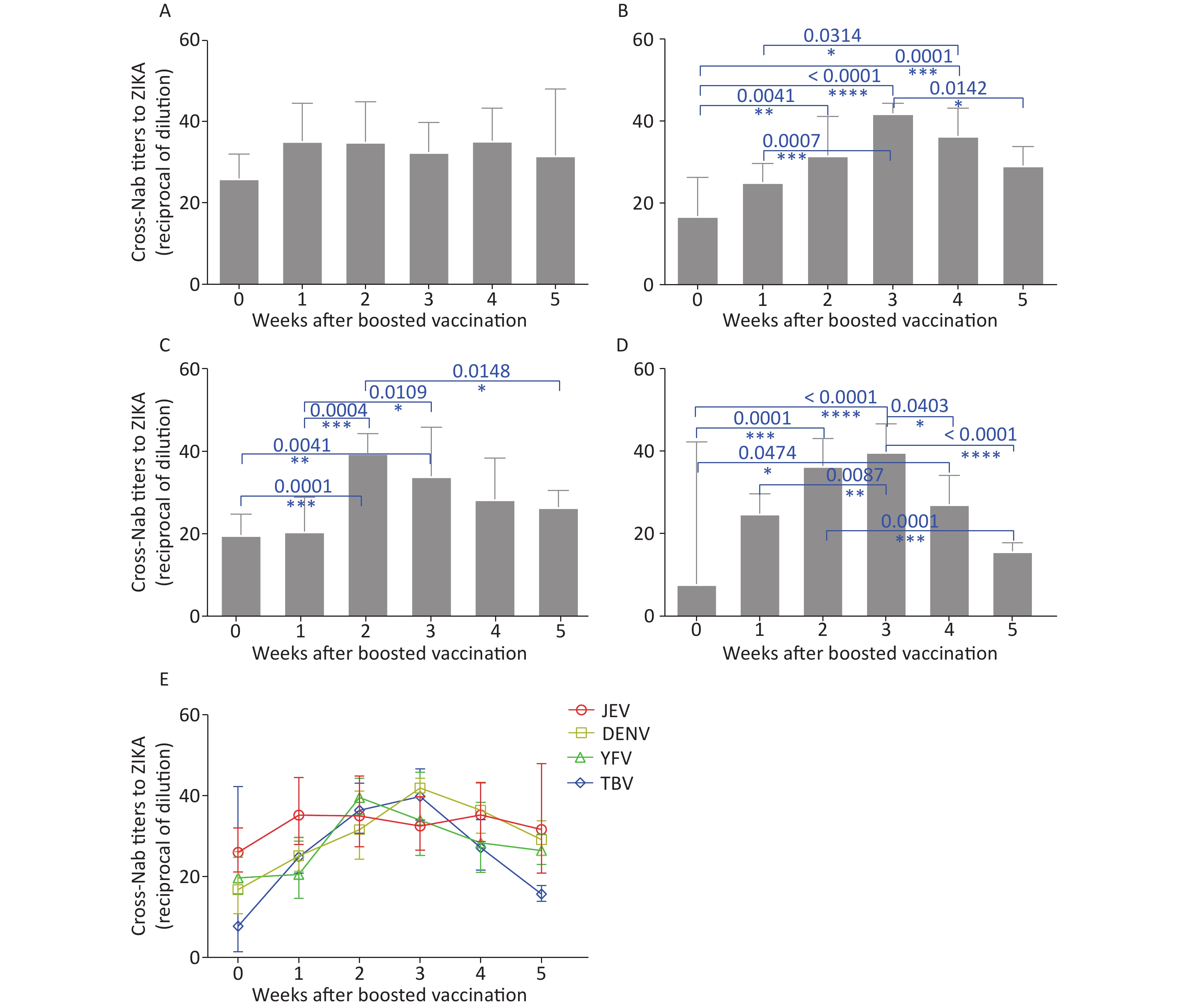
Figure S1. The cross-reactive T-cell responses to ZIKV by flow cytometry. Proportion of CD4+ IL-2+ (A), CD4+ IFNγ+ (B), CD8+ IL-2+ (C), CD8+ IFNγ+ (D) in splenocytes were determined at different time-points when stimulated with Zika virus particles after different flavivirus vaccination
Antibody-based correlates of protection for additional flavivirus vaccines, such as YFV, TBV, and JEV, have been determined, consisting of neutralizing antibody (Nab) titers of > 10 (The titer refers to the maximum dilution ratio that can neutralize 50% of the virus in Plaque reduction neutralization test). When cross-Nab titers against ZIKV were higher than 10, cross-protection to ZIKV was considered present. The cross-NAb titer in the control group did not exceed 1:10. However, cross-neutralization of ZIKV was efficient in all the vaccine groups. The sera-conversion rate in all vaccine groups was 100% at all time points, except for the TBV group (66.7%) in week zero (before the booster vaccination), when the positive rate was based on the 1:10 titer. In the JEV group, cross-NAb titers to ZIKV at different time points did not differ significantly from each other (Figure 1A), and the geometric mean titers (GMT) were 1:26.03, 1:35.23, 1:35.05, 1:32.52, 1:35.28, and 1:31.69 at weeks 0, 1, 2, 3, 4, and 5, respectively. The cross-NAb titers to ZIKV were different between the DENV, YFV, and TBV groups, but changed with a similar trend over time. Initially they increased gradually and then decreased (Figure 1E). In the DENV group, GMT peaked at 1:41.92 in week three, and the most significant statistical difference occurred between week three and zero (P < 0.0001) (Figure 1B). In the YFV group, there was a marked increase in cross-NAb titers between weeks one and two (P = 0.0004) (Figure 1C). YFV elicited the highest cross-NAb level against ZIKV in week two, with a GMT of 1:39.56. In the TBV vaccine group, there was a marked increase in the cross-NAb titer between week zero and two (P = 0.0001), and a marked decline between week three and five (P < 0.0001) (Figure 1D), in which the GMT was from 1:7.70 to 1:36.42, then from 1:39.81 to 1:15.73. Antibodies against ZIKV were significantly cross-reactive in all vaccination groups, which is consistent with previous studies. The highest cross-Nab titers were found in the DENV group, followed by those from the JEV and YFV vaccines, which is likely due to their phylogenetic distance to ZIKV.
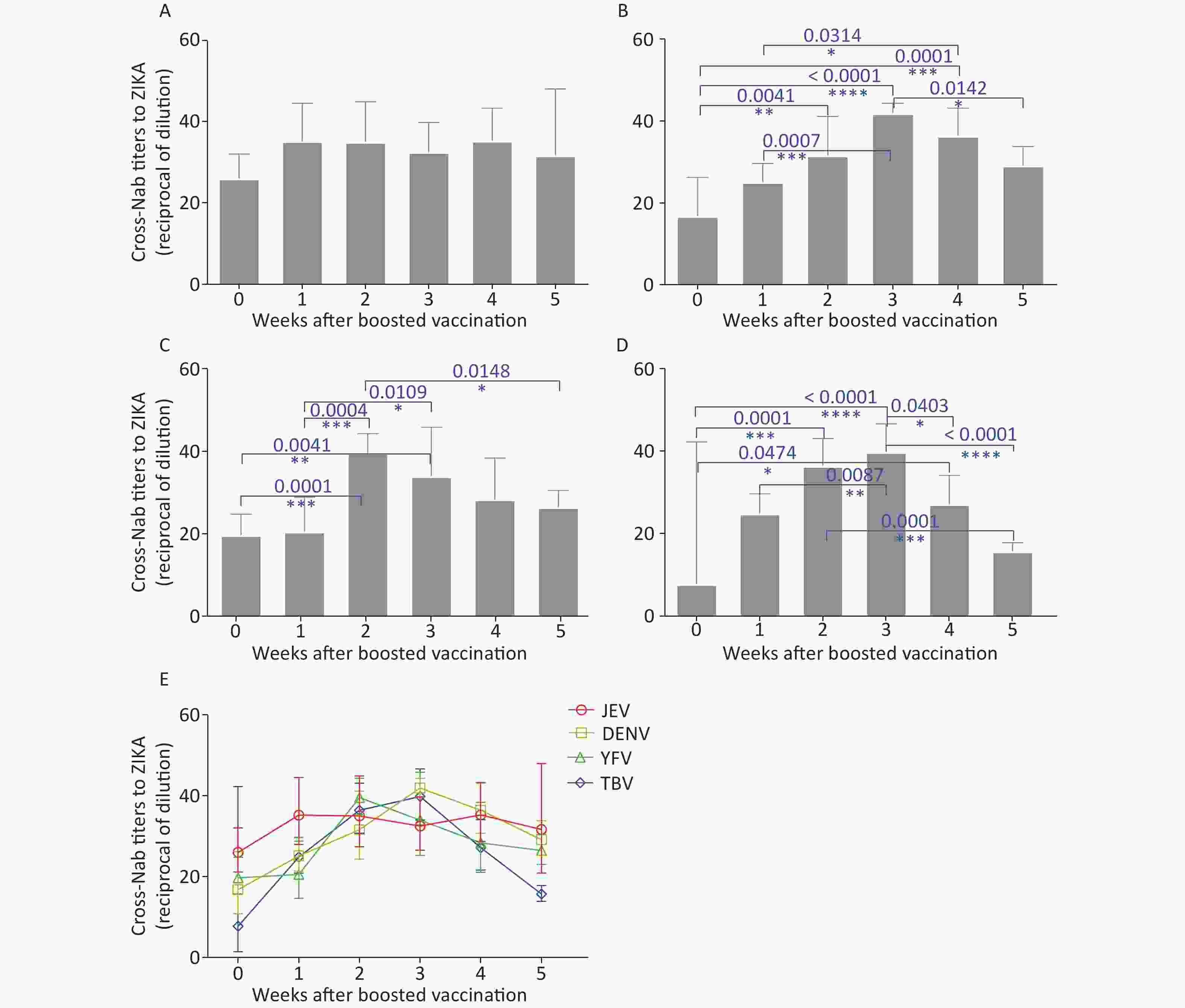
Figure 1. The titer of cross-neutralizing antibodies (NAbs) to Zika virus (ZIKV) by plaque reduction neutralization test (PRNT)among different vaccine groups. The cross-reactive cellular immune responses to ZIKV of the Japanese encephalitis virus (JEV) vaccine group (A), the Dengue virus (DENV) vaccine group (B), the Yellow fever virus (YFV) vaccine group (C), the Tick-borne encephalitis virus (TBV) group (D), and (E) is the summary data. P values of statistically significant differences between different time points in the same vaccine group were shown graphically.
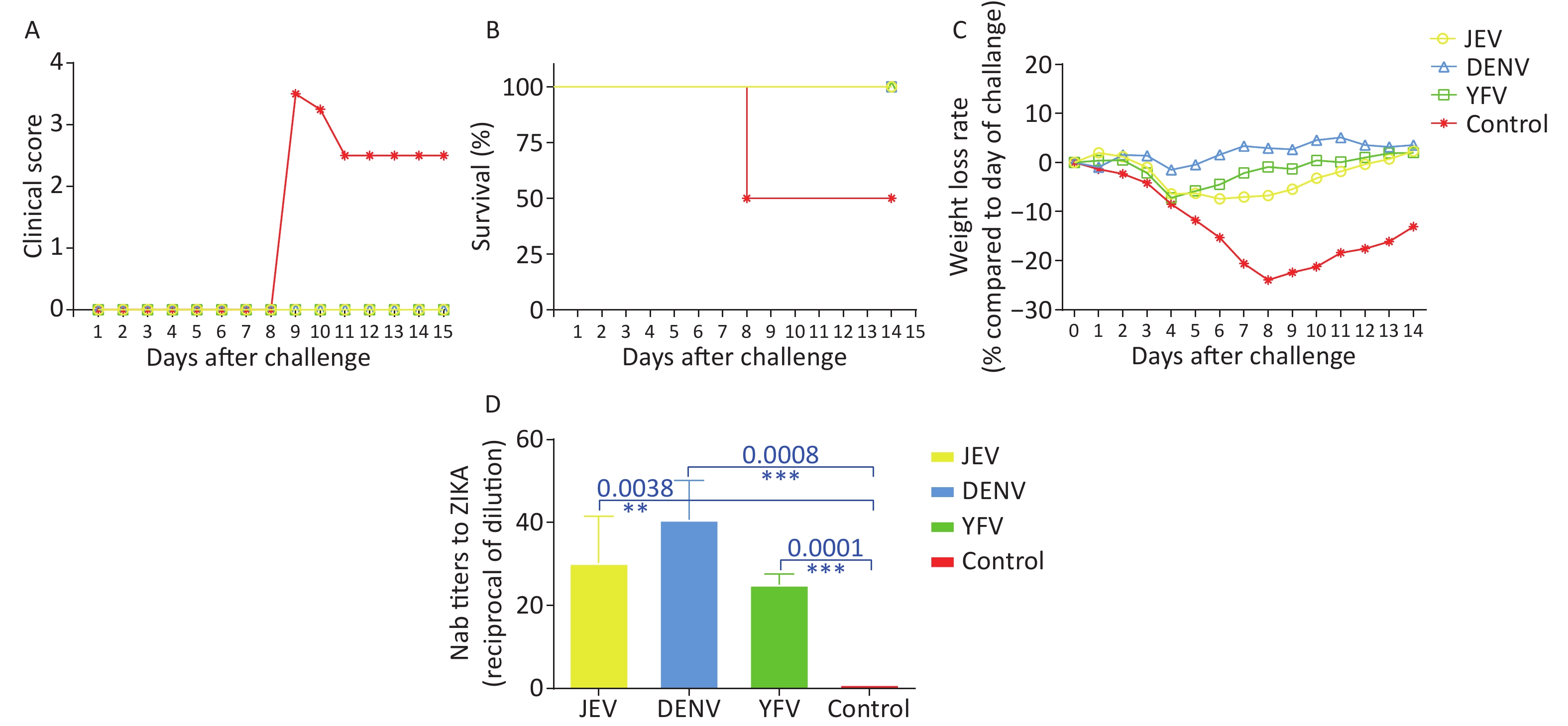
We next tested whether pre-vaccination with the flavivirus vaccine was adequate to protect against ZIKV challenge in IFNα−/β− mice. These mice support high levels of ZIKV replication or succumb at a particular viral challenge dosage, based on the occurrence of cross-reactivity during in vitro testing. Because IFNα−/β− mice are less productive than standard laboratory mice, only 16 IFNα−/β− mice were used in this investigation, with four animals each in the JEV, DENV, YFV, and control groups. Groups of 6–9-week-old IFNα−/β− mice were intravenously (i.v.) challenged with 106 tissue culture infectious dose 50 (TCID50) of ZIKV PRVABC59 two weeks after the final immunization. IFNα−/β− mice in the control group all exhibited signs of disease post-challenge, first noted with an average clinical score of 3.5 on day 8 post-challenge. However, none of the mice in the vaccine groups had any clinical signs recorded during the 14-day study period (Figure 2A). The survival rate in all vaccine groups was 100%, but only 50% of control mice survived (Figure 2B). The control mice started to lose weight rapidly after day two post-challenge, whereas the vaccine groups demonstrated only a temporary weight loss between days two and six post-challenge, followed by an increasing trend throughout the study. These data indicate that the mice were healthy (Figure 2C). Mice in the DENV group exhibited temporary weight loss of -0.96% on day two and −1.48% on day four post-challenge. In the YFV group, weight loss began on day three (−2.15%), reached its lowest point (−7.17%) on day four, and then recovered on day 10 (0.44%). In the JEV group, the weight reached its lowest level on day six post-challenge (−7.39%), recovered slowly, and reached 0.73% on day 12 post-challenge (Figure 2C). Serum samples from IFNα−/β− mice were collected before the challenge. The GMT of cross-NAb titers against ZIKV were 1:30.21, 1:40.67, and 1:25.08 in the JEV, DENV, and YFV groups, respectively. Conversely, the cross-NAb titer in the control group did not exceed 1:10. Compared with the control group, significant differences were observed in the JEV, DENV, and YFV groups with P = 0.0038, P = 0.0008, and P = 0.0001, respectively (Figure 2D).
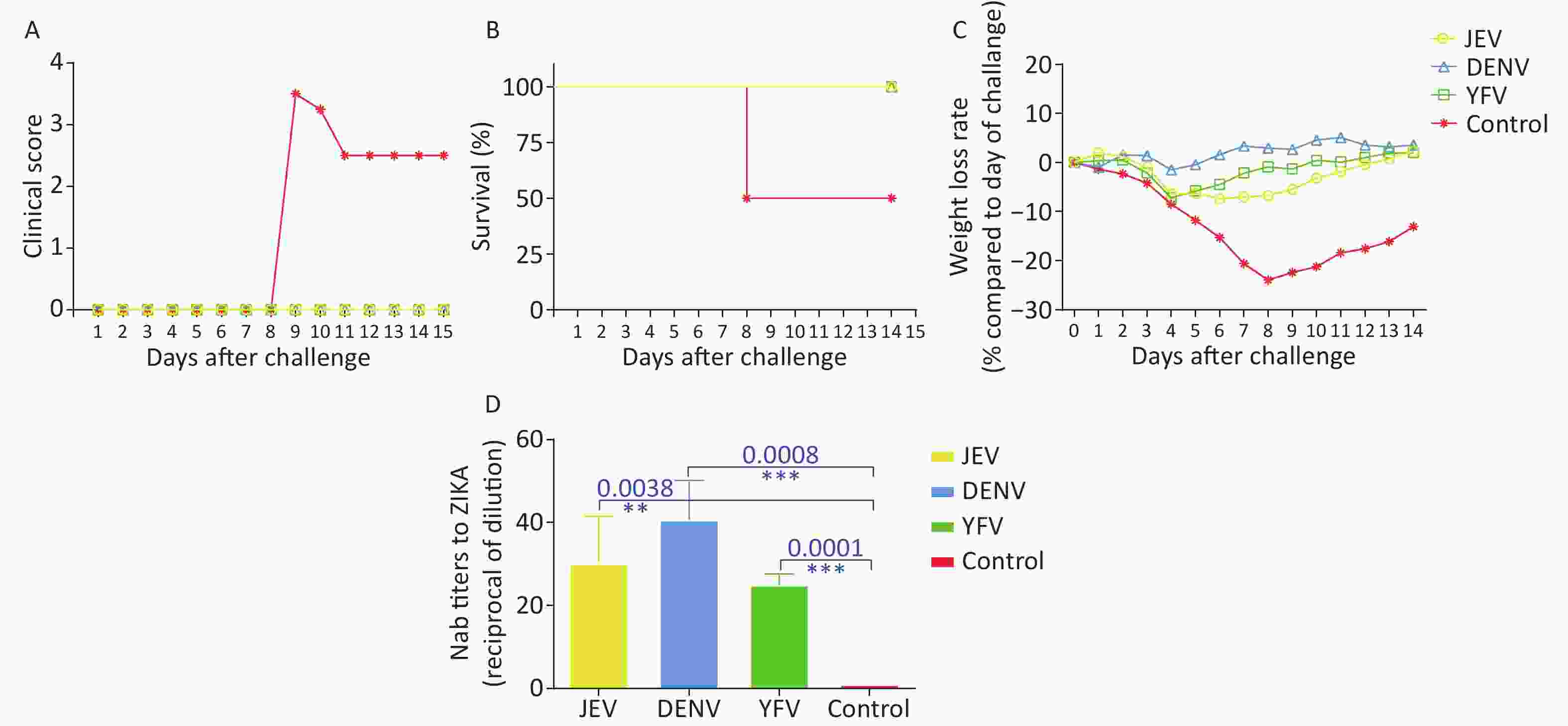
Figure 2. 6–9 weeks-old interferon (IFN)α−/β− mice were used to study the cross-protection to Zika virus (ZIKV) induced by other flavivirus vaccines. (A) Average clinical scores post-challenge; (B) mortality after infection; (C) weight loss of each group; (D) the titer of cross-neutralizing antibodies (Nabs) to ZIKV by PRNT.
Antigenic cross-reactivity across flaviviruses is common, with shared immunogenic epitopes stimulating humoral and cell-mediated immune responses. The interaction between ZIKV and other medically important flaviviruses remains an important issue. Many studies have indicated cross-reactivity between flaviviruses. Our findings, particularly the cross-reactivity between ZIKV and Dengue virus, were consistent with prior findings. Nonetheless, concerns exist that pre-flavivirus immunization may result in more severe Zika infection and antibody-dependent enhancement (ADE). Apart from evidence acquired in vitro, ADE has been documented primarily between ZIKV and the dengue virus, and it has not been described between ZIKV and other flaviviruses.
Furthermore, whether the relationship between dengue and ZIKV is useful or harmful to the viral defense remains controversial. The flavivirus vaccine has been in use for a long time. The 17D yellow fever vaccine has been in use in the United States for 80 years, the Japanese encephalitis vaccine for approximately 60 years, and the TBV vaccine has been used since 1937. In numerous countries, ZIKV has co-circulated with these flaviviruses, and the flavivirus vaccine has been used in several epidemic regions. However, additional flaviviruses and pre-vaccination have not been linked to differences in clinical outcomes. Therefore, more research is needed to determine the effect of the primary flavivirus vaccine on ZIKV infection, which may be considerably more effective than previously thought. According to recent epidemiological investigations, patients with previous DENV infections have no ADE-associated symptoms [9]. Furthermore, epidemiological evidence suggests that YFV immunization may be linked to protection against ZIKV[10].
Elucidating the cross-interaction mechanisms between flaviviruses will enable the development of a ZIKV vaccine. We anticipate that ZIKV vaccination will become available shortly. Nonetheless, approved flavivirus vaccines, such as JEV, YFV, and TBV would be a reasonable alternative for emergency use in ZIKV epidemics since they, are cross-reactive and exert a low risk of ADE.
The authors declare no conflict of interest.
We would like to thank Linlin Bao (Institute of Laboratory Animal Science, Chinese Academy of Medical Sciences) for providing IFNα-/β-mice and ZIKV strain PRVABC59, Liqiang Feng (Guangzhou Institutes of Biomedicine and Heath, Chinese Academy of Sciences) for providing ZIKV GZ02, and thank Editage (www.editage.cn) for English language editing.
WANG Ling and LI Yu Hua conceived of the study. WANG Ling, LIU Jing Jing, FANG En Yue, LI Ming, and LIU Ming Lei performed experiments. WANG Ling, LIU Jing Jing, and FANG En Yue analyzed the data. WANG Ling and LIU Jing Jing wrote this manuscript. All authors have contributed to the manuscript and approved the submitted version.
HTML
Reference
 22047Supplementary Materials.pdf
22047Supplementary Materials.pdf
|

|








 Quick Links
Quick Links
 DownLoad:
DownLoad: