-
Adiponectin, a specific hormone protein secreted by adipocytes, is a protective factor in the human body and has attracted great attention due to its anti-obesity activity[1,2]. The adiponectin gene (ADIPOQ), which is located on human chromosome 3q27, is a single-copy gene. Various single-nucleotide polymorphisms (SNPs) of the human ADIPOQ play an important role in energy balance and are associated with body mass index (BMI) and obesity[3,4]. Long-chain polyunsaturated fatty acids (LC-PUFAs) have been identified to exert an important role during the first 1,000 days of life on infant growth, neurodevelopment, and visual acuity. Fatty acids, such as α-linoleic acid (ALA, C18:3n-3) and linoleic acid (LA, C18:2n-6), which cannot be synthesized de novo from other lipids, are necessary for biological reactions. Some fatty acids such as docosahexaenoic acid (DHA, C22:6n-3) and eicosapentaenoic acid (EPA, C20:5n-3) can be synthesized in neonates from their precursors (LA and ALA). Because of the very low conversion rates in the neonatal liver, it is crucial to provide adequate LC-PUFAs in the diet from early infancy. Therefore, we can infer that fatty acid intake in exclusively breastfeeding infants is entirely dependent on the abundant supply of breast milk. However, the association between ADIPOQ SNPs and maternal fatty acids has not been well explored. Studies have shown that maternal characteristics directly affect the concentration in breast milk, specifically including macronutrients, n-6/n-3, and expression of inflammatory markers in breast milk. However, to the best of our knowledge, this is the first analysis of the association between SNPs in ADIPOQ and breast milk PUFA concentration.
Our study hypothesized that maternal genetic background and pre-pregnant BMI may affect the amount of PUFAs in breast milk during the lactation period. Based on this hypothesis, our study investigated the interaction effect of BMI and the presence of SNPs, rs1501299 and rs1063538, on the secretion of maternal breast milk PUFAs.
The participants were lactating mothers recruited from a postpartum care center in Changchun who agreed to participate in the study. This study was conducted in accordance with the guidelines of the Declaration of Helsinki, and all procedures involving human subjects were approved by the Ethics Committee of the School of Public Health, Jilin University, China (No. 2016-08-01). A total of 423 exclusively breast-feeding mothers aged 20–39 years were recruited between March 2017 and December 2019. Exclusion criteria were as follows: 1) mothers with maternal pregnancy complications or lactation contraindications; 2) preterm infants or twins were excluded from the study. Ten women with incomplete information and 43 women in whom the genotype could not be detected because of the limited amount of breast milk provided were excluded from the study. Trained researchers interviewed the subjects to investigate their basic information, including age, pre-pregnancy weight, height, and health-related behavioral habits. Furthermore, we investigated the infant anthropometric measurements at birth, including birthweight, length, sex and gestational age. All participants signed an informed consent form and agreed to provide detailed basic dietary information.
Information on habitual food intake was obtained using the 24-h recall method. A questionnaire was administered to conduct analyses. Fatty acid intake data were calculated as mean values from a 3-day food record (before collecting breast milk) through the 24-h recall method. This was done by using the database of computerized food codes based on Chinese food composition tables.
Breast milk was collected from 8:00 a.m. to 11:00 a.m. between the 22nd and 25th postpartum days using an electric milk pump to extract all the milk from the unilateral breast. Then collected milk was placed in a 20 mL centrifuge tube, transported to the laboratory at low temperature, and stored at –80 °C for the detection of PUFAs and genomic DNA in the milk.
The internal standard method was used to calculate the fatty acid methyl esters (FAMEs). Eight PUFAs were detected in breast milk using the Japanese Shimazzu gas chromatograph (GC-14C, SHIMADZU Corp.) equipped with a capillary column (SP-2560, Supelco, Bellefonte, PA, USA; 100 m × 0.25 mm × 0.20 μm). C17:0 was used as an internal standard, and specific experimental parameters were set by gas chromatography. Thereafter, FAME was obtained by diluting the FAME mixed standard solution with different concentration gradients. The sample entry numbers for each standard concentration were recorded. After detecting the mixed standards at each concentration, the peak area of each FAME at different concentrations was recorded. A FAME standard curve was obtained using the ratio of FAME at each concentration as an ordinate. After the breast milk PUFAs test, the peak areas of the eight methyl PUFAs in the breast milk samples were determined by comparing the retention time ratio with that of the mixed standard, and the peak area/internal standard ratio of each breast milk sample was introduced into the obtained regression equation. Specific experimental steps have been described in a previous study[5].
Two SNPs closely related to plasma fatty acid levels were identified using the NCBI SNP database (dbSNP Build 126; http://www.ncbi.nlm. nih.gov/). The participants were genotyped for two ADIPOQ cluster variants, including two SNPs (rs1501299 and rs1063538). The milk samples were thawed at 4 °C and the fat layer, skim milk layer, and cellular layer were separated[6]. Genomic DNA was extracted from 300 μL of the cell layer using a DNA kit (Beijing, TIANGEN) according to the manufacturer's instructions. Genotyping was performed using the Sequenom Mass Array system (BOMIAO Biological Technological Company, Beijing, China).
Participants were separated according to the Chinese BMI criteria as follows: underweight (BMI < 18.5 kg/m2), normal weight (BMI 18.5–23.9 kg/m2), or overweight (BMI ≥ 24.0 kg/m2). Continuous basic information variants among the groups were tested using one-way ANOVA. All statistical analyses were performed using SPSS version 26.0 (SPSS Corp., Armonk, IL, 100 USA). Categorical variables were tested using the chi-square test.
The normal distribution of fatty acids was tested using the Kolmogorov-Smirnov test and distribution plots. The skewed measurements of γ-linolenic acid (GLA, C18:3n-6), Dihomo-γ-linolenic acid (DGLA, C20:3n-6), EPA, and DHA concentrations were expressed as square roots to obtain a normal distribution. Spearman’s correlation coefficients were used to examine the relationship between dietary fatty acid intake and the fatty acid content of breast milk. Hardy-Weinberg equilibrium was tested using the chi-square goodness of fit test for each SNP locus. M and m represent the major and minor alleles, respectively, with three genotypes: MM, Mm, and mm. The BMI × genotype interactions were tested for breast milk PUFAs and investigated using a univariate general linear model. In this model, breast milk PUFAs were the dependent variable. BMI and genotype were the fixed factors. BMI and genotype were also tested individually with the general linear model. Dietary fatty acids correlated with breast milk fatty acid concentrations, and maternal age was adjusted for in the model. Data are presented as means ± SD. Statistical significance was set atP < 0.05. Data were displayed using Origin version 2021.
The demographic and clinical information of the mother-infant dyads is presented in Table 1. The participants were classified into three groups according to their maternal BMI. As expected, infants in the overweight (OW) group had significantly higher birth weights than those in the other two groups. In addition to birth weight, infant birth length in the OW group was significantly longer than that in the under weight (UW) and normal weight (NW) groups. Meanwhile, mothers’ ages differed among the groups.
Characteristics Maternal BMI P UW
(n = 96)NW
(n = 256)OW
(n = 61)Mothers Age (year) 28.97 ± 2.94 30.74 ± 3.58 30.75 ± 3.45 < 0.001 Prepregnancy BMI (kg/m2) 17.43 ± 0.94 20.83 ± 1.50 27.18 ± 2.90 < 0.001 Delivery patterns, n (%) Vaginal delivery 41 (9.93) 70 (16.95) 16 (3.87) 0.012 Cesarean 54 (13.08) 186 (45.04) 45 (10.90) Missing value 1 (0.24) − − Infants Sex, n (%) Male 39 (9.44) 136 (32.93) 34 (8.23) 0.073 Female 57 (13.80) 119 (28.81) 27 (6.54) Gestational age (week) 39.31 ± 1.10 39.22 ± 1.03 39.18 ± 1.00 0.678 Birthweight (kg) 3.28 ± 0.39 3.49 ± 0.53 3.69 ± 0.86 < 0.001 Birth length (cm) 50.24 ± 1.70 50.44 ± 1.17 50.94 ± 1.45 0.016 Note. Continuous data are presented as means ± SD; categorical variables are presented as a percentage; BMI, body mass index; UW, underweight; NW, normal weight; OW, overweight; −, no content. Table 1. Demographic and clinical characteristics of mothers and infants
The characteristics of the detected SNPs, including their positions on chromosome 3 and their genotypes, are summarized in Table 2. Minor allele frequencies ranged from 27.1% to 47.7%. The genotype distributions for each SNP were consistent with Hardy-Weinberg equilibrium (P > 0.05).
SNP Allele M/m Genotype (n) MAF, % P M/M M/m m/m rs1501299 G/T 220 159 32 27.1 0.710 rs1063538 T/C 110 185 92 47.7 0.420 Note. MAF, minor allele frequency; M/M, major/major allele; M/m, major/minor allele; m/m; minor/minor allele; SNP, single nucleotide polymorphism. Table 2. Characteristics of the selected SNPs
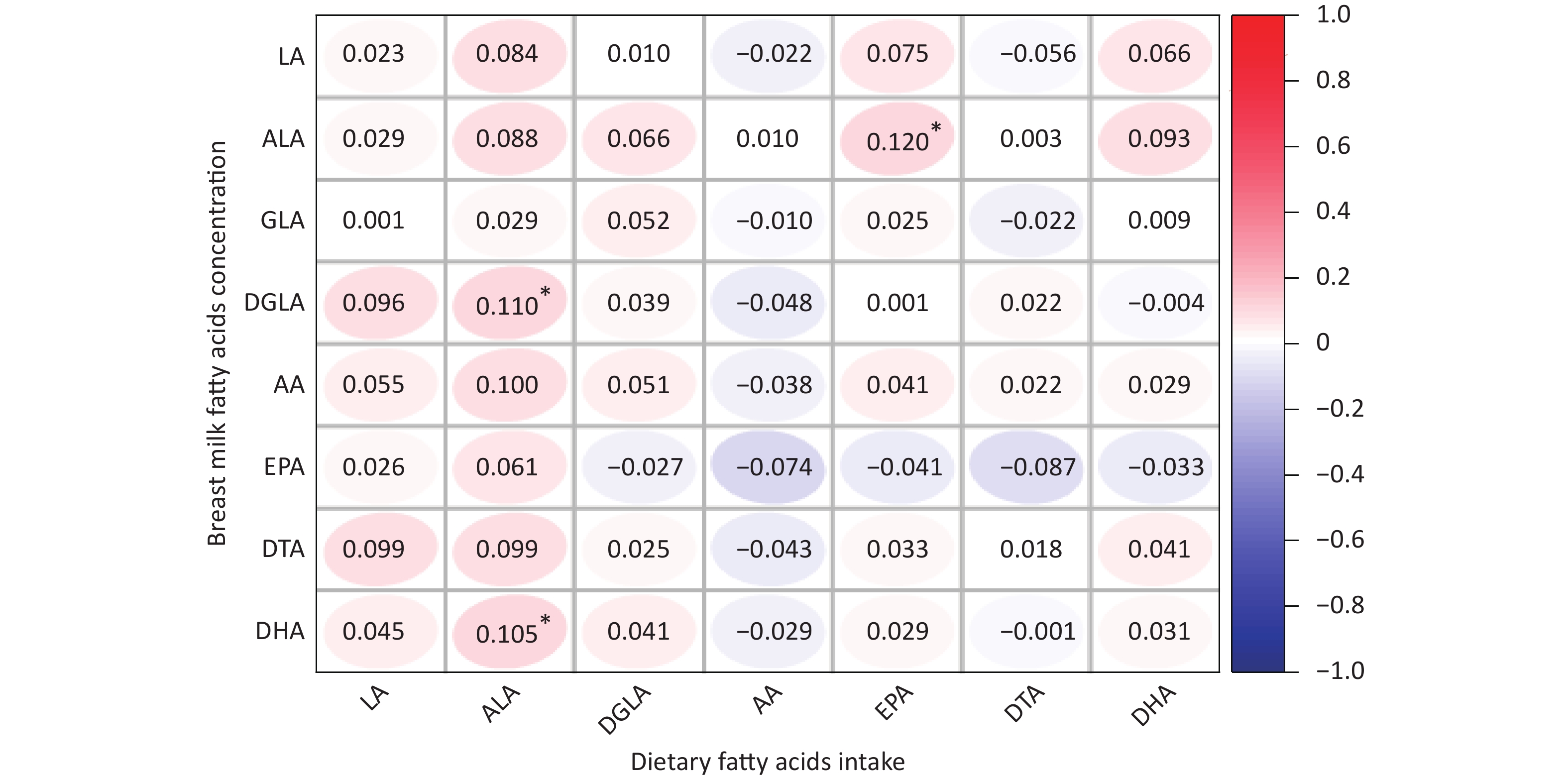
The average concentrations of PUFAs in breast milk among the genotypes are summarized in Supplementary Table S1, (available in www.besjournal.com). However, we did not observe any differences in breast milk PUFAs among the different genotypes. The correlation between the dietary fatty acid intake of lactating mothers and the fatty acid content of their breast milk is shown in Supplementary Figure S1, (available in www.besjournal.com). The fatty acid contents of breast milk, LA, GLA, AA, EPA, and DTA, were not related to dietary fatty acid intake. However, ALA, DGLA, and DHA contents in breast milk were positively correlated with dietary fatty acid intake.
SNP LA GLAa DGLAa AA DTA ALA EPAa DHAa rs1501299 G/G 0.37 ± 0.20 0.19 ± 0.06 0.22 ± 0.06 0.08 ± 0.04 0.02 ± 0.01 0.15 ± 0.09 0.09 ± 0.03 0.22 ± 0.08 G/T 0.37 ± 0.17 0.20 ± 0.06 0.23 ± 0.07 0.08 ± 0.04 0.02 ± 0.01 0.14 ± 0.07 0.09 ± 0.03 0.22 ± 0.06 T/T 0.38 ± 0.16 0.20 ± 0.05 0.23 ± 0.06 0.09 ± 0.03 0.02 ± 0.01 0.15 ± 0.06 0.09 ± 0.03 0.22 ± 0.06 P 0.913 0.801 0.398 0.685 0.998 0.670 0.976 0.812 rs1063538 T/T 0.38 ± 0.18 0.20 ± 0.06 0.23 ± 0.06 0.08 ± 0.04 0.02 ± 0.01 0.15 ± 0.07 0.09 ± 0.03 0.22 ± 0.07 T/C 0.37 ± 0.18 0.20 ± 0.06 0.22 ± 0.06 0.08 ± 0.04 0.02 ± 0.01 0.14 ± 0.08 0.09 ± 0.03 0.22 ± 0.06 C/C 0.35 ± 0.20 0.19 ± 0.06 0.22 ± 0.07 0.08 ± 0.04 0.02 ± 0.01 0.15 ± 0.08 0.09 ± 0.03 0.22 ± 0.08 P 0.619 0.757 0.578 0.789 0.863 0.944 0.399 0.889 Note. LA, Linoleic acid; GLA, γ-Linoleic acid; DGLA, Dihomo-γ-linolenic acid; AA, Arachidonic acid; DTA, Docosatetraenoic acid; ALA, α-linolenic acid; EPA, Eicosapentaenoic acid; DHA, Docosahexenoic acid. SNP, Single nucleotide polymorphism. aThe data were normalized by square root transformation. Table S1. Relationship between the selected SNPs and the concentrations of PUFAs in breast milk (g/100 g, mean ± SD)
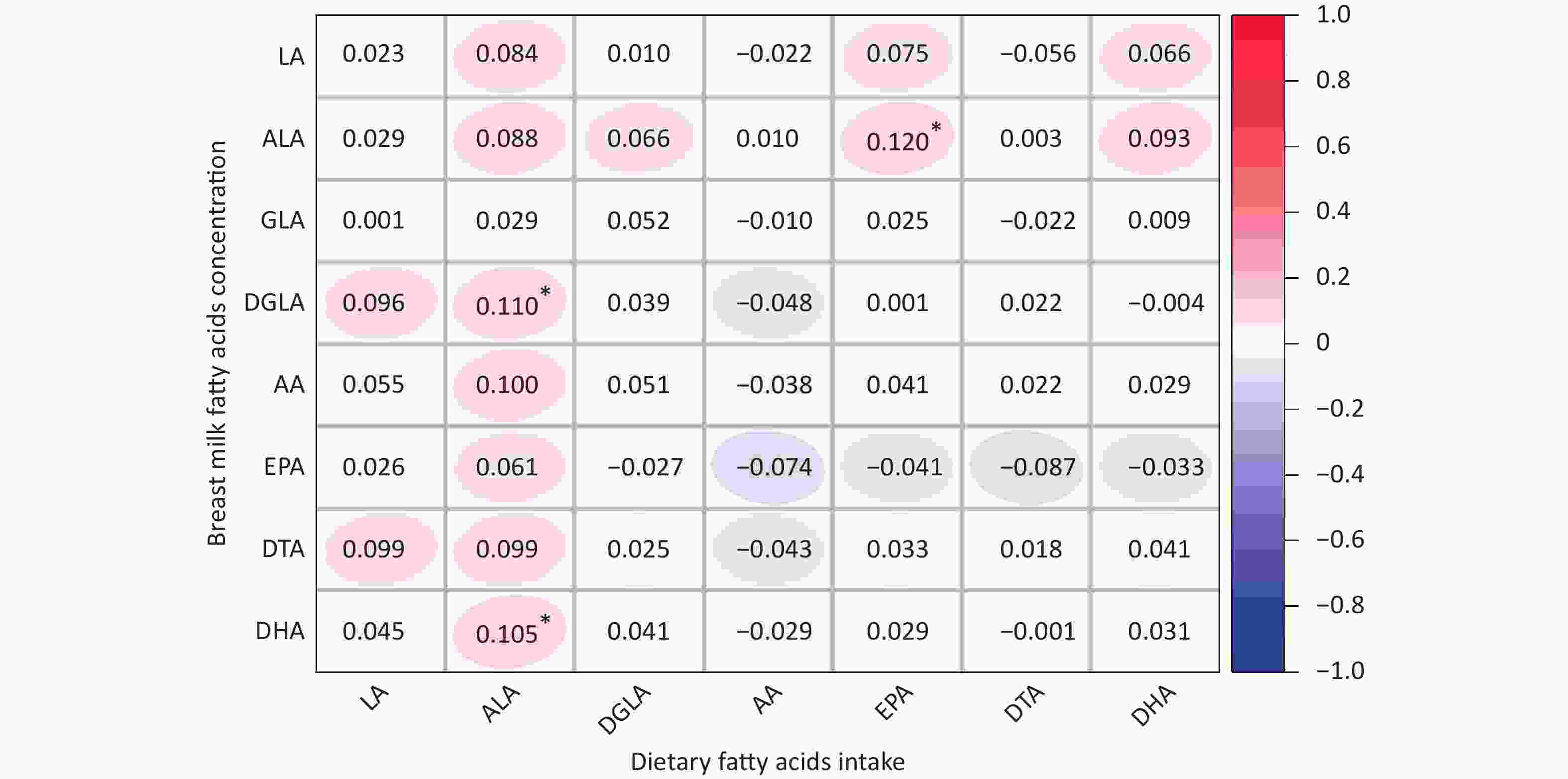
Figure S1. Correlations between dietary fatty acids intake and fatty acids content of breast milk. LA, Linoleic acid; GLA, γ- Linoleic acid; DGLA, Dihomo-γ-linolenic acid; AA, Arachidonic acid; DTA, Docosatetraenoic acid; ALA, α-linolenic acid; EPA, Eicosapentaenoic acid; DHA, Docosahexenoic acid. *P < 0.05.
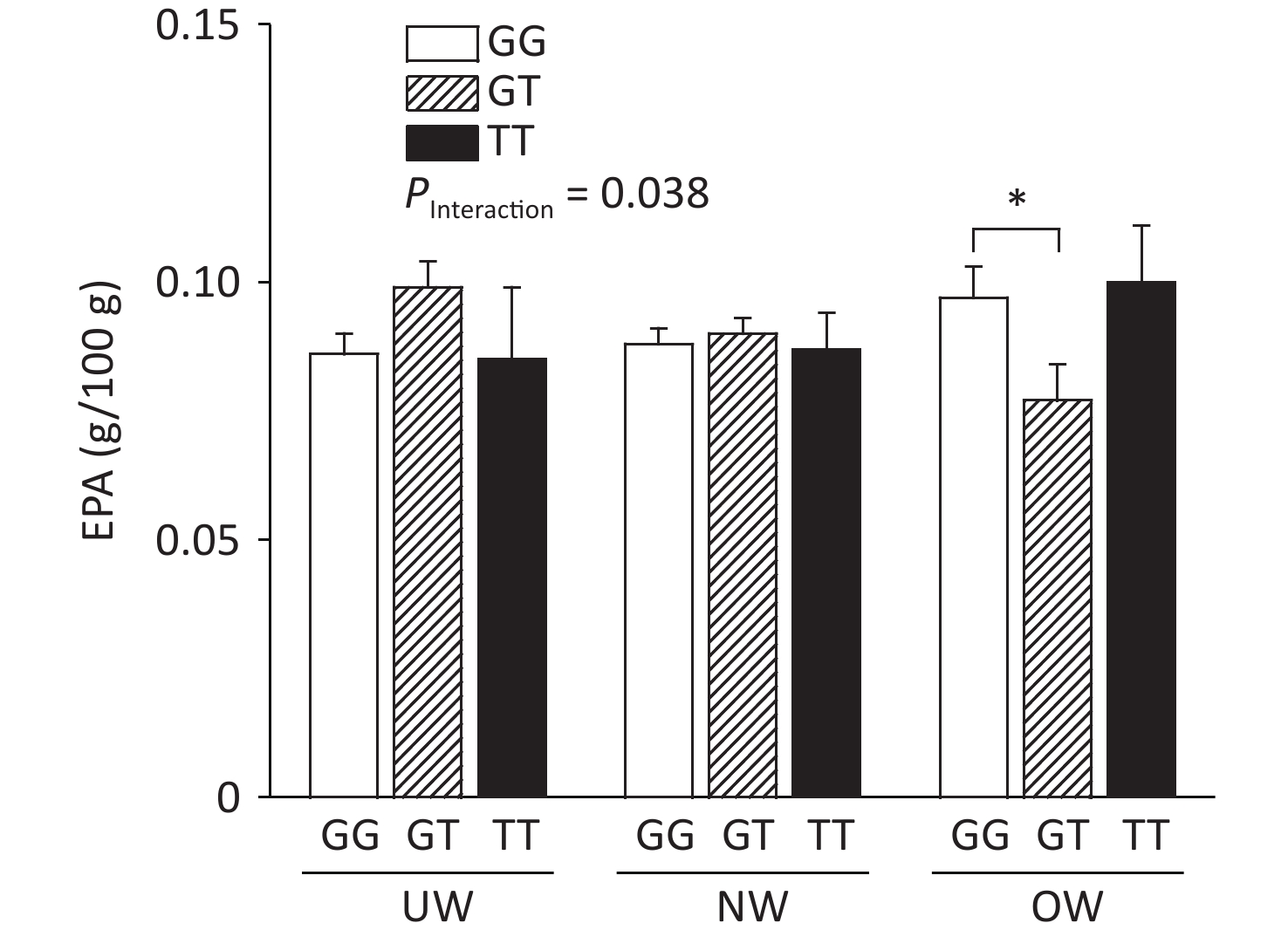
The relationship between ADIPOQ SNPs and breast milk PUFA concentrations according to BMI is shown in Figure 1, where rs1501299 interacted with BMI to influence EPA concentrations in breast milk (adjusted by age). The concentration of breast milk EPA in G/T genotype mothers was significantly lower than that in G/G genotype mothers (0.08 ± 0.01 vs. 0.10 ± 0.01, P = 0.020). The lower concentration of breast milk EPA in rs1501299 G/T overweight individuals might reflect a lower EPA reserve in these participants, as there were no differences in their dietary EPA intake in the baseline analysis. Consistent with our findings, Elvira et al.[7] reported that the EPA levels in the plasma of G/T carriers were lower than those in G/G homozygotes. Interestingly, this phenomenon was only observed in the OW group. This suggests that a lower EPA level in mature milk is associated with pre-pregnancy obesity. This suggests that the rs1501299 SNP is closely associated with BMI.
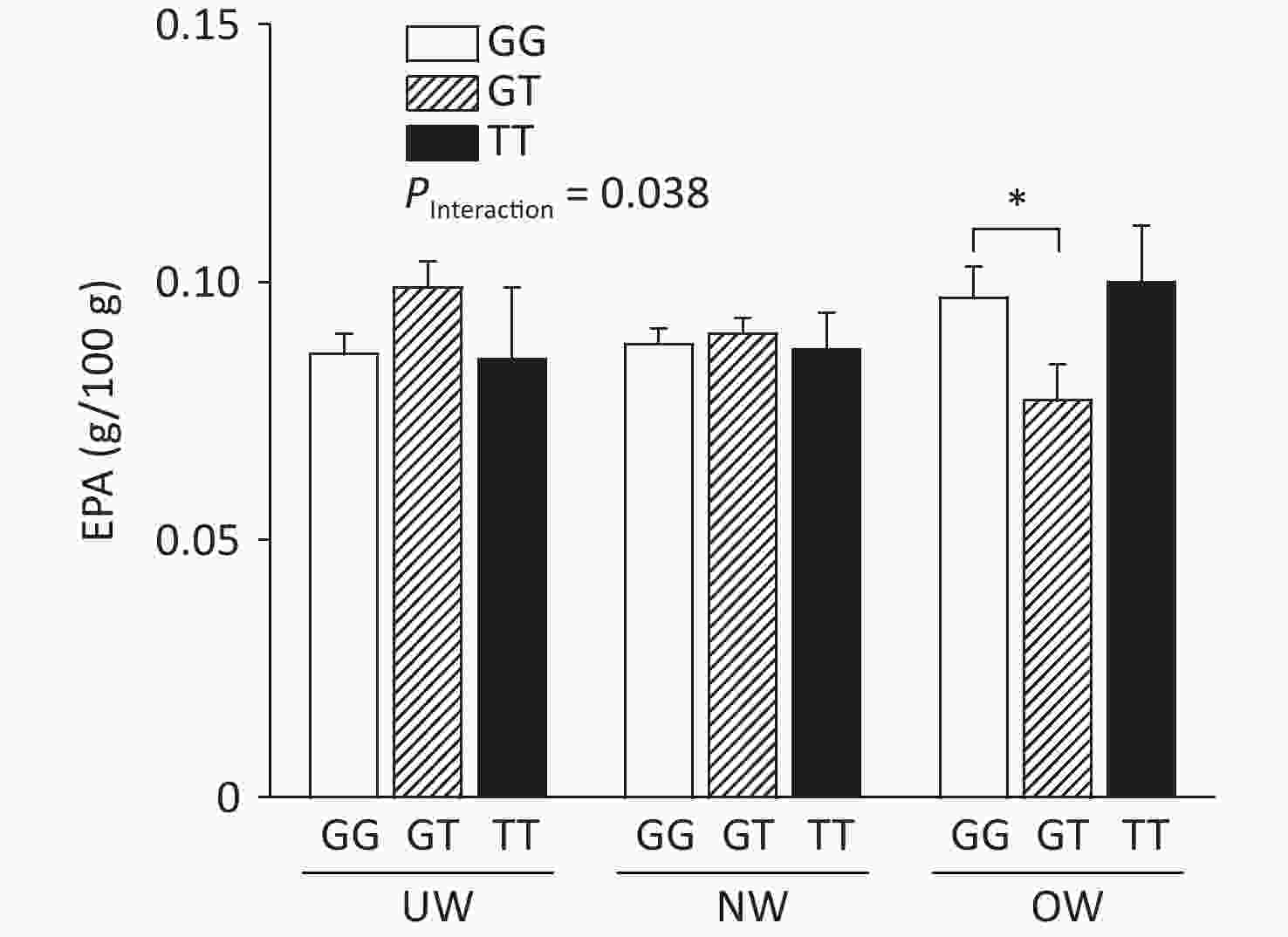
Figure 1. Breast milk EPA level according to BMI and ADIPOQ SNP rs1501299 genotype adjusted by age. EPA, Eicosapentanoic acid; BMI, Body mass index; UW, Underweight; NW, Normal weight; OW, Overweight. *P = 0.020 between OW group GG and GT for EPA (0.10 ± 0.01 vs. 0.08 ± 0.01).
In our study, the concentration of EPA was decreased, whereas BMI increased in participants with the G/T genotype. One potential explanation for this reduction is that compared with normal weight participants, overweight participants had lower concentrations of adiponectin[8]. This effect could have been exacerbated in G/T subjects and contributed to the decrease in EPA levels in breast milk among overweight participants. The main biological function of adiponectin is dependent on the activation of AMP-activated protein kinase in skeletal muscle, which increases fatty acid oxidation and glucose uptake. This study hypothesized that serum phospholipid n-3 PUFAs affect the concentration of peripheral blood adiponectin. In our study, lower breast milk EPA levels were observed in overweight G/T carriers.
EPA plays a role in brain metabolism, where it crosses the blood-brain barrier through lipoproteins, may be consumed by rapid-oxidation in the mitochondrial matrix, and provides energy for neural activity. This suggests that mothers with low EPA levels should supplement their diet with foods rich in n-3 PUFAs, such as seafood, as studies have shown that the amount of n-3 PUFAs in breast milk is easily influenced by diet.
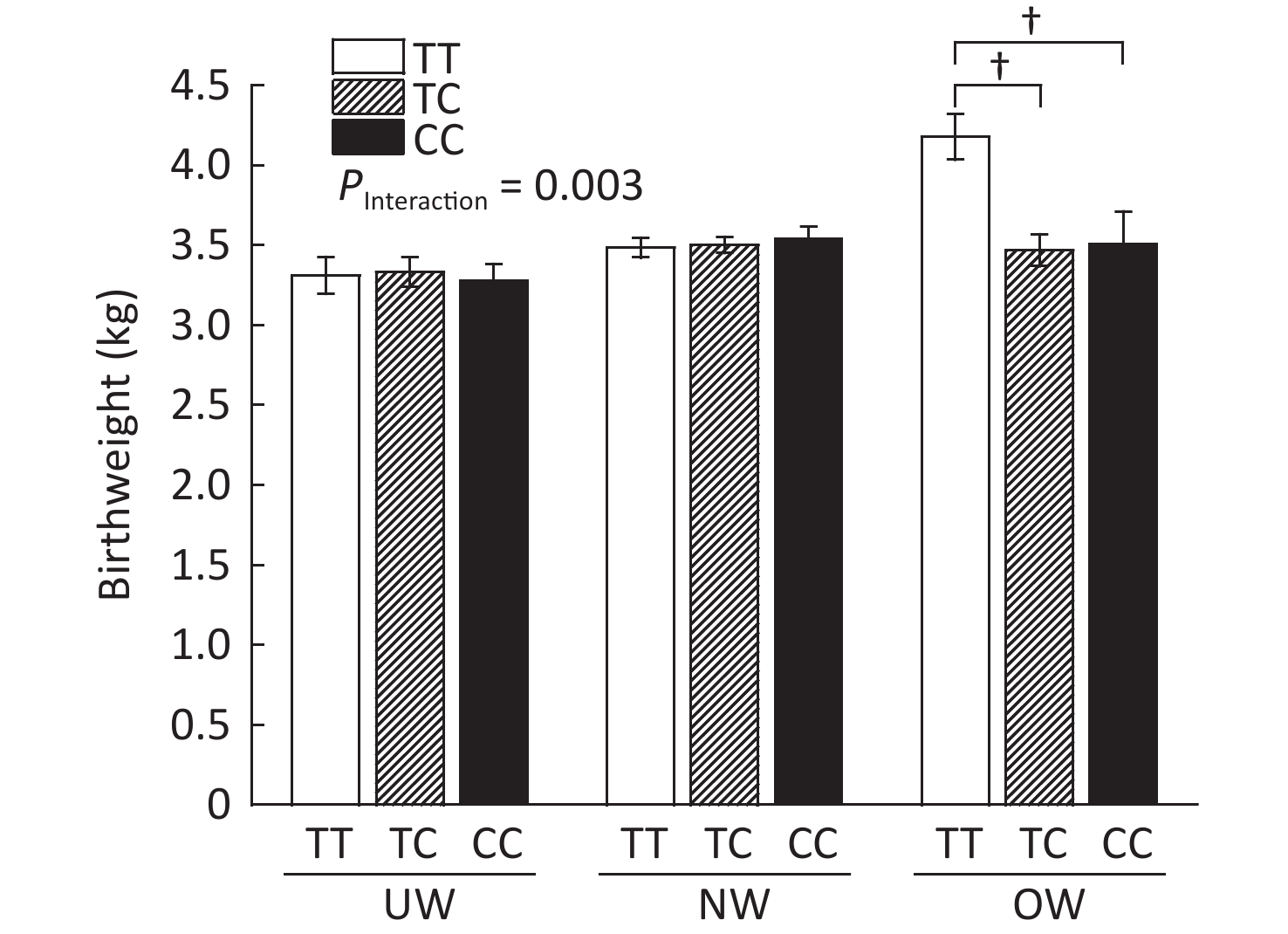
As shown in Supplementary Figure S2, (available in www.besjournal.com), rs1063538 interacted with BMI to influence infant birthweight. Infants born to mothers with the T/T genotype were significantly heavier than the infants born to mothers with minor genotypes (4.18 ± 0.14 vs. 3.47 ± 0.10, P < 0.001; 4.18 ± 0.14 vs. 3.51 ± 0.20, P < 0.001). The SNP rs1063538 is located in the 3'UTR region of ADIPOQ, and previous studies have shown that polymorphisms in the 3'UTR have a significant effect on the level of adiponectin. For example, in genome-wide linkage and association scan studies on adiponectin levels, rs6773957 and rs3774261 were found to be the sites most strongly associated with adiponectin levels[9], and they were both within the 3'UTR region of the gene, where rs1063538 was also located. Given the effect of adiponectin on obesity, and the role of 3'UTR in regulating gene expression, we reasoned that the SNPs of ADIPOQ 3'UTR may perform a function in infant weight at birth.
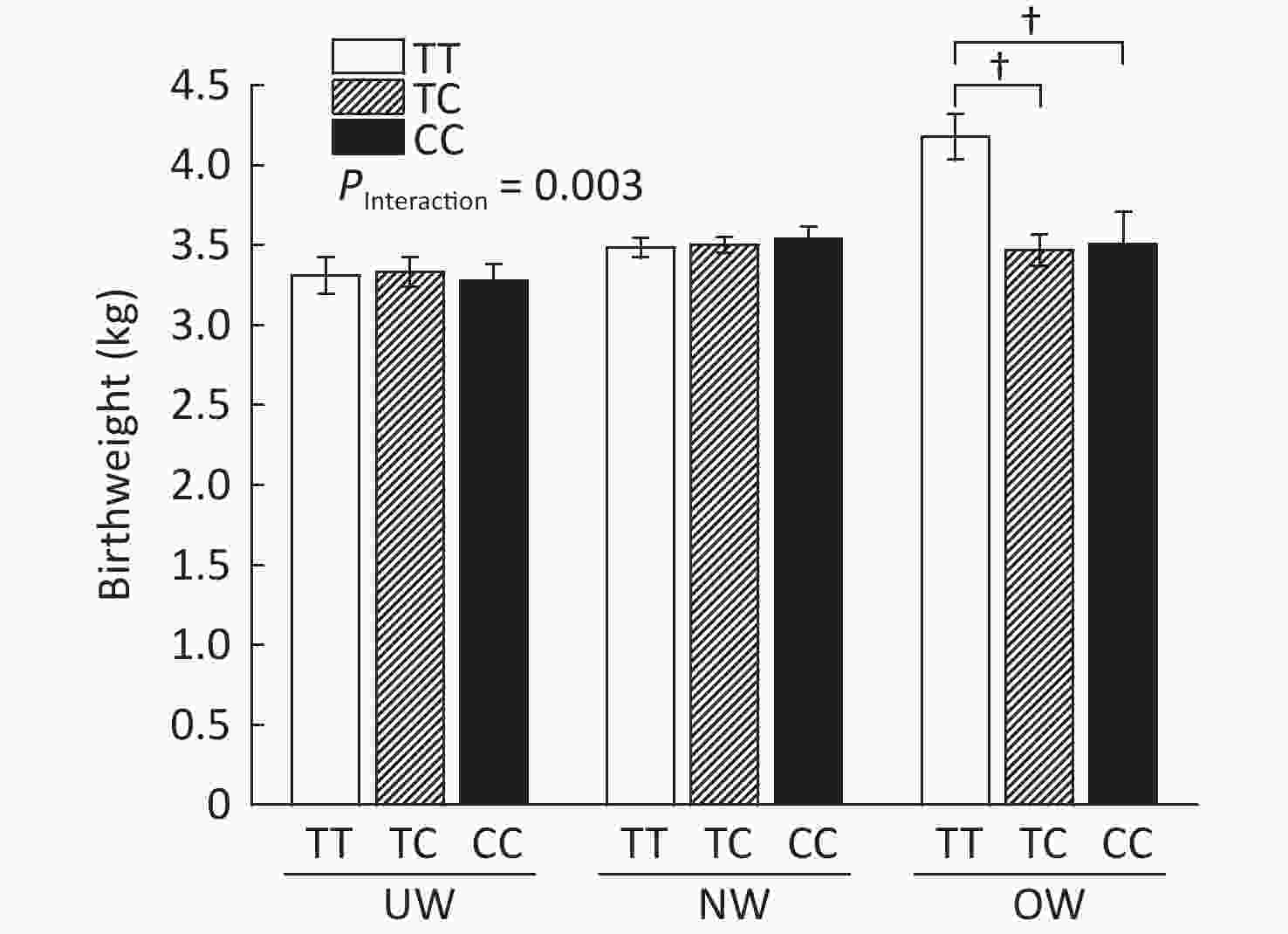
Figure S2. Infant birthweight according to BMI and ADIPOQ SNP rs1063538 genotype adjusted by age. BMI, Body mass index; UW, Underweight; NW, Normal weight; OW, Overweight. †P < 0.001 between OW group T/T and TC/CC for birthweight (4.18 ± 0.14 vs. 3.47 ± 0.10; 4.18 ± 0.14 vs. 3.51 ± 0.20).
Our study has the limitations of being cross-sectional and observational in nature; therefore, the causal relationships and mechanistic interactions among ADIPOQ genetic variation, maternal BMI, milk fatty acids, and infant weight cannot be easily established. In addition, we did not measure maternal adiponectin concentration. In future, we will further explore the influence of ADIPOQ on breast milk adiponectin levels. Despite these limitations, we found that ADIPOQ gene-environment interactions were associated with reduced EPA concentrations in mature milk and heavier birthweight in neonates. These findings may help guide the diet of lactating mothers more specifically. BMI-related guidance can also be provided to women preparing for pregnancy to ensure healthy development of infants both in utero and during lactation.
HTML
 23024+Supplementary Materials.pdf
23024+Supplementary Materials.pdf
|

|







 Quick Links
Quick Links
 DownLoad:
DownLoad: