-
Group B streptococcus (GBS) is a Gram-positive, encapsulated bacterium that belongs to the group of pyogenic streptococci, and it is an asymptomatic colonizer of the digestive and genitourinary tracts of healthy human adults. However, it can cause severe invasive infections in neonates and immunocompromised adults. In the 1960s, GBS was identified as a leading cause of life-threatening neonatal infections[1,2].
In neonatology, there are two distinguishable clinical syndromes: early onset disease (EOD), in which a GBS infection occurs within the first week of life (particularly within the first 24 h), and late-onset disease (LOD), in which GBS infection occurs after the baby is 7 days old (7–90 days postpartum). EOD is caused by vertical transmission through either an ascending infection from the genital tract or during labor and birth. Numerous studies have shown that up to 30% of pregnant women worldwide are colonized with GBS, and that vertical transmission occurs in approximately 50% of colonized mothers. Approximately 1% of colonized newborns develop EOD, which may be connected to ruptured membranes, because fetal infection can occur only when exposed to GBS. Bacteremia without a particular focus was the most common clinical syndrome, followed by pneumonia and meningitis. Currently, the case fatality rate of EOD is estimated to still be 2%–10%, and fatal outcomes are more frequent among premature neonates[3].
Most cases of EOD occur due to the contact of the neonate with GBS during delivery. Therefore, intrapartum antibiotic prophylaxis (IAP) administered to GBS carriers is useful for preventing vertical transmission in the vast majority of cases, and its widespread use has resulted in a significant reduction in the incidence of EOD. LOD, however, is most likely acquired from breast milk or from nosocomial or community sources. Prematurity is the main risk factor for the development of LOD, and bacteremia without a particular focus of infection is the most common presentation. The mortality rate of LOD is low; however, meningitis and subsequent sequelae are more frequently associated with LOD[4].
GBS has also been known to cause significant maternal morbidities, including endometritis, chorioamnionitis, bacteremia, and postpartum wound infections. GBS tract infections can cause miscarriages, preterm births, and low birth weights in newborns. Although GBS seldom causes diseases in healthy adults, it is responsible for serious infections in patients with diabetes, elderly individuals, residents of nursing homes, and immunocompromised patients. The successful administration of IAP and the treatment of severe GBS infections rely on the efficient and reliable detection of GBS in clinical samples[5].
Recently, there has been a rapid increase in the availability of chromogenic media for GBS detection. These contain enzyme substrates linked to indoxyl chromogens, and the target microorganisms are characterized by specific enzyme systems that metabolize the substrate, resulting in release of a chromogen. Subsequently, indigoid dye forms upon the oxidation and dimerization of the indoxyl molecules in the presence of oxygen precipitates within the colonies, leading to typical brightly contrasting colors[6]. Therefore, the contrast amplitude is proportional to the GBS concentration. However, traditional visual identification usually requires at least 48 h in order to determine whether GBS colonizes; thus, rapid and highly sensitive detection of GBS is urgently needed.
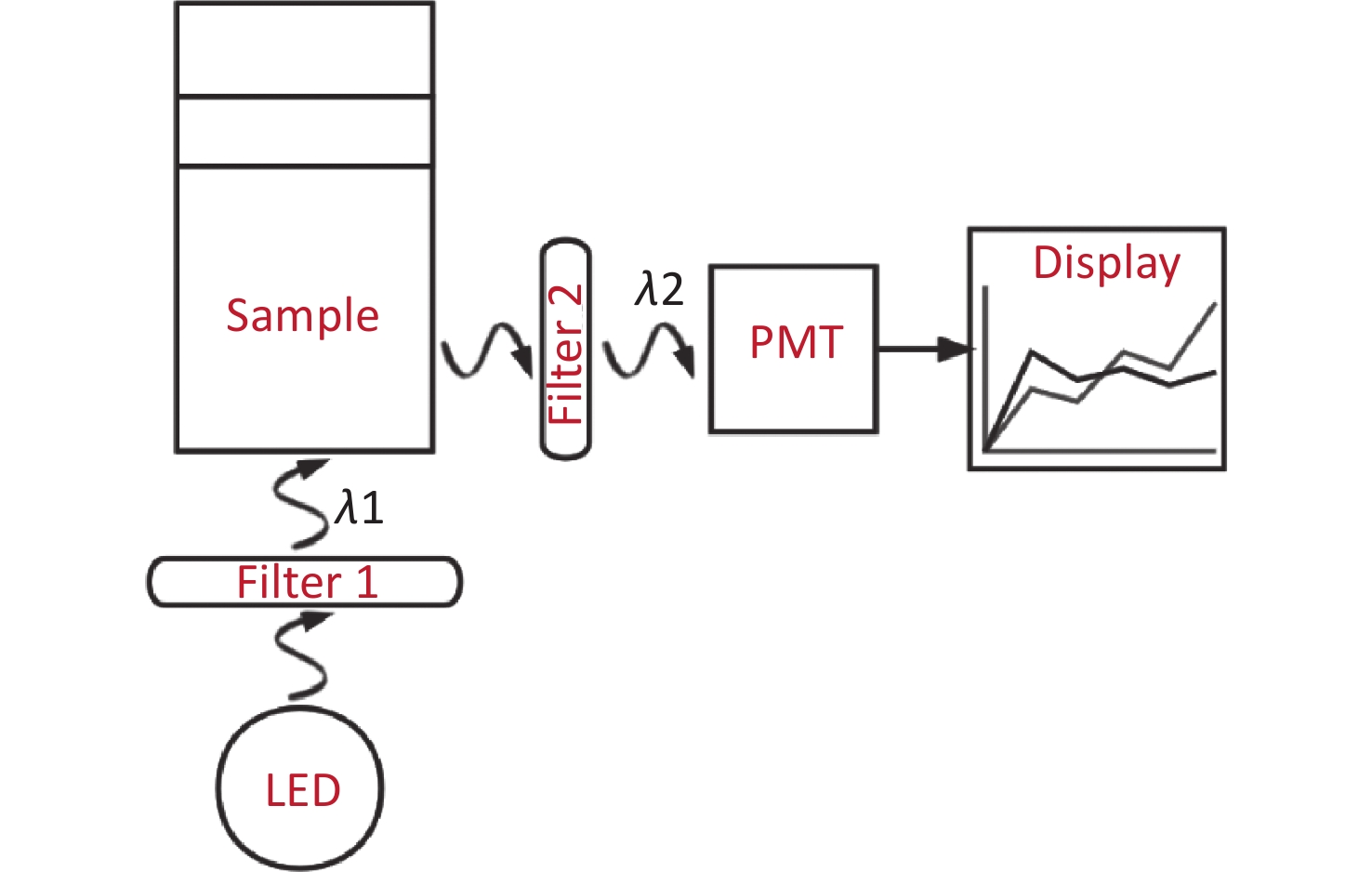
With the emergence of photocatalytic nanoparticles[7], quantitative detection technologies are being developed rapidly. In this study, we developed a rapid trace analysis of GBS [a point-of-care testing (POCT)[8] as shown in Figure 1] based on chromogenic media, which meets clinical needs for highly sensitive and rapid identification.
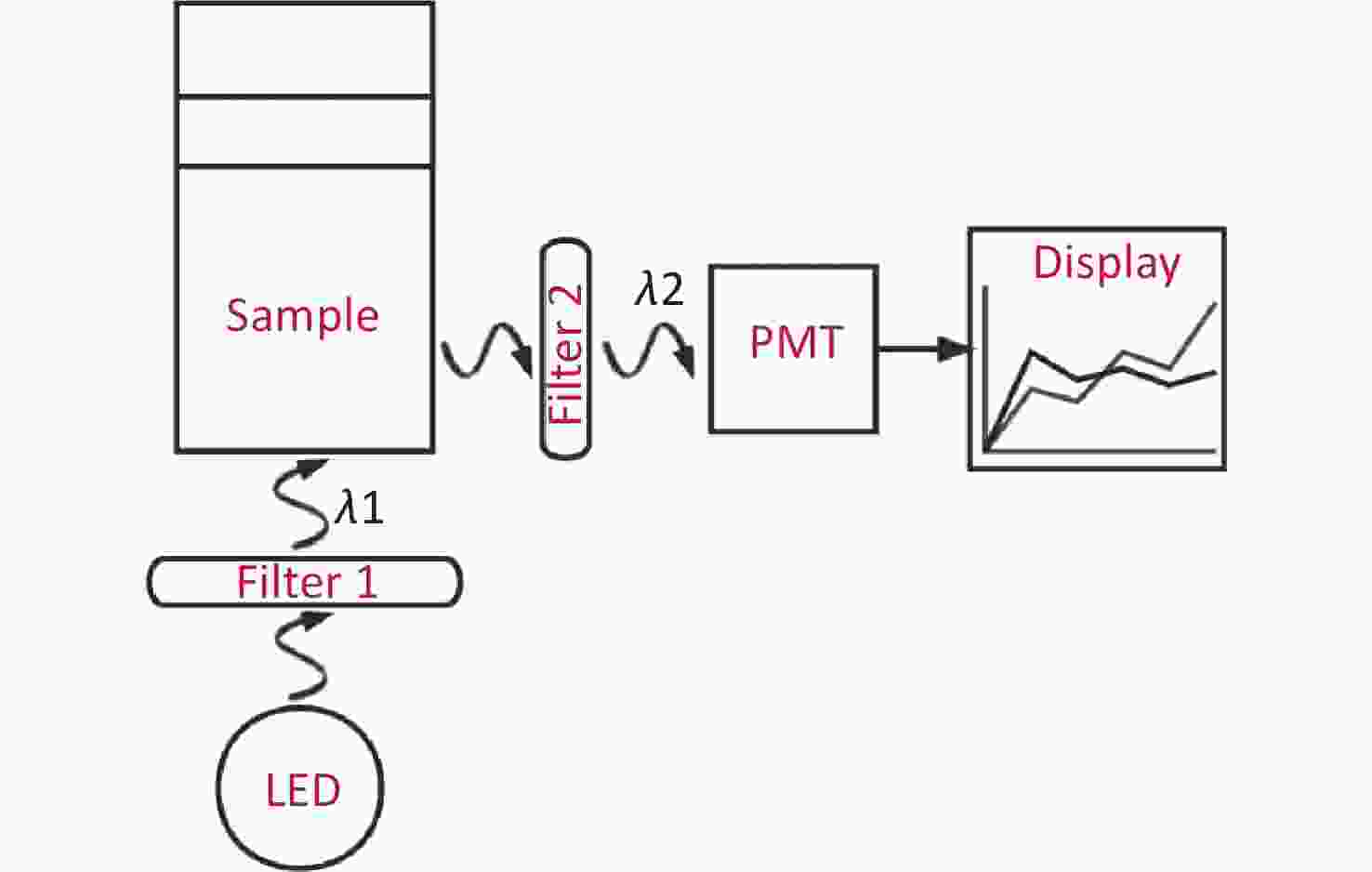
Figure 1. The mechanism of the POCT (a rapid trace analysis with chromogenic media). The use of an LED is the source of excited light. A parallel excitation light with a wavelength of λ1 is filtrated from filter 1, and then the incident light from the sample is perpendicular to the bottom of tube. As a result, the irradiated sample emits another light (λ2), which is the so-called emission light, however, a certain amount of the light of λ2 will be absorbed by indoxyl chromogen, and the rest of the emission light will be finally detected by PMT through filter 2. The absolute value of the progress time is directly displayed in Figure 2.
Two tubes of culture media with 1.2 mL were supplied by Wenzhou Beikete Medical Equipment Co., Ltd. One contained indoxyl chromogens with a higher concentration and was labeled “5”, another was without any chromogens and labeled “0”. Figure 2 shows the samples of chromogenic media with different concentrations of the chromogen mixture and the corresponding measured absolute light intensity, which was prepared and measured simultaneously as follows:
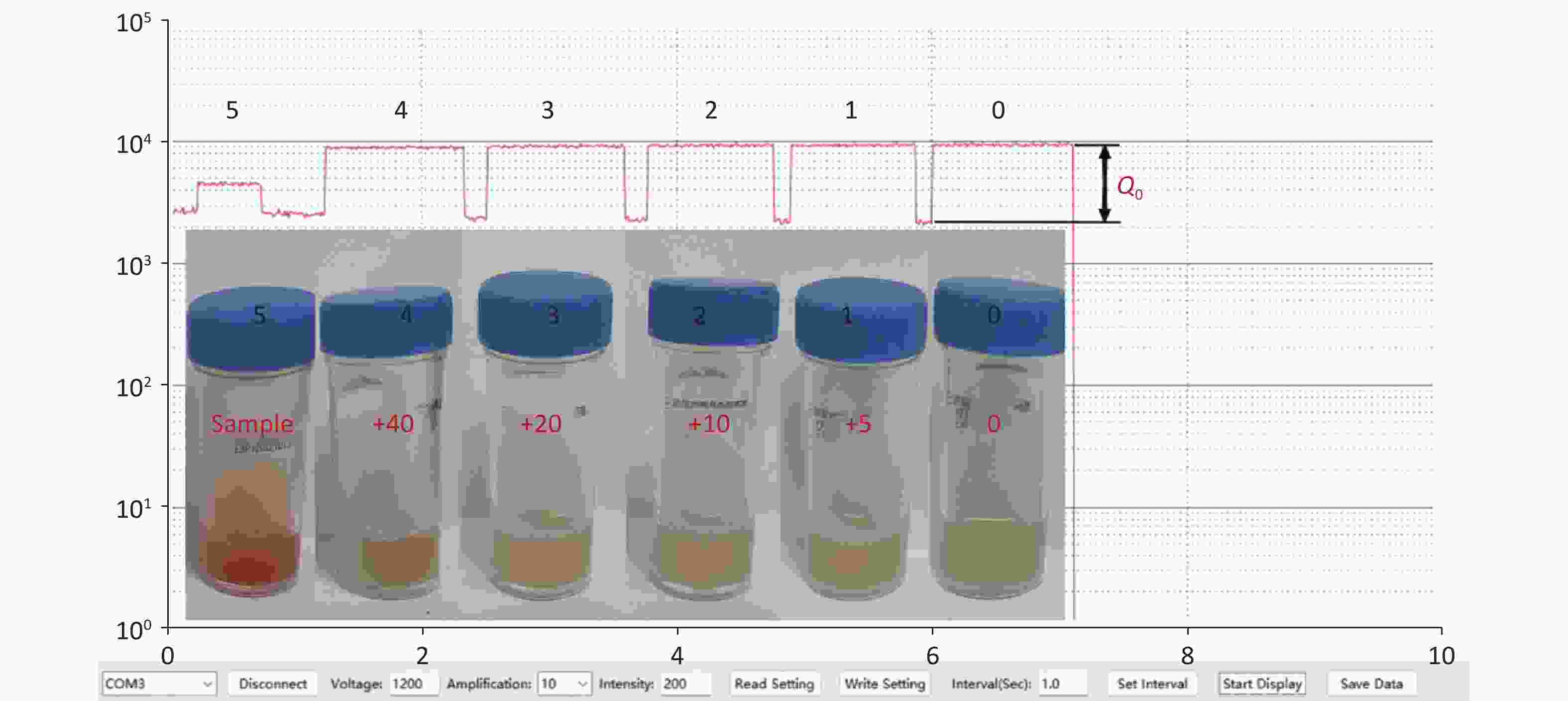
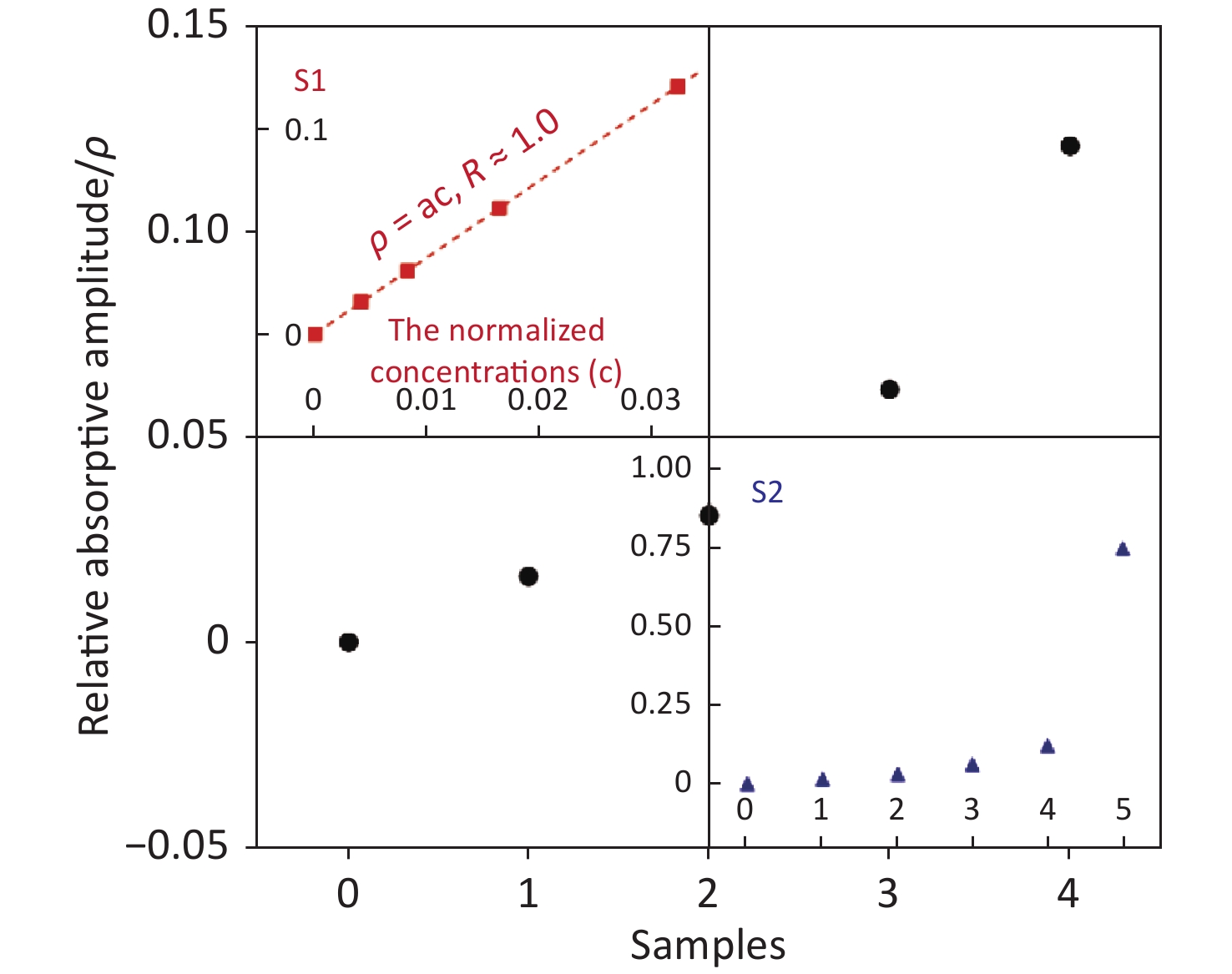
Figure 2. The measured absolute light intensity of the samples by the POCT used vs. time. Each higher platform from right to left corresponds to the value of sample 0, 1, 2, 3, 4 and 5 respectively, while the lower platform is the background value. This means that the absolute light intensity is the higher one minus the lower one. The relative absorptive amplitude of sample i, ρi, can be defined by Equation 1, and shown in Figure 3. The value of samples 1–4 is clearly higher than ''0'', which means the method has excellent sensitivity. Insert: Analysis samples of chromogenic media were mixed with different volume of sample ''5''. The corresponding injected volumes of the mixture of chromogens were 0, 5, 10, 20, 40 µL respectively from right to left as described in text, and the samples were labeled 0, 1, 2, 3, 4 correspondingly. It is very difficult using the traditional visual identity method to determine whether GBS colonized in samples 1–4.
Firstly, we measured the value of the emission light from tube ''0'', and the platform ''0'' in Figure 2 is its time process;
Secondly, a 5 µL sample of ''5'' was injected in tube ''0'', the new sample was labeled ''1'', and the measured value is shown in platform ''1'' in Figure 2;
Thirdly, another 5 µL sample of ''5'' was injected in tube ''1'', the new sample was labeled ''2'', and the measured value is shown in platform ''2'' in Figure 2;
Fourthly, a 10 µL sample of ''5'' was injected in tube ''2'', the new sample was labeled ''3'', and the measured value is shown in platform ''3'' in Figure 2;
Fifthly, a 20 µL sample of ''5'' was injected in tube ''3'', the new sample was labeled ''4'', and the measured value is shown in platform ''4'' in Figure 2;
Finally, we measured the value of the emission light from tube ''5'', and the platform ''5'' in Figure 2 was its process time.
The normalized concentrations of chromogens of sample ''0–5'' (with respect to the sample ''5''), c, are approximately 0, 4.15 × 10−3, 8.26 × 10−3, 1.64 × 10−2, 3.23 × 10−2, and 1.00 respectively.
The absolute intensity of the remaining emission light (Qi) was found to be equal to the value of the higher platform minus the background noise (corresponding to the value of the lower platform). Thus, the absorptive amplitude of the sample is Q0−Qi, where Q0 is the value at which there are no indoxyl chromogens in chromogenic media, such as sample 0.
The relative absorptive amplitude of sample i, ρi, can be defined by
$$ {\rho }_{i}\equiv 1-\frac{{Q}_{i}}{{Q}_{0}} , $$ (1) and this is shown in Figure 3. The relationship between the relative absorptive amplitude (ρ) and the normalized concentrations of samples (c) was almost linear [ρ = ac, where a (=3.75) is the fitted constant] at lower concentrations, with a regression coefficient (R) of 1.0. The value of samples 1–4 is clearly higher than "ρ0 (=0)", which means that the sensitivity of our method is much higher than that of the traditional one as shown in Figure 2. The enormous quantitative difference between samples 0 and 5 further indicates the advancement made in regard to the trace analysis of GBS. It is possible for chromogenic media to significantly shorten the identification time if the POCT is loaded.
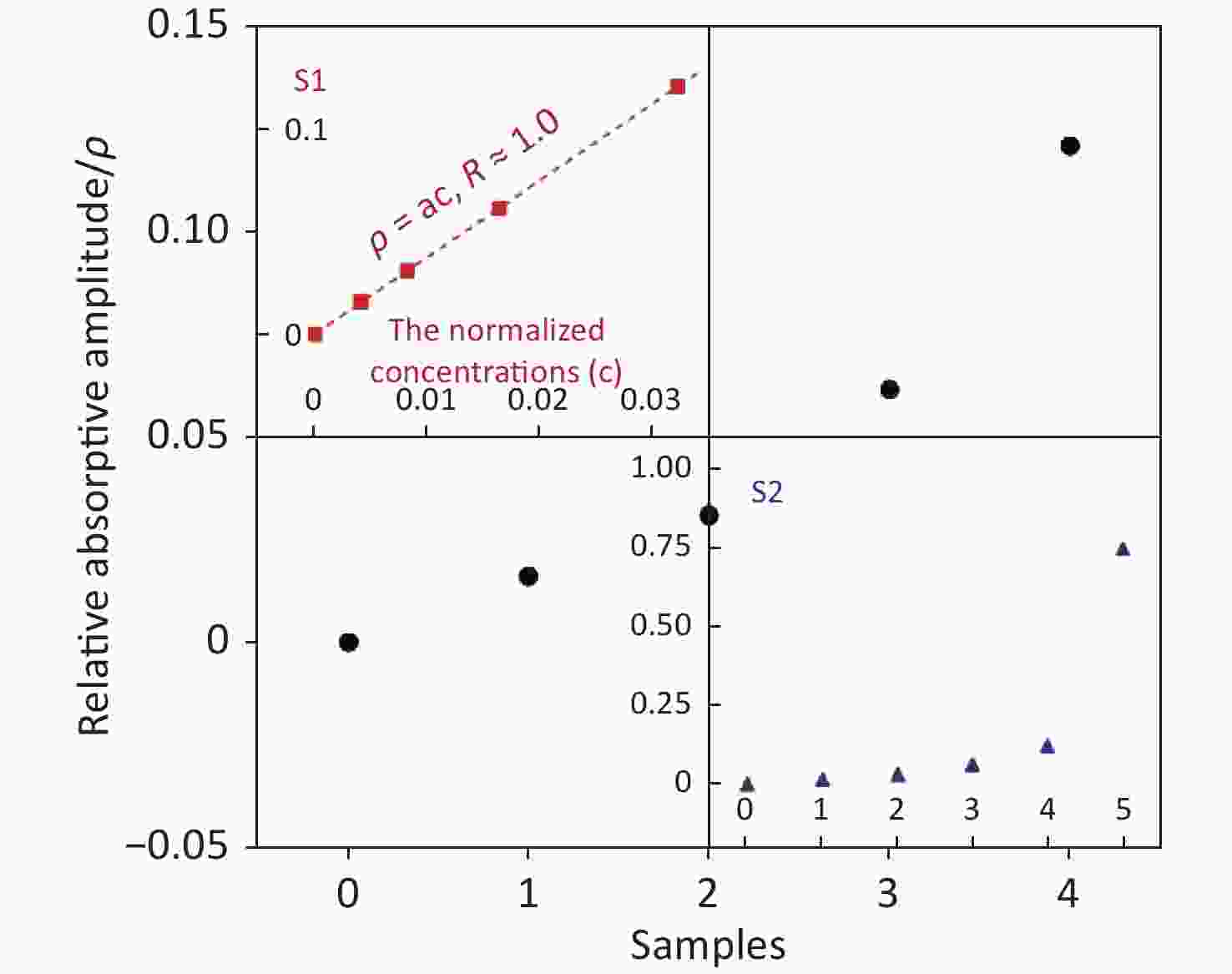
Figure 3. The relative absorptive amplitude (ρ) vs. different samples ''0–4'' (black dot). The value can be calculated using Equation (1). The amplitude of samples 1–4 is clearly higher than ''0'', which means our method has a higher sensitivity. The inserted panel S1 shows that the relation between ρ and c (normalized concentrations of samples) is almost linear ρ = ac, where a (= 3.75) at lower concentrations with a regression coefficient (R) of 1.0 (red square). The inserted panel S2 also displays the clearly very large difference between sample 0 and 5 (blue triangle).
In summary, the sensitivity of screening methods based on culture identification of the maternal carriage of GBS depends on the timing of specimen collection, the source of the specimen, and finally the culture technique used by the microbiology laboratory performing the analysis. Chromogenic media is a good alternative for identifying GBS carrier status among near-term pregnant women. Shortening the identification time of GBS and improving its sensitivity were the aims of this study.
It is very difficult for traditional visual identification to determine whether GBS has colonized the samples in samples 1–4, as shown in Figure 2. However, our POCT achieved a rapid trace analysis of GBS and reinforced the advancement made using chromogenic media. Each platform of the time course displays differences in altitude, as shown in Figure 2, which indicates that our method is more sensitive than the traditional one.
It should be noted that if a quantitative measurement of the CFU of sample "5" is done, then, the relation between ρ and CFU of GBS can be determined because this information is important for clinical trials.
The authors would like to thank Wenzhou Beikete Medical Equipment Co., Ltd. for supplying the samples and chromogenic media.
A Rapid Trace Analysis of GBS using the Specific Spectral Absorption of Chromogenic Culture Media
doi: 10.3967/bes2023.093
- Received Date: 2023-02-09
- Accepted Date: 2023-05-08
| Citation: | CHEN Yang, ZHANG Yan Yan, SHU Yao Gen. A Rapid Trace Analysis of GBS using the Specific Spectral Absorption of Chromogenic Culture Media[J]. Biomedical and Environmental Sciences, 2023, 36(7): 649-652. doi: 10.3967/bes2023.093 |








 Quick Links
Quick Links
 DownLoad:
DownLoad: