-
Chromium is a major industrial pollutant and its environmental level keeps increasing due to the extensive usage in leather tanning, stainless-steel production, and electroplating[1].Chromium can accumulate in human's food chain to impact human physiology, and causes many diseases. It may result in severe health problems ranging from simple skin irritation to lung carcinoma when contact with chromium[2]. The removal of highly toxic Cr(Ⅵ) has been proposed using membrane filtration[3], chemical agents and precipitation[4], ion exchange resins[5] among many others. However, these techniques are costly, energy-intensive and not efficient for removing Cr(Ⅵ) at low concentration. With the rapid development of nanotechnology, nanoparticles have been widely used in wastewater purification[6-9].
The studies on magnetite nanoparticles (MNPs) have shown good potential in the removal of metals via surface adsorption. This benefits its physicochemical properties, such as the high ratio of surface-to-volume that results in a better and more efficient on adsorption ability. Magnetite-supported adsorbents can be easily separated from the treated water using an external magnet. Therefore an efficient, economic, scalable synthesis of MNPs has been widely demanded in wastewater purification industry[10].
Although nanomaterials (NPs) have shown a profound impact on the practical applications, their potential biological and environmental toxicity have not been sufficiently studied yet. The adsorption of pollutants to nanoparticles alters the properties for both pollutant and nanoparticle, resulted adducts may cause an immediate threat to human health and ecology. For instance, the toxicity effects of titanium dioxide NPs + glucose to rats (Chen et al.[11]) showed that oral exposure of NPs + glucose induced more evident toxicity than NPs alone due to the effects of excessive glucose and the interactions between NPs and glucose. Wang et al.[12]have reported that vitamin C promoted the toxicity of ZnO NPs to gastric epithelial cell line and neural stem cells because the vitamin C accelerated uptake of Zn ions and the dissolution of ZnO NPs. Both indirect mechanisms and synergistic or inhibitory effects can enhance or suppress the expected responses from the specific classes of pollutants. Therefore, the biological impacts of nanoparticles-adducts need to be cautious evaluated. Recently, significant research efforts have been made toward the investigation of nanoparticle toxicity, very little attention has been paid to nanoadducts though the nanoadduct formations has been broadly used in environmental remediation which also has potentials to cause pollution in even broader areas. Thus, the impact of nanoadducts also needs to be thoroughly studied and it is just as important as the study of nanomaterials and pollutants.
The objective of the present study was to evaluate the effect of MNPs/Cr(Ⅵ) adducts on human embryonic kidney cell line HEK293 by assessing cell viability, apoptosis, oxidative stress induction, and cellular uptake. HEK293 was used as a model because Cr(Ⅵ) is known to induce nephrotoxicity[13]. they are essential in defining the toxicological response of the in vitro culture models to nanoparticle adducts exposure. Our results indicated that the cytotoxicity of the MNPs/Cr(Ⅵ) adducts was remarkably reduced compared to Cr(Ⅵ) anions. And the cellular uptake of MNPs/Cr(Ⅵ) adducts was rare. The particles were endocytosed from the extracellular fluid and could not enter into the cell nucleus. In this case, MNPs/Cr(Ⅵ) adduct formation significantly reduces its associated cytotoxicity.
-
Magnetite Fe3O4nanoparticles (MNPs) were synthesized by coprecipitation of ferric and ferrous ion according to Laurent et al.[14]. A stock of solution of Cr(Ⅵ) at concentration of 500 mg/L was prepared by dissolving a known quantity of potassium dichromate (K2Cr2O7) in ultrapure water. MNPs/Cr(Ⅵ) adduct was prepared by mixing appropriate amount of MNPs with Cr(Ⅵ) solutions of varying concentration from 0 to 30 mg/L in 15 mL centrifuge tubes. After formation of MNPs/Cr(Ⅵ), MNPs/Cr(Ⅵ) adduct was washed by centrifugation and re-dispersion in distilled water for three times.
-
The size of morphology of MNPs and MNPs/Cr(Ⅵ) adducts were characterized by scan electron microscopy (SEM, FESEM 6700F, JEOL, Japan). Their hydrodynamic size was analyzed using a dynamic light scattering instrument (DLS, Nano ZS 90, Malvern, UK). Samples were sonicated prior to DLS determination. The structure phases of MNPs and MNPs/Cr(Ⅵ) adducts were analyzed using X-ray diffraction (XRD, Bruker D8, Germany).
-
The human embryonic kidney cell line HEK293 was purchased from the Cell Resource Center of Shanghai Institute of Biological Sciences, Chinese Academy of Sciences (Shanghai, China). HEK293 cells were cultured with DMEM media, supplemented with 10% FBS (Gibco, Grand Island, NY, USA), 100 IU/mL penicillin, and 100 μg/mL streptomycin, incubated at 37 ℃ under 95% air and 5% CO2. The cells were regularly monitored with an inverted light microscope (Leica, DMI3000B) and the culture medium was changed every day. The cells were normally subcultured at a ratio of 1:3 every 2 days to sustain the exponential growth phase.
-
HEK293 cells were placed on 96-well cell culture plates with 5 × 103 cells in 200 μL medium per well. The plates were incubated at 37 ℃ for 12 h under a 5% CO2 atmosphere and humid chamber until the cells were adhered to the surface of the cell culture dish. Twenty μL of each followings, Cr(Ⅵ) at 0, 10, 20, and 30 μg/mL, MNPs at 4 g/L, and synthesized MNP/Cr(Ⅵ) adduct, were added to individual wells respectively. Blank medium wells serviced as negative control in all cases. The culture plates were further incubated for 24 h.
Cell mitochondria dehydrogenase activity, which was proportional to the cell viability, was determined using the WST-1 assay (Beyotime Institute of Biotechnology, China). When color of WST-1 reagent is converted to yellow formazan, it indicates the mitochondria in living cells. Cell morphology after being treated with Cr(Ⅵ) solution, magnetite Fe3O4, and the corresponding adducts were also monitored using a microplate spectrophotometer (Biotek, Epoch, USA).
-
Cells positive of apoptosis and necrosis were estimated by the annexinV/propidium iodide (PI) assay[15]. In brief, the HEK293 cells that treated with Cr(Ⅵ), MNPs, and their adducts were collected by trypsinization and washed twice with PBS. After resuspending the cells with the binding buffer, Annexin V-FITC and PI were added onto the cells successively. Cells were analyzed via FlowSight® Imaging Flow Cytometer (Amnis, Washington, USA). The data were analyzed with the IDEAs software (Amnis Corporation).
-
Cells were exposed to Cr(Ⅵ), MNPs, and their adducts for 24 h, washed once with ice-cold PBS, and lysed in ice-cold RIPA lysis buffer for 30 min. The lysates were centrifuged at 8, 000 × g for 10 min. The supernatants were collected for the measurements of malondialdehyde (MDA) contents, activities of superoxide dismutase (SOD), and glutathione (GSH, GSSG) content. Biochemical parameters for oxidative stress, including the activity of superoxide dismutase (SOD), glutathione (GSH, GSSG) content, and malondialdehyde (MDA) levels, were determined using a Diagnostic Reagent Kit (Nanjing jiancheng Bioengineering Institute, Nanjing, China) according to the manufacturer's instruction. Protein content was measured with a Bradford Protein Assay Kit (Beyotime, China).
Briefly, the SOD activity assay was based on its ability to inhibit the oxidation of oxymine by O2-produced by the xanthine-xanthine oxidase system. The red product (nitrite) produced by the oxidation of oxymine had an absorbance at 550 nm. GSH is the major intracellular low-molecular weight thiol that plays a critical role in the cellular defense against oxidative stress. GSH acts as an electron donor in the reduction of peroxides governed by glutathione peroxidase, leading to the formation of oxidized glutathione (GSSG). The lipid peroxidation products (measured as MDA) were quantified using the thiobarbituric acid (TBARS) method, and the MDA concentration was expressed as nmol.mg/protein.
-
Qualitative analysis of internalized nanoparticles was performed by TEM. Cells were collected and fixed with 2.5% glutaraldehyde in 0.1 mol/L PBS for 30 min at room temperature and postfixed in 1% osmium tetroxide (pH 7.4). Then the cells were dehydrated in increasing grades of ethanol and embedded. Ultrathin sample sectioning was performed using a diamond knife, and the sections were observed under TEM (Tecnai G20 TWINW, FEI, USA). HEK293 were washed three times with PBS and trypsinized. After centrifugation, the cell pellets were digested in 3 mL of nitric acid (HNO3) and 1 mL of hydrogen peroxide (H2O2) with ultrasound. Quantification of cellular uptake was performed by ICP-MS (NexION 350X, USA).
-
Each experiment was performed at least in triplicate. Reproducibility of each experiment was confirmed independently for three times. Values are expressed as means ± standard deviation (SD). Statistical differences were determined by analysis of variance followed by Student's t test. All statistical analyses were performed using SPSS 13.0. Differences considered to be statistically significant if P-value is less than 0.05.
-
Size, shape, and chemical composition are the important properties in the nanoparticle toxicity investigation[16]. The particle size and appearance of MNPs and MNPs/Cr(Ⅵ) adducts were determined by SEM. The images clearly showed spherical morphology and the diameters of MNPs and MNPs/Cr(Ⅵ) ranged from 10 to 20 nm (Figure 1A-B). The hydrodynamic diameter of MNPs and MNPs/Cr(Ⅵ) in culture medium was 216 nm and 352 nm respectively, as determined by DLS (Table 1). The size of nanoparticles in aqueous suspension was much higher compared to SEM. This might be due to the particle agglomeration tendency in the aqueous state. This finding was consistent with our previous observation[17-19].The crystalline structure and magnetic properties of MNPs and MNPs/Cr(Ⅵ) adducts were verified by XRD patterns (Figure 1C).
Table 1. Characterization of MNPs and MNPs/ Cr(Ⅵ) Adducts
Characterization MNPs Adducts Primary size/SEM (nm) 10-20 Hydrodynamic size/DLS (nm) 216 352 Absorption/K2Cr2O7μg/mg MNPs NA 13.4 -
Adsorption of Cr(Ⅵ) onto Fe3O4MNPs reduced the quantity of Cr(Ⅵ) that was readily taken up by cells. Cr(Ⅵ)-induced cytotoxicity was demonstrated by the optic microscopic observation in which the cell shrinkage was aggravated as increase of Cr(Ⅵ) concentration. MNPs/Cr(Ⅵ) adducts-induced cytotoxicity was significantly reduced as showed in the Figure 2A. We therefore expected that MNPs/Cr(Ⅵ) might result in less cytotoxicity compared to free Cr(Ⅵ). To verify this, we identified the viability of cells by analyzing the activity of mitochondria dehydrogenase, which is proportional to the number of live cells. Figure 2B showed the cell viabilities of different treatments. The Cr(Ⅵ) clearly induced a dose-dependent cytotoxic effect on HEK293 cells. As the Cr(Ⅵ) concentration increased to 30 mg/L, the cell viability was reduced to 55.3% compared to the controls. For the cells treated with both Cr(Ⅵ) and MNPs, the cell viabilities were not significantly different from the control cells and from the cells treated with MNPs only. Therefore, the Cr(Ⅵ) inducted cytotoxicity was markedly ameliorated after it was adsorbed onto the MNPs to form the MNPs/Cr(Ⅵ) adducts, which reduced the free-Cr(Ⅵ) level in the system.
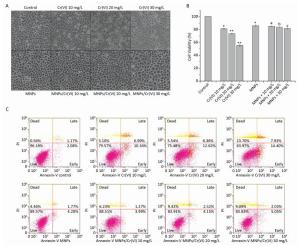
Figure 2. Cells were treated with different concentrations of Cr(Ⅵ), MNPs and corresponding MNPS/Cr(Ⅵ) adducts for 24 h. (A) Changes of cell morphology in Cr(Ⅵ)-exposed cells observed by optic microscope, magnification is × 200. (B) Cell viability assessment by quantifying mitochondria dehydrogenase activity. Significant differences are indicated by *vs. control. a, b, cvs. the corresponding Cr(Ⅵ) groups without MNPs (*P < 0.05, **P < 0.01). (C) Cell apoptosis analysis by Flow cytometry.
To identify whether Cr(Ⅵ) and MNPs/Cr(Ⅵ) adducts induced apoptosis, the treated cells were stained with Annexin V-FITC/PI. The living cells were Annexin V -FITC and PI double-negative, whereas the late apoptotic or secondary necrotic cells were double-positive. The early apoptotic cells were only Annexin V -FITC positive, whereas the isolated nuclei or cellular debris were only PI positive. An increase in the FITC-conjugated Annexin V -positive cells is an early marker for apoptosis. PI staining was used to investigate the loss of cell membrane integrity. The rate of apoptosis in HEK293 cells (i.e., the proportion of AnnexinV+ cells) was shown in Figure 2C. Twenty-four hours after Cr(Ⅵ) exposure, higher percentages of early-and late-apoptotic cells were observed compared with the control. However, after MNPs/Cr(Ⅵ) adducts exposure, the percentage of apoptotic cells were significantly decreased.
Oxidative stress has been recommended as an ordinary pathway for nanoparticle-induced toxicity[20-21].This parameter was evaluated with three indicators, SOD, GSH, GSSG, and MDA in our study. The Indicator activities impacted by Cr(Ⅵ), MNPs, and MNPs/Cr(Ⅵ) adducts, were compared in Figure 3. Results indicated that the GSSG and MDA levels were significantly enhanced and the SOD and GSH concentrations were significantly inhibited in Cr(Ⅵ) treated HEK293 cells relative to the control group. Comparing the four parameters among all treated groups, the SOD and GSH concentrations were remarkably higher and both GSSG and MDA levels were significantly declined in HEK293 cells treated with MNPs and MNPs/Cr(Ⅵ) adducts relative to the groups treated with the high level of Cr(Ⅵ), but there is no significant differences between MNPs group and MNPs/Cr(Ⅵ) adducts group.
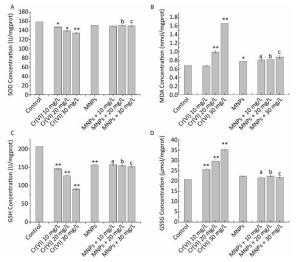
Figure 3. Effects of Cr(Ⅵ) (0-30 mg/L) with and without MNPs (4 g/L) on (A) superoxide dismutase (SOD), (B) malondialdehyde (MDA), (C) reduced glutathione (GSH), and (D) oxidized glutathione (GSSG) in HEK293 cells. Significant differences are indicated by *vs. control. a, b, cvs. the corresponding Cr(Ⅵ) groups without MNPs (*P < 0.05, **P < 0.01).
Our results are in accordance with those of previous studies in which the MNPs treatment caused an obvious increase in SOD and GSH levels and a significant reduction in MDA levels[22-25].
-
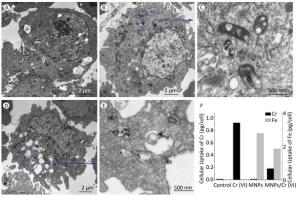
To further evaluate the cell-specific cytotoxicity, we took a series of TEM images for HEK293 cell to observe the entire internalization process of MNPs and MNPs/Cr(Ⅵ) adducts into the HEK293 cell. (Representative images after 24 h incubation were showed in Figure 4) As it can be seen in the figures, after the cells were cultured with MNPs and MNPs/Cr(Ⅵ) adducts for 24 h, they were rarely detectable inside the HEK293 cells and the cell nucleus remained intact. To confirm the uptake of the MNPs and MNPs/Cr(Ⅵ) by the cells, elemental analysis was employed by ICP-MS to determine the iron content of the cells. The concentration of iron atoms in acid digesting solution was directly measured. The results were consistent with the TEM observations. The cellular iron levels of MNPs and MNPs/Cr(Ⅵ) adducts were less than 2.78 pg/cell and 1.85 pg/cell respectively, as determined by ICP-MS (Figure 4F).
-
As to apply the nanomaterials in environmental remediation, nanoparticles spontaneously adsorb the chemicals including environmental pollutants. The subsequently generated nanoadducts cause an instant threat to human health and ecology. Since the MNPs is being widely used in water pollution remediation to remove the toxic Cr(Ⅵ), it is necessary to confirm the combined biological activity of MNPs/Cr(Ⅵ) adducts. HEK293 is a promising model for assessing cytotoxicity because the Cr(Ⅵ) is known to induce the nephrotoxicity.
To elucidate the toxicity of nanoadducts, it is essential to consider the properties of MNPs/Cr(Ⅵ) adduct. The particle size, surface area, and chemical form of the nanomaterials are important parameters for assessing nanoparticle toxicity[26]. The shapes of MNPs and MNPs/Cr(Ⅵ) were spherical with average diameter around 10-20 nm as observed through the scan electron microscopy. The hydrodynamic diameter of MNPs and MNPs/Cr(Ⅵ) in culture medium was 216 nm and 352 nm as determined by DLS. The hydrodynamic diameter of MNPs increase notably after reacting with Cr(Ⅵ). This phenomenon could be ascribed to the co-precipitation of Cr(Ⅵ) and MNPs. The XRD patterns of MNPs showed that the Fe3O4peak intensity dropped significantly after the reaction with Cr(Ⅵ). Meanwhile, the new particles Fe2O3 and (Fe0.6Cr0.4)2O4 were detected in the reaction media. This clearly indicated the formation of MNPs/Cr(Ⅵ) adducts and demonstrated MNPs capability to adsorb Cr(Ⅵ) in the aqueous medium.
In vitro toxicological assessment is an extremely vital tool for nanotoxicology. The most commonly used methods in vitro assessment include three major class of cell based assays, cell viability, apoptosis, and oxidative stress. The in vitro study was conducted using human embryonic kidney cell line to evaluate the nanoadducts were evaluated ability to induce the cell toxicity. The nanoadducts suspensions were collected and applied on the HEK293 cells. After 24 h treatment, the effects of Cr(Ⅵ), MNPs, and their adduct on cell morphology were assessed via microscopic observations. The WST-1 reduction was calculated for each case. Our data indicated that the Cr(Ⅵ) could enter the cells and induce morphologic alternation. The cell damage rate was Cr(Ⅵ) dose level dependent. Simultaneously, the cell viability also showed the Cr(Ⅵ) dose level dependent reduction. In contrast to Cr(Ⅵ), HEK293 cells only expressed a slight sensitivity towards to the MNPs and MNPs/Cr(Ⅵ) adducts.
MNPs reduced apoptosis was clearly demonstrated in HEK293 cells. The results showed statistically significant difference in the apoptotic rates of the different Cr(Ⅵ) concentrations group, but not in the MNPs/Cr(Ⅵ) adducts group. In apoptosis assay, the apoptotic rate in HEK293 cells was reduced in MNPs treated group but accelerated in Cr(Ⅵ) treated group. The acceleration was Cr(Ⅵ) dose dependent and the apoptosis was statistically significant difference among three levels of Cr(Ⅵ) groups. However, dose dependent apoptosis was not observed in MPNs/Cr(Ⅵ) adducts treated cells. The adsorption of Cr(Ⅵ) onto MNPs diminished the number of free Cr(Ⅵ) in the system, weakened Cr(Ⅵ) toxicity effect, and consequently reduced the number of apoptotic cell. Therefore, it revealed that MNPs/Cr(Ⅵ) adducts didn't have obvious effect on the apoptosis of HEK293 cells as Cr(Ⅵ) did. The slight toxicity effect of MNPs/Cr(Ⅵ) adducts may be caused by desorbed Cr(Ⅵ) anions. Cytotoxic observations on HEK293 cells from our study were in alignment with the findings reported by other researches on the aspects of cell viability and apoptosis. For instance, Schulze et al.[27]reported that the dextran-stabilized MNPs (20 nm) were not toxic to phagocytic C6 cells at concentration of 0.73 mol/L iron for up to 10 days. Similarly, Ferumoxtran-10 was not toxic to human hematopoietic progenitor cells after 2 h at a concentration of 0.25 mg/mL[28].
Oxidative stress is considered as one of the most important mechanisms for cytotoxic effects of NPs[29]. As a result of Cr(Ⅵ) is a powerful oxidizing agent that restrains cell growth by disturbing nitrogen metabolism. Not only particle toxicity but also oxidative damage can cause toxic effects[30-31]. It is well-known that an oxidizing agent will restrain cell growth by disturbing nitrogen metabolism. Cr(Ⅵ) is a powerful oxidizing agent. Its cytotoxic effects will come from both particle toxicity and the oxidative damage. Therefore, the reduction of cell viability may be the attribution of the interactions of NPs with metabolic intermediates, such as H2O2 or H+[32].Nevertheless, oxidative damage of MNPs or MNPs/Cr(Ⅵ) has not been widely studied yet. The outcomes of the present study provided the evidence that MNPs addition together with Cr(Ⅵ) significantly ameliorated Cr(Ⅵ)-induced damaging effects on HEK293 cells. MNPs/Cr(Ⅵ) adducts did not initiate the oxidative damage. Previously, Heike et al.[33].had shown that Pd/magnetite NPs did not initiate oxidative damage and caused little impact on viability of selected mammalian and a fish cell line which was consistent with our suggestion. Therefore, the possible reasons for MNPs ameliorating the toxicity could be: (1) MNPs decreased Cr(Ⅵ) accumulation; (2) MNPs decreased the oxidative damage; (3) MNPs/Cr(Ⅵ) adducts was not toxic.
Characterization of nanoparticle uptake and localization is inherently related to the cell toxicity studies owing to the uptake provides evidences of the nanoparticle-cell interaction. Quantifying number of nanoparticles taken up by the cells and describing the localization of these materials presents analytical challenges that research groups have approached with a variety of techniques.
The result showed that the cellular uptake of MNPs/Cr(Ⅵ) adducts was rare. There were only few cells that were able to internalize the nanoparticles and the particles were accumulated through the whole cytosol. Most nanoparticles could be attached to the membrane of the cells, but these nanoparticles were easily washed out before imaging. The particles are endocytosed from the extracellular fluid. Due to relatively large size of MNPs/Cr(Ⅵ) adduct, when the cells are exposed to these particles, the membrane of the cell needs more effort to initiate the process of endocytosis. Therefore, it is more difficult to find MNPs/Cr(Ⅵ) adducts in the cells compared to MNPs and there is no MNPs/Cr(Ⅵ) adducts accumulation in the cell nucleus. The particles were aggregated, as shown in the cytoplasm. A portion of the plasma membrane was invaginated and pinched off to form a membrane-bound vesicle, as the arrows pointed in Figure 4C and 4E, respectively. As intracellular localization plays an important role in nanoparticles-induced toxicity, localization of nanomaterials in mitochondria and in the nucleus may induce interference in cellular functions. However, recent studies reported that endosomes and lysosomes were the most ordinary areas for nanomaterial localization[34-35].Studies with Ag nanomaterials showed that the nanoparticles did not induce significant cytotoxicity with the evidence of them entering endosomes and lysosomes[36]. Our study suggested that only few MNPs/Cr(Ⅵ) nanoparticles were endocytosed from the extracellular fluid which sequestered in digestive vacuoles, metabolized in a normal path and would not lead to the apoptosis[37].
-
Our study results indicated that MNPs/Cr(Ⅵ) adducts do not lead to the oxidative stress. It only causes a minor impact on the cell toxicity. The cellular uptake of MNPs/Cr(Ⅵ) adduct is rare. Only few particles are endocytosed from the extracellular fluid and not accumulated in the nucleus of cells.
Our findings have important implications for the environmental usage of the nanotechnology. Compare to the traditional methods of wastewater purification such as membrane filtration, chemical agents and precipitation and ion exchange resins, magnetic nanoparticle method is highly efficient, more sensitive, cost effective, highly selective and easily separated from loaded metals using magnetic field. MNPs have superb adsorption capacity and minor cytotoxicity. After they are contacted to the Cr(Ⅵ), formed MNPs/Cr(Ⅵ) adducts express the little cytotoxic behaviors. Meanwhile, the formation of adducts removes the free Cr(Ⅵ) anions from the water system, remediates the environmental risks. Therefore, the MNPs will be continuously used in pollutant removal applications. The presented study results support that the magnetite nanoparticles can be implemented for a wastewater treatment technology in which advantageous properties outweigh the risks.
-
We thank Environmental Monitoring Center of Jilin province for the assistance on measurements. This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
-
No conflict of interest to declare.
doi: 10.3967/bes2017.106
Combined Toxicity of an Environmental Remediation Residue, Magnetite Fe3O4 Nanoparticles/Cr(Ⅵ) Adduct
-
Abstract:
Objective This paper aims to elucidate the combined toxicity of magnetite nanoparticles/Chromium[MNPs/Cr(Ⅵ)] adducts. Methods The HEK293 cell was exposed to either Cr(Ⅵ) or MNPs, or their adducts MNPs/Cr(Ⅵ). The cytotoxicity was evaluated by assessing the cell viability, apoptosis, oxidative stress induction, and cellular uptake. Results The toxicity of formed adducts is significantly reduced when compared to Cr(Ⅵ) anions. We found that the cellular uptake of MNPs/Cr(Ⅵ) adduct was rare, only few particles were endocytosed from the extracellular fluid and not accumulated in the cell nucleus. On the other hand, the Cr(Ⅵ) anions entered cells, generated oxidative stress, induced cell apoptosis, and caused cytotoxicity. Conclusion The results showed minor effects of the nanoadducts on the tested cells and supported that magnetite nanoparticles could be implemented in the wastewater treatment process in which advantageous properties outweigh the risks. -
Key words:
- Magnetite Fe3O4 /
- Chromium /
- Adduct /
- Toxicity
-
Figure 2. Cells were treated with different concentrations of Cr(Ⅵ), MNPs and corresponding MNPS/Cr(Ⅵ) adducts for 24 h. (A) Changes of cell morphology in Cr(Ⅵ)-exposed cells observed by optic microscope, magnification is × 200. (B) Cell viability assessment by quantifying mitochondria dehydrogenase activity. Significant differences are indicated by *vs. control. a, b, cvs. the corresponding Cr(Ⅵ) groups without MNPs (*P < 0.05, **P < 0.01). (C) Cell apoptosis analysis by Flow cytometry.
Figure 3. Effects of Cr(Ⅵ) (0-30 mg/L) with and without MNPs (4 g/L) on (A) superoxide dismutase (SOD), (B) malondialdehyde (MDA), (C) reduced glutathione (GSH), and (D) oxidized glutathione (GSSG) in HEK293 cells. Significant differences are indicated by *vs. control. a, b, cvs. the corresponding Cr(Ⅵ) groups without MNPs (*P < 0.05, **P < 0.01).
Table 1. Characterization of MNPs and MNPs/ Cr(Ⅵ) Adducts
Characterization MNPs Adducts Primary size/SEM (nm) 10-20 Hydrodynamic size/DLS (nm) 216 352 Absorption/K2Cr2O7μg/mg MNPs NA 13.4 -
[1] Kantar C, Cetin Z, Demiray H. In situ stabilization of chromium (Ⅵ) in polluted soils using organic ligands:The role of galacturonic, glucuronic and alginicacids. Hazard Mater, 2008; 159, 287-93. doi: 10.1016/j.jhazmat.2008.02.022 [2] Jha MK, Kumar V, Bagchi D, et al. Processing of rayon waste effluent for the recovery of zinc and separation of calcium using thiophosphinic extractant. Hazard Mater, 2007; 145, 221-6. doi: 10.1016/j.jhazmat.2006.11.015 [3] Aliane A, Bounatiro N, Cherif AT, et al. Removal of chromium from aqueous solution by complexation-ultra filtration using a water-soluble macroligand. Water Res, 2001; 35, 2320-6. doi: 10.1016/S0043-1354(00)00501-7 [4] Gheju M, Balcu I. Removal of chromium from Cr(Ⅵ) polluted wastewaters by reduction with scrap iron and subsequent precipitation of resulted cations. Hazard Mater, 2011; 196, 131-8. doi: 10.1016/j.jhazmat.2011.09.002 [5] Rengaraj S, Yeon KH, Moon SH. Removal of chromium from water and wastewater by ion exchange resins. Hazard Mater, 2001; 87, 273-87. doi: 10.1016/S0304-3894(01)00291-6 [6] Guan X, Chang J, Chen Y, et al. A magnetically-separable Fe3O4 nanoparticle surface grafted with polyacrylic acid for chromium(Ⅲ) removal from tannery effluents. RSC Advances, 2015; 5, 50126-36. doi: 10.1039/C5RA06659J [7] Luther S, Brogfeld N, Kim J, et al. Study of the Thermodynamics of Chromium(Ⅲ) and Chromium(Ⅵ) Binding to Fe3O4 and MnFe2O4 nanoparticles. Colloid Interface Sci, 2013; 400, 97-103. doi: 10.1016/j.jcis.2013.02.036 [8] Tang SC, Lo IM. Magnetic nanoparticles:Essential factors for sustainable environmental applications. Water Res, 2013; 47, 2613-32. doi: 10.1016/j.watres.2013.02.039 [9] Guan X, Yan S, Zeng Q, et al. Polyacrylic acid-grafted magnetite nanoparticles for remediation of Pb(Ⅱ)-contained water. Fiber Polym, 2016; 17, 1131-9. doi: 10.1007/s12221-016-6529-1 [10] Shen YF, Tang J, Nie ZH, et al. Preparation and application of magnetic Fe3O4 nanoparticles for wastewater purification. Sep Purif Technol, 2009; 69, 312-9. [11] Chen Z, Wang Y, Zhuo L, et al. Interaction of titanium dioxide nanoparticles with glucose on young rats after oral administration. Nanomedicine, 2015; 11, 1633-42. doi: 10.1016/j.nano.2015.06.002 [12] Wang Y, Yuan L, Yao C, et al. A combined toxicity study of zinc oxide nanoparticles and vitamin C in food additives. Nanoscale, 2014; 6, 15333-42. doi: 10.1039/C4NR05480F [13] Sahu BD, Koneru M, Bijargi SR, et al. Chromium-induced nephrotoxicity and ameliorative effect of carvedilol in rats:Involvement of oxidative stress, apoptosis and inflammation. Chem Biol Interact, 2014; 223, 69-79. doi: 10.1016/j.cbi.2014.09.009 [14] Laurent S, Forge D, Port M, et al. Magnetic iron oxide nanoparticles:synthesis, stabilization, vectorization, physicochemical characterizations and biological applications. Chem Rev, 2008; 108, 2064-110. doi: 10.1021/cr068445e [15] van Engeland M, Ramaekers FC, Schutte B, et al. A novel assay to measure loss of plasma membrane asymmetry during apoptosis of adherent cells in culture. Cytometry, 1996; 24, 131-9. doi: 10.1002/(ISSN)1097-0320 [16] Auffan M, Rose J, Bottero JY, et al. Towards a definition of inorganic nanoparticles from an environmental, health and safety perspective. Nat Nanotech, 2009; 4, 634-41. doi: 10.1038/nnano.2009.242 [17] Dwivedi S, Alkhedhairy AA, Ahamed M, et al. Biomimetic Synthesis of Selenium Nanospheres by Bacterial Strain JS-11 and Its Role as a Biosensor for Nanotoxicity Assessment:A Novel Se-Bioassay. PLOS ONE, 2013; 8, e57404. doi: 10.1371/journal.pone.0057404 [18] Wahab R, Khan ST, Dwivedi S, et al. Effective inhibition of bacterial respiration and growth by CuO microspheres composed of thin nanosheets. Colloid Surface B, 2013; 111, 211-7. doi: 10.1016/j.colsurfb.2013.06.003 [19] Sigh BR, Dwivedi S, Al-Khedhairy AA, et al. Synthesis of stable cadmium sulfide nanoparticles using surfactin produced by Bacillus amyloliquifaciens strain KSU-109. Colloid Surface B, 2011; 85, 207-13. doi: 10.1016/j.colsurfb.2011.02.030 [20] Foldbjerg R, Dang DA, Autrup H. Cytotoxicity and genotoxicity of silver nanoparticles in the human lung cancer cell line, A549. Arch Toxicol, 2011; 85, 743-50. doi: 10.1007/s00204-010-0545-5 [21] Kim S, Choi JE, Choi J, et al. Oxidative stress dependent toxicity of silver nanoparticles in human hepatoma cells. Toxicol in Vitro, 2009; 23, 1076-84. doi: 10.1016/j.tiv.2009.06.001 [22] Culcasi M, Benameur L, Mercier A, et al. EPR spin trapping evaluation of ROS production in human fibroblasts exposed to cerium oxide nanoparticles:Evidence for NADPH oxidase and mitochondrial stimulation. Chem Biol Interact, 2012; 199, 161-76. doi: 10.1016/j.cbi.2012.08.007 [23] Benameur L, Auffan M, Cassien M, et al. DNA damage and oxidative stress induced by CeO2 nanoparticles in human dermal fibroblasts:Evidence of a clastogenic effect as a mechanism of genotoxicity. Nanotoxicology, 2014; 9, 696-705. [24] Zhu MT, Wang Y, Feng WY, et al. Oxidative Stress and Apoptosis Induced by Iron Oxide Nanoparticles in Cultured Human Umbilical Endothelial Cells. J Nanosci Nanotechno, 2010; 10, 8584-90. doi: 10.1166/jnn.2010.2488 [25] Naqvi S, Samim M, Abdin M, et al. Concentration-dependent toxicity of iron oxide nanoparticles mediated by increased oxidative stress. Int J Nanomed, 2010; 16, 983-9. [26] Peralta-Videa JR, Zhao L, Lopez-Moreno ML, et al. Nanomaterials and the environment:a review for the biennium 2008-2010. Hazard Mater, 2011; 186, 1-15. doi: 10.1016/j.jhazmat.2010.11.020 [27] Schulze E, Ferrucci JT Jr, Poss K, et al. Cellular uptake and trafficking of a prototypical magnetic iron oxide label in vitro. Invest Radiol, 1995; 10, 604-10. https://www.researchgate.net/publication/14642026_Cellular_Uptake... [28] Daldrup-Link HE, Rudelius M, Oostendorp RA, et al. Targeting of hematopoietic progenitor cells with M R contrast agents, Radiology, 2003; 28, 760-7. [29] Hoet P, Boczkowski J. What's new in Nanotoxicology? Brief review of the 2007 literature. Nanotoxicology, 2009; 2, 171-86. [30] Schiavon M, PilonSmits EA, Wirtz M, et al. Interactions between chromium and sulfur metabolism in Brassica juncea. J. Environ. J Environ Qual, 2008; 37, 1536-45. doi: 10.2134/jeq2007.0032 [31] Gangwar S, Singh VP. Indole acetic acid differently changes growth and nitrogen metabolism in Pisumsativum L. seedlings under chromium (Ⅵ) phytotoxicity:Implication of oxidative stress. Sci HorticAmst, 2011; 129, 321-8. doi: 10.1016/j.scienta.2011.03.026 [32] Moore MN. Do nanoparticles present ecotoxicological risks for the health of the aquatic environment. Environ Int, 2006; 32, 967-76. doi: 10.1016/j.envint.2006.06.014 [33] Heike H, Dana K, Annegret P, et al. Evaluating the cytotoxicity of palladium/magetitenano-catalysts intended for wastewater treatment. Environ Pollut, 2010; 158, 65-73. doi: 10.1016/j.envpol.2009.08.021 [34] Chithrani BD, Chan WCW. Elucidating the mechanism of cellular uptake and removal of protein-coated gold nanoparticles of different sizes and shapes. Nano Lett, 2007; 7, 1542-50. doi: 10.1021/nl070363y [35] Peckys DB, de Jonge N. Visualizing gold nanoparticle uptake in live cells with liquid scanning transmission electron microscopy. Nano Lett, 2011; 11, 1733-8. doi: 10.1021/nl200285r [36] Jiang XM, Foldbjerg R, Miclaus T, et al. Multi-platform genotoxicity analysis of silver nanoparticles in the model cell line CHO-K1. Toxicol Lett, 2013; 222, 55-63. doi: 10.1016/j.toxlet.2013.07.011 [37] Lewinski N, Colvin V, Drezek R. Cytotoxicity of Nanoparticles. Small, 2008; 4, 26-49. doi: 10.1002/(ISSN)1613-6829 -





 下载:
下载:




 Quick Links
Quick Links