-
Organoids are primary tissue or stem cells derived cell aggregates that have the capacity for self-organization, self-renewal, and the capacity to mimic cellular and tissue level functions. Organoids can overcome the shortcomings of traditional 2D cell culture models and closely mimic 3D primary tissue composition, architecture, and biologically relevant models making excellent in vitro systems. Organoids can be derived from primary tissues, and pluripotent stem cells [both induced pluripotent stem cells (iPSCs) and embryonic stem cells (ESCs)] by supplementing with appropriate physical and biochemical cues. Physical cues in the form of extracellular matrices such as collagen, fibronectin, entactin, and laminin are supplied to provide the appropriate environmental conditions for the development of 3D architecture, and cell-to-cell communications and help in the functional survival of the organoids. Biochemical cues such as the variety of growth factors, EGF, Noggin, Activin A, and R-spondins[1], help in regulating the cell signaling pathways leading to control of cell proliferation, differentiation, and self-renewal[2].
Organoids are very relevant in basic research as well as translational applications. Stem cell-derived organoids show homology with embryonic developmental stages, lineage specifications, and tissue homeostasis hence it has helped to understand the developmental biology of organs such as the brain, heart, pancreas, and stomach[3]. Since organoids represent most of the components of the organs, they can be utilized to study the genetic, non-genetic, metabolic, and disease pathology of infectious diseases. Recent advances in gene editing technologies (CRISPR/Cas9, TALEN, etc.) and single-cell genomics provides invaluable insights for the application of organoids to study disease and development of the organ[4]. Drug discovery, efficacy, and toxicity against specific organs can be tested using vascularized organ-on-chip and microfluidics technology[5-7]. Hence, organoid technology is now becoming an important tool in regenerative medicine and personalized medicine.
Organoids have proved to be a major tool during the COVID-19 pandemic to mimic SARS-CoV-2 pathogenesis in the dish and to discover newer drugs against the virus. Humans and bat intestinal organoids were developed to grow SARS-CoV-2 reproducibly and study the biology of coronaviruses[8-10]. Organoids had also allowed the cultivation and identification of coronaviruses in the most natural environment that was not earlier possible to cultivate using immortalized cell lines. The development of airway, neuronal, kidney, cardiac, and intestinal organoids has helped to understand the biology of the virus more clearly and enable the finding of the appropriate drug targets[11,12]. SARS-CoV-2 can easily grow in the VERO cell line but the capillary and kidney organoids had helped understand the virus strategy to damage the kidney in severely ill patients[13]. Intestinal organoids helped in understanding the ACE2 expression level in the gastrointestinal tract establishing it as another entry point for viruses other than the respiratory route[10]. SARS-CoV-2 exposure to the human brain organoid revealed that the virus alters the Tau distribution from axons to the stroma, hyperphosphorylation and it can lead to neuronal death[14]. Similarly, Mills et al.[15] have used cardiac organoids to identify pathways associated with cardiac injury in patients with coronavirus and inhibitors to rescue cardiac dysfunction. The iPSC cell line was developed from the fibroblasts of the adult zebrafish by using the doxycycline-inducible lentiviral delivery system and chemical molecules[16]. The zebrafish induced pluripotent cells (ziPSCs) were stable and the features were similar to fish embryonic stem cells within in vivo and in vitro pluripotent nature[16]. The study of stem cells was best explored in three vertebrate organisms including mice, humans, and medaka. The endothelial cells were enriched from the embroid body derived from the blastocyst of fli: GFP and kdrl: GFP transgenic zebrafish[17]. The endothelial cell enrichment can increase the chances of the development of functional circulation leading to the development of more organ-like organoid tissues. Zebrafish embryonic explant was shown to specify ectoderm, mesoderm, and endoderm and form a mesendoderm lineage even when prepared before the germ layer formation and without any extraembryonic tissues. The zebrafish explants showed genetically regulated self-assembly and the order of structure formation seems to be regulated by intrinsic genetic programs[18]. Embroid body from transgenic zebrafish kdrl: GFP was shown to develop longer and wider branches of endothelial cells compared to organ explants[19]. Interestingly, a long-term proliferating venom gland organoid was established representing several snake species with secretory venom peptides and biological activities[20]. Figure 1 summarizes the organoids which have been derived and well established so far from pluripotent stem cells/adult stem cells and their germ layer identities.
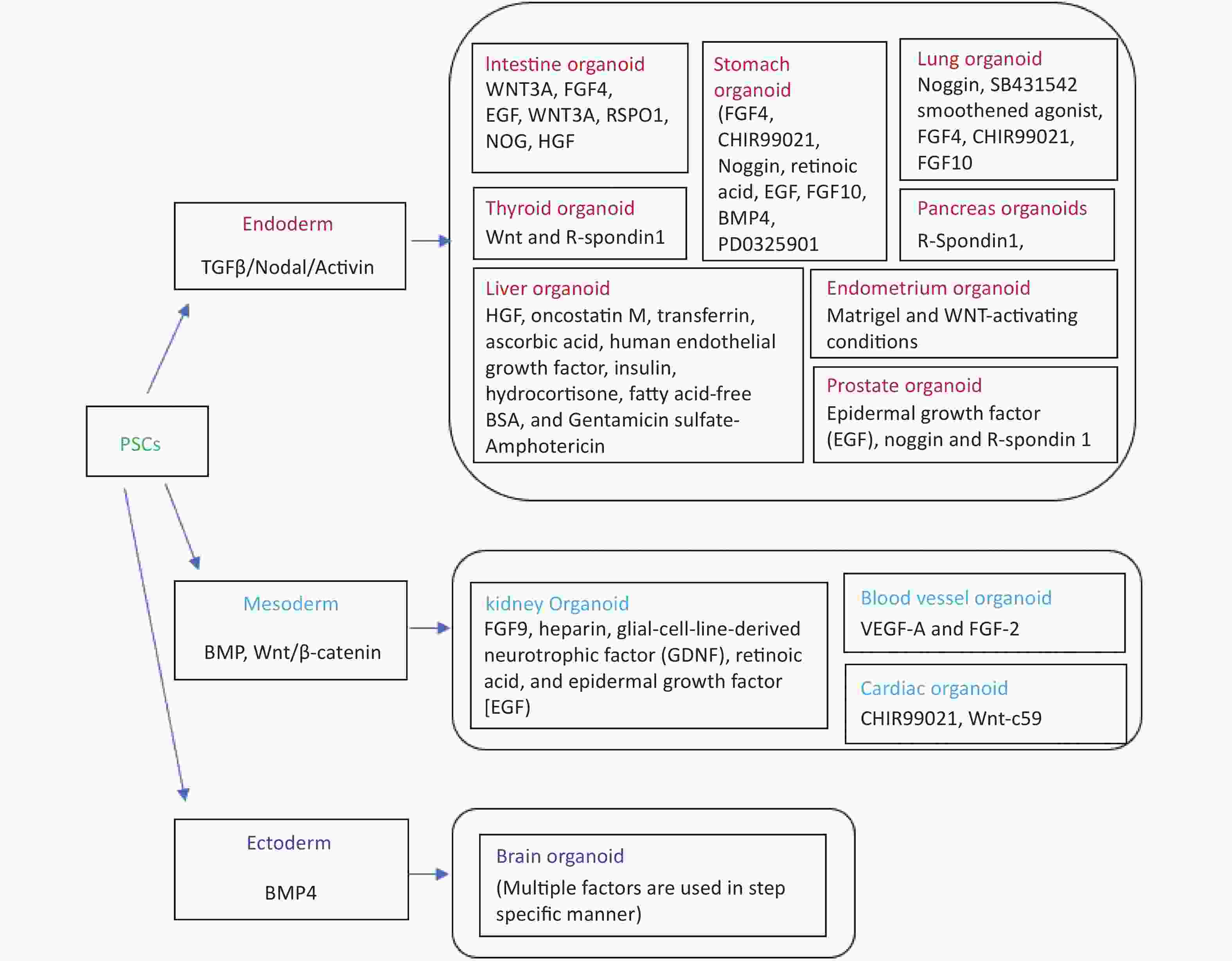
Figure 1. Fundamental chart of various categories of human tissue-specific organoids along with relevant factors. Inducers and/or differentiation factors might vary depending upon on the starting material (viz., iPSC/Adult stem cells/iPSC cell line), while iPSCs refers to “induced pluripotent stem cells”.
-
Intestinal organoids have been used to model different intestinal diseases such as Inflammatory Bowel Disease (Crohn’s disease) and Ulcerative colitis[21-23]. Intestinal organoids can be derived from human primary intestinal tissue as well as pluripotent stem cells. Insulin-like growth factor 1 (IGF-1) and fibroblast growth factor 2 (FGF-2) combination have been shown to increase the plating efficiency of human intestinal organoids, making the CRISPR genome engineering easier and single-cell RNA sequencing has confirmed that refined conditions have improved the native cellular diversity in human small intestinal organoids[24,25]. Recombinant IL-22 targets intestinal stem cells and supports the growth of both mouse and human intestinal organoids. IL-22 induces the phosphorylation of STAT3 in Lgr5(+) intestinal stem cells[26]. The retinoic acid nuclear receptor controls the exit from the regenerative state and drives enterocyte differentiation[27]. Enteroids are in vitro 3D structures that have a very similar cellular composition and architecture to the small intestine. Enteroids can be derived from small intestinal crypt cells which are lgr5+ and in the presence of Epithelial Growth Factor (EGF), R-Spondin, and Noggin, it can be induced to differentiate. Human enteroids development requires the addition of Wnt-3A, SB202190 (p38 inhibitor), and A83-01/SB431542 (TGF-β inhibitor)[28,29]. Recent advances showed expression of ACE2 in differentiated enterocytes readily infected by SARS-CoV-2 serves as a great model to investigate anti-viral therapy[30]. Colonoids have been successfully cultured but their maintenance is very difficult. A method was developed to culture the mouse intestinal crypt organoids and use it for the study of the crypt metabolic profile by the measurement of oxygen consumption, glycolysis, ATP utilization, and the rate of respiration. These crypts can be utilized for the study of their modulation by nutritional and pharmacological interventions[31]. The intestinal epithelium extracted from the 3-day-old chicken embryo was used to generate the three-dimensional culture of intestinal organoids grown on the Matrigel matrix[32]. The propagation of organoids was facilitated by the addition of R-spondin 1, prostaglandin E2, and Noggin and the intestinal enteroids can be generated from the intestine of an adult chicken. The small intestine crypts from chicken were isolated and propagated into intestinal organoids under the external stimulus[4,33]. Prostaglandin E2 promotes the growth of chicken embryo intestinal organoids and induces sustained growth and survival of the epithelial spheroids. The villus crypt obtained from the intestine of the chicken was found to be capable of the formation of enteroids under the influence of chemicals and growth factors. The developed enteroids have all the cell types such as epithelial cells, goblet cells, and enteroendocrine cells[34]. The organoids developed to form the gut epithelium of chicken were shown to migrate in the Matrigel matrix and the static organoids had rotational movements[35].
-
Human gastric organoids can be generated by temporal modulation of Wnt, BMP, EGF, FGF, and retinoic acid signaling pathways in human pluripotent stem cells[36]. Gastric organoids can be derived from normal tissue/stomach cancer and iPSC or ESC. These can be used for gene editing, host-microbe interaction study, omic profiling, biobanking, and high throughput screening. In Helicobacter pylori-infected gastric organoid, CagA protein (of H. pylori) was found to bind to c-Met receptor of organoid epithelial cells and induced cell proliferation. H. pylori infection was also shown to induce the expression of PDL-1 on gastric epithelial cells through Shh signaling pathway. Gastric organoids might help better understand peptic ulcers, gastric cancer, the molecular basis of stomach development, and cell lineage differentiation of gastric cells.
-
It is reported that leucine-rich repeat-containing G protein-coupled receptor 5-expressing and leucine-rich repeat-containing G protein-coupled receptor 6-expressing stem/progenitor cells of taste bud can be reprogrammed into mature taste cells[37]. It was also shown that single-progenitor cells have the potential to develop into all types of mature taste cells and that differentiated taste cells can develop without innervation[38]. The development in the ex-vivo model was shown to mimic the cells of tastebud papillae and it was very similar to the renewal of adult taste stem cells to mature taste cells[39]. Human submandibular gland stem/progenitor cells were propagated into organoids and transplanted into mice after inducing with FGF10 for the development of salivary gland tissues[40].
-
Human pluripotent stem cells were induced to differentiate into human lung organoids and bud tip progenitor organoids under defined factors[41]. Airway organoids were established from broncho-alveolar tissues which could be grown and maintained for a longer time[42]. Some of the important region-specific progenitor cells in the lungs are basal cells (proximal airways), neuroendocrine cells and variant club (bronchioles), alveolar type 2 epithelial cells, and bronchoalveolar stem cells (BASCs) in bronchoalveolar duct junction[43]. These progenitor cells can be utilized for the epithelial regeneration of lungs and differentiated into different types of lung cells in controlled conditions. Krt5-GFP+ basal cells can form tracheospheres when grown for a week[43]. Distal EpCAMhi CD49fpos CD104posCD24low lung epithelial cells can be differentiated into spheres when it is grown along with d EpCAMnegSca-1pos lung mesenchymal cells[44]. Chapman et al. 2011[45] could grow the organoid culture from integrin α6β4+ alveolar epithelial progenitor cells. Kim et al. 2019[46] have developed a protocol to grow lung cancer organoid that produces acinar or large glandular pattern and express TTF-1, napsin-A, and cytokeratin. Lung tumor organoids can be used to establish the model system that can be utilized to study T cell-based therapies[47]. To show the effects of Olaparib, organoids with BRCA2 p.W2619C and BRCA2 p.M965I mutations were created and the former was shown to have lower IC50[46]. Human adult primary bronchial epithelial cells, lung microvascular endothelial cells, and lung fibroblast cells were used together in controlled conditions to generate airway organoids and it was shown that these cells can undergo rapid condensation and self-organization to form the epithelial and endothelial structures that can be maintained for a longer time in culture[48]. Airway organoids are generated after the condensation and it generates an invasive multicellular tubular structure that mimics branching morphogenesis and has expression of YAP/TAZ activation[48].
-
Human pluripotent stem cells derived from hepatic organoids were developed that had the property of self-renewal and were functionally competent[49] iPSCs can be differentiated into different types of hepatocytes such as endothelial cells, Kupffer cells, and cholangiocytes[50]. Mouse and human primary hepatocytes could be grown for multiple months and they retained the morphology as well as gene expression and function[51]. Chemically defined hydrogels were used for the derivation of mouse and hepatic organoids and the development of the organoids was found to be sensitive to stiffness and independent of actin-myosin contractility and required Src family of kinases (SFKs) and yes-associated protein 1 (YAP)[52]. Biopsy-derived human liver organoids were derived without the use of animal components which can be a promising technology in regenerative medicine[52]. Human iPSCs (hiPSCs) were induced to develop into hepatobiliary organoids that mimic hepatogenesis and show the properties such as indocyanine green uptake, accumulation of lipid and glycogen, and secretion of albumin and urea as well drug metabolic activity. The biliary structures were able to show the gamma-glutamyltransferase activity, efflux of rhodamine, and storage of bile acids. The organoids could survive more than 8 weeks when transplanted in mice[53].
-
Mouse PSCs were used to develop thyroid progenitors and could be matured into thyroid follicular organoids[54]. Human and murine thyroid-derived cells were cultured to develop thyroid organoids that were capable of self-renewal and they had shown the characteristics of stem cells and thyroid tissues[55]. Papillary thyroid cancer organoids were shown to preserve histopathological profiles, genetic constitution of the original tumors, patient-specific drug response, and mutations. Estradiol promoted the growth of organoids that was related to estrogen receptor α but independent of ERβ and G protein-coupled ER[56]. Ogumdipeet et al. 2021[55,57] cultured the cells from thyroid gland tissue in a defined thyroid gland medium (TGM) containing Wnt and R-spondin 1 for 7 days which differentiated from the thyrosphere and ultimately aggregated to form thyroid organoids. Recently, a long-term culture system of human fetal thyroid organoid was established which can maintain thyroid lineage and molecular signatures and it can generate functional human thyroid follicles after mice renal transplantation. Forskolin induced cAMP activation plays an important role in follicle maturation and secretion of T4 thyroid hormone[58].
-
Kidney organoids were developed from hiPSCs-derived metanephric mesenchyme and ureteric bud-like cells. Here aldosterone, arginine, and vasopressin were used to promote the differentiation into principal cells and intercalated cells[59]. Kidney organoids were developed from human induced pluripotent cells to study the renin-angiotensin system and shown to have kidney-specific cells such as podocytes, proximal tubule cells, distal tubule cells, stromal cells, and endothelial cells[60]. Renin production by the organoid was also shown to be responding to the parathyroid hormone[60].
Pluripotent stem cell-derived kidney organoid closely resembles nephrogenesis and its differentiation into ureteric bud and metanephric mesenchyme and patterning into nephron structure resemble fetal kidney[61]. Adult stem or progenitor-derived kidney tubuloids provide a better model to study adult kidney tubule renewal and repair. Kidney tubuloids are genetically stable and can be propagated for a longer time and consist of the adult proximal tubule, distal tubule, collecting duct epithelium, and loop of Henle[62]. Kidney organoids and tubuloids can be used for the study of physiology, disease modeling, drug screening, and tissue transplantation. A tumor tubuloids bank has been established to study drug targets[62,63]. Pediatric cancer organoid biobanks of different kidney tumors are developed to study renal cell carcinoma, malignant rhabdoid tumors, congenital mesoblastic nephroma, and Wilms tumor[64]. The methods of metabolomics and transcriptomics were used to study the metabolic dynamics during the differentiation of kidney organoids and the role of important amino acids was shown in the regulation and lineage maturation of the organoid[65].
-
Human blood vessel organoids have been used as a model to study diabetic vasculopathy[66] and hyperglycemia and inflammatory cytokines have been shown to induce the thickening of the vascular basement membrane[67]. The human blood vessel organoid contains different cell types such as pericyte and endothelial cells and it self-assembles into capillary networks with an envelope of basement membrane[68]. Endothelial cells and mural cells require blood vessel function and can be generated from human pluripotent stem cells. Human pluripotent stem cells were induced to develop into blood vessel organoids which mimic microvasculature in morphology, function, and molecular signature[68].
-
hPSC-derived 3D organoids called cerebral organoids were developed having discrete interdependent brain regions and used to model microcephaly[69]. iPSCs from a patient were used to generate whole brain organoids and endothelial cells from the patient itself were used to vascularize the organoid[70]. The organoid models of the dorsal forebrain are populated with a rich diversity of cell types that are normally present in the cerebral cortex. It indicates that the cellular diversity of the brain can be reproduced in the lab[71]. Human cerebral organoid has been demonstrated to model glioblastoma[72]. Vascularized human cortical organoids were developed to model the development of the cortex which can be used to study brain pathology and provide a platform to develop cell therapies and model the nervous system disorder and injuries[73]. Brain organoids were engineered to mimic fetal brain development and used in modeling Zika virus-induced microcephaly[74]. Also, human brain organoids were used to demonstrate the neurotoxic effect of SARS-CoV-2[14,75]. The dorsal-ventral axis of the brain was established by fusing dorsal forebrain and ventral forebrain organoids in a coculture. These organoids can be a good model to recapitulate complex interactions among different parts of the brain[76]. Microfilament engineered cerebral organoids can form neuroectoderm and cortical region and reconstitution of the basement membrane leads to the formation of polarized cortical plate and radial units[77,78]. A human cortical organoid was developed showing dynamic changes in the cell population and electrical activity[79]. A miniaturized multi-well spinning bioreactor was used to generate the forebrain, midbrain, and hypothalamus organoid from the hiPSCs[74,80]. Blood-brain barrier organoids were developed by coculture of endothelial cells, pericytes, and astrocytes by culturing them in low adhesion conditions[81]. The patient-derived glioblastoma cells were grown inside the cerebral organoids to form a model wherein glioblastoma cells get an environment similar to the natural microenvironment to understand the disease pathology and targeted therapy. The glioblastoma cerebral organoid (GLICO) model provides a system mimicking the primary human glioblastoma multiforme in ex vivo and can be useful for high-throughput drug screening[82]. A human pluripotent stem cell-derived cerebral organoid has been developed which can produce cortical neurons and recapitulates cortical development and patient-specific iPSCs have also been used to develop the model for microcephaly[69,83]. A Sonic Hedgehog (SHH) protein gradient was established in forebrain organoids that can enable the formation of the anteroposterior and dorsoventral axis in the human brain organoid[84]. The toxic effect of alcohol on cell signaling pathways and neurons was studied at the cellular, metabolic, and gene expression levels. For instance, alcohol-induced neurotoxicity has been studied in iPSC-derived cerebral organoids to model the fetal alcohol spectrum disorder in the fetus of alcoholic mother[85]. Cerebral organoids developed from the iPSC cells of schizophrenia patients were used to understand the molecular basis of the disease by studying the transcriptomic changes and metabolic changes in these organoid[86]. Various imaging and analysis methods have been developed and image acquisition methods have been improved to study and adapt to the increasing demands of cerebral organoid research data[87]. Primary embryonic stem cells derived from zebrafish and medaka have been shown to form the anterior neural structures[88] and blastula stage cell aggregates could mimic eye developmental stages such as the retinal specification, morphogenesis, and differentiation. The number of cell aggregates, genetic factors, and the changes in morphology was mimicking the in vivo conditions.
-
Studies with human and mouse ES cells have shown that these cells can self-assemble to form the cell aggregates in the 3D suspension culture to form retinal tissues under the low serum concentration[89-91]. Retinal organoids from human pluripotent stem cells were derived and used to investigate retinal development, retinal disease modeling, and therapeutic development[92]. Light-sensitive retinal organoids were developed that had multiple nuclear and synaptic layers. The cell types of the retinal organoids developed in vitro demonstrated stable structure at a rate similar to the human retinal development in vivo[93]. Cone-rich retinal organoids were developed and their generation, transcriptome profiling, and functional validation demonstrated their resemblance to macula/fove[94]. The reproducibility and quicker development of fish-derived organoids along with advanced gene-editing technology makes it convenient to understand the development and diseases in animals. Fish-derived organoids can also help in understanding the effects of the physical environment and chemical factors on the morphogenesis and differentiation of embryonic tissues. Lucie Zilovafish’s primary embryonic stem cells can self-assemble to form retinal tissue which mimics the in-vivo early eye development[88].
-
Protein C receptor-positive (Procr+) cells, present in the islet of the pancreas in an undifferentiated state and have epithelial-to-mesenchymal transition characteristics can differentiate to form all four types (α, β, δ, and PP cells) of endocrine cells. These Procr+ cells can form islet-like organoids and can be maintained for a longer time on serial passaging[95]. Pancreatic organoid models have been established from tumors and biopsies and shown to survive cryopreservation. Pancreatic organoids could mimic the pancreatic duct and orthotopically transplanted pancreatic cancer organoid shows similarity in disease progression and metastasis[96]. These organoids can be used to study alterations in genes and changes in specific pathways responsible for cancer progression[96].
-
Novel and promising endometrium organoid was developed from mouse and human endometrium tissue which mimics the molecular and histologic architecture of the respective tissues[97]. The developed organoid responded to estrogen and progesterone and human endometrial organoids also mimic the menstrual cycle on hormonal treatment[97]. Human endometrial organoids were developed to study the embryo implantation process and these organoids were able to differentiate into pinopodes, large cytoplasmic apical protrusions[98]. 3D culture of normal and deciduous endometrium was developed which could respond to reproductive hormones and showed the early characteristics of pregnancy[99]. Mammary tumor organoids have been developed from mouse models engineered for the deficiency of BRCA1 and BRCA2[100].
-
Prostate organoids are generally responsive to androgen and it mimics the prostate epithelium[101]. Prostate organoids can be generated from adult stem cells and pluripotent cells from mice, human benign, and malignant tumors and can be manipulated at the molecular level using CRISPR/Cas9 and shRNA systems[102]. In the mouse model, luminal C cells located at distal prostate invagination tips were shown to have a higher potential to form the organoid and regeneration of the prostate epithelial duct and it forms the distal prostate luminal lineage by self-renewal and differentiation[103]. A single luminal stem/progenitor cell was shown to generate 3D prostate organoids and it had shown structural similarity with basal cells and luminal cells, long-term expansion, and functional androgen receptor signalling[104]. Luminal cells are favoured for organoid generation and form basal cells in culture[105]. Prostate cancer cell lines LNCaP and C4-2B were used to form organoids with glandular structure and the organoid has androgen receptor-positive adenocarcinoma cells but p63-positive basal cells were absent[106]. Mouse and human bladder organoids have been cultured efficiently and genetically manipulated[107]. These can be established from the cancer tissues, biopsies, and passaged for a longer period[108].
-
Testicular organoids have been used to study cell-cell interactions, germ-cell niches, disease modelling, and testicular cancer. Testicular organoids consisting of germ cells, Sertoli cells, Leydig cells, and peritubular myoid cells were developed from pigs, mice, macaque, and humans[109]. Sertoli-like cells[110] and Leydig-like cells[107,111] have been derived from pluripotent cells. iPSCs can be induced to form Sertoli cells, Leydig cells, germ cells, peritubular cells, and endothelial cells, and a combination of these can give a functional testis[112]. Microwell culture system was used to generate thousands of homogeneous porcine testicular organoids which had testis-specific architecture and cell associations[113]. Moreover, testicular cells isolated from testes tissue were cultured in the presence or absence of a scaffold which further led to the development of testicular organoids[114].
-
Cardiac organoids were developed using human-induced stem cell-derived cardiomyocytes[115]. The derived embryoid bodies were cast on rat-engineered heart tissue (EHT) to check the human induced pluripotent stem cell-derived cardiomyocytes (hiPSC-CM) embryoid body (EB) controlled beating activity and it was found that EB-controlled beating activity was a regular beating. Action potential and calcium transient transmission from EB were directly related to rat EHT. Heart forming organoid (HFOs) was generated from human pluripotent stem cells aggregated by directed cardiac differentiation through modulation of the Wnt pathway and small molecules[116]. HFOs structures had a myocardial layer that was lined by endocardial-like cells and encircled by septum transversum-like Anlagen. The composition of HFOs closely mimics early native heart Anlagen. Human cardiac organoids have been generated to model myocardial infarction and screening of drug toxicity[117].
-
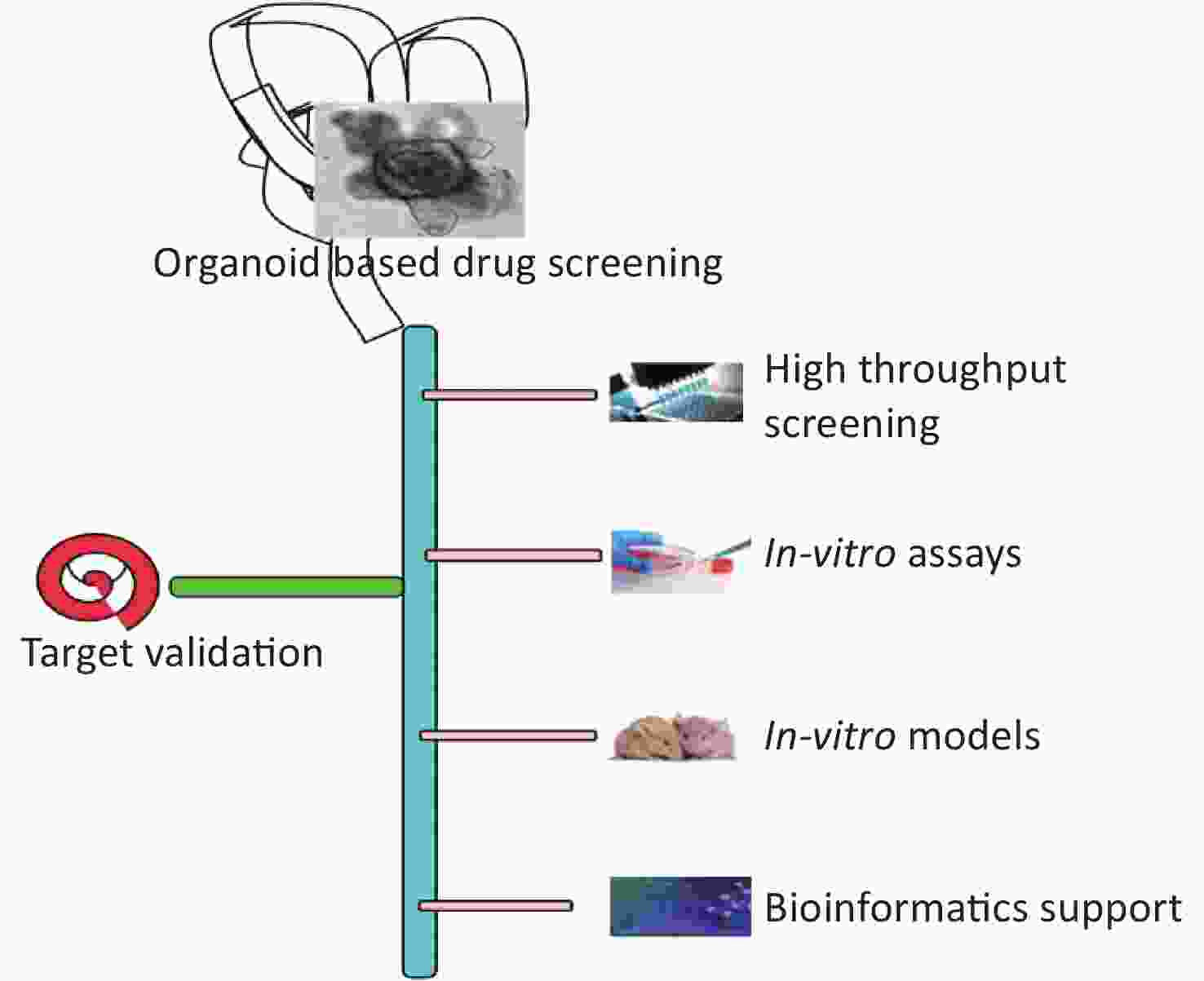
Most of the drugs failed in clinical trials in phase II and phase III from 2013 to 2015[118], because of the effectiveness and safety of the candidate drugs. Hence, the development of a model system that can provide a better estimate of effectiveness and safety is required which can minimize the cost of drug development as well as enhance the rate of success. Patient-derived organoids as well as adult stem cell/iPSC-derived organoids have played a pivotal role as a drug screening model in the last decade. Organoids can recapitulate the morphological, structural, and functional characteristics of the body organs, and hence act as a reliable and better model compared to the animal models and cell lines[119]. Organoids can help eliminate the biases of modern medicine because of the differences in the genetics of patients, anomalies in the prediction of outcomes, and the longer time taken in the novel drug development. Organoids developed from a specific diseased sample or a specific individual can improve the treatment and act as powerful precision therapy tools. Organoid biobank can be used for high throughput screening (Figure 2). Drug screening for cystic fibrosis using organoids has resulted in two new cystic fibrosis drugs and the model has also identified the unique mutations that can benefit from the unique set of treatment options[120]. Organoid models can not only help in identifying new drugs but also classify the patients who can benefit or not benefit from the particular treatment protocol. Organoids can be used in the future to partially or completely replace animal models for drug testing and development. Organoids grown from the rectal biopsy of two patients had shown a positive response to the drug ivacaftor and the patients had shown a positive response to treatment with the drug[121]. Organoids derived from primary human pancreatic ductal adenocarcinoma have been used to develop new drugs against the disease[122]. Organoids were derived from intrahepatic cholangiocarcinoma, gall bladder cancer, and neuroendocrine carcinoma of the ampulla of Vater and it was shown on compound library screening that antifungal drugs amorolfine and fenticonazole can suppress the organoids derived from biliary tract carcinomas with minimal toxicity on normal biliary epithelial cells[123]. Organoid studies can be a useful preclinical model for pharmacodynamic profiling and drug screening platforms. This way, the organoid research domain can help to improve the efficacy and specificity of drugs; and to find out novel drug targets. Seahorse XF analysis was optimized and used to investigate the bioenergetics of organoids and characterize the responses to drugs, and gene knockdowns and track the metabolic changes in specific cell types[124]. One of the best examples of organoid uses for drug screening is the Forskolin-induced Swelling in Intestinal Organoids. Cystic fibrosis transmembrane conductance regulator (CFTR)-modulating drugs correct surface expression and/or function of the mutant CFTR channel in subjects with cystic fibrosis (CF) but the major challenge is the identification of subject that can benefit from the drug as CFTR gene shows heterogeneity and other factors might be involved in drug efficacy. To address this problem, an epithelial organoid is developed from the rectal biopsies of patient in organoid growth medium and drug screening is done to identify the correct drug for the individual patient. Forskolin-induced swelling is monitored in organoid using calcein green. In vitro extent of swell response is related to the clinical response to the treatment in the patient. The assay is a cost-effective approach for identification of patients who will respond to the drug independent of their CFTR mutations. This assay can also help in development of future new CFTR modulators[96,125].
-
In this era, the wide application of various machine learning (ML) techniques and/or the employment of deep learning (DL) algorithms in the sphere of healthcare services has accelerated the computational procedure of detection along with the commencement of early diagnosis of different diseases/disorders to enhance the survival rate of human beings as well as prolong the lifetime of the patients. Several ML and DL methods including Logistic Regression (LR), Random Forest (RF), Support Vector Machine (SVM), Decision Tree (DT), K-Nearest Neighbor (KNN), Convolutional Neural Network (CNN) are rapidly deployed to detect the presence of disease and diagnose the disease for shielding the human organs. Table 1 explores some related research works regarding the usage of the principles of ML and DL for the diseases responsible for affecting various human organs.
Table 1. Computational approaches of ML and DL for detecting and diagnosing disease
Employed ML approaches Associated organ Advantages Future challenges Reference Neural Networks, Logistic Regression Head and Neck Effective diagnosis of head and neck cancer-affected patients. Producing better diagnostic accuracy considering smoking history along with the perineural invasion of patients. [126] DT, RF Heart Heart disease detection with high accuracy. A diverse mixture of ML techniques to predict heart diseases with better accuracy. [127] KNN Liver Early and effective prediction of chronic liver infections. Development of a model for obtaining more accuracy to detect chronic liver infections. [128] DL Pancreas DL-based Nucleus Classification of images for predicting pancreas cancer with high accuracy. Employment of a larger dataset to produce better accuracy for predicting pancreas cancer. [129] Bayes classifiers, SVM Stomach Creation of a model by dint of ML approaches to initially detect stomach cancer. Avoid the statistical assumptions and consideration of larger datasets to detect stomach cancer with better accuracy. [130] LR, RF Kidney Effective diagnosis of chronic kidney diseases (CKD) with superior accuracy. Improvement in diagnosis of CKD with better accuracy considering more categories of severe CKD and more complex data samples of patients. [131] CNN Lung Lung cancer prediction from data of CT images. Consideration of the smoking history of patients. [132] Shallow convolutional
neural networkBreast Breast cancer identification with higher accuracy. This works compared with only limited CNN and deep learning methods, while these number could be extended in future for better prospective and better efficiency. [133] DL Brain Effective diagnosis of brain cancer by highly efficient deep learning model depending upon CNN for glioblastoma multiforme (GBM) subtype detection
with superior accuracy.This work could be extended to utilize in other human cancers for designing DL-oriented diagnostic methods through more high-throughput experimental data profile. [134] DL/CNN Brain Explanation-driven DL model through the use of CNN, local interpretable model-agnostic explanation (LIME) as well as Shapley additive explanation (SHAP) to predict discrete subtypes of brain tumours from MRI image dataset. Classification techniques with higher accuracy and better optimizer could be applied and superimposed on proposed technique. [135] The current methods of manufacturing organoids are yet to demonstrate consistency and robustness. ML can help design and test organoids utilizing computers rather than the traditional lab method. Scalable production of high-grade organoids can be possible with the help of ML/DL. Mechano-transduction pathways can be applied to regulate the manufacturing of organoids and ML methods can help in the identification of key signature cytoskeleton states associated with the phenotype of the organoid.
-
Organoid technology provides a great opportunity for the study of organogenesis, cell differentiation, cell-cell interactions, and physiological functions but it has some inherent limitations. Organoid culture doesn’t have mesenchymal cells, immune cells, and vasculature hence it can’t exactly mimic the organs or tissues of the body but the coculture can improve the architecture of the organoids[136]. Organoids can’t model multi-organ pathologies and it recapitulates only part of the entire body hence studies in animal models should be used to complement the organoid studies[137]. Organoid studies can’t mimic the developmental axis of the organ and lack a functional immune system and complete physiological conditions. Protocols and culture conditions for organoid needs to be well defined to enhance reproducibility and cell types and structures in organoid should be compared with the respective organ. Maintenance of organoid and derived structures need to be standardized for maintenance for a longer time. The establishment of a tissue bank and distribution hub can provide uniformity in the work performed at different labs. The results obtained from organoids might vary depending on the source of initiating cells (such as cell lines, fetal or adult primary stem cells, or iPSC cells), culture media, and growth conditions used for iPSC or organoid maintenance. The reproducibility of the organoid varies according to the type and complexity of the assay, source of initiating cells, and hence proper quality control is required for minimizing the variability[138]. In the organoids structure of the colon, the function of cystic fibrosis transmembrane conductance regulator (CFTR) protein and its rescue by CFTR modulators can be quantified using the forskolin-induced swelling assay and it has been widely used to screen drugs or find out the patient-specific drugs[121,125]. Similar kinds of organoid-based assay/readout methods for other human organs are lacking.
Currently organoid technology is having difficulty in homogenization and lacks the scalability for high throughput screens and large-scale cell therapy. Improvement in cell culture methods and ECM support may produce more scalable and reproducible organoids. For cell transplantation therapy using organoids, Matrigel scaffold needs to be replaced by a more biocompatible material.
For the stable scaling up of organoids and their reproducibility, we need to ensure a microbe free cell culture form cell production to validation, development of organoid storage and delivery methods, and methods to validate the safety and efficacy of the in vivo organoids.
Current protocols have limitations of readability as optical monitoring provides unclear information about functionality of the organoids. The readouts of metabolites, secreted factors are variable because of the organoid formation methods. The integration of miniature biosensors into developing organoids can solve this problem but it needs to undergo intensive research and development to achieve that goal. Readouts can be improved by controlled imaging and automated analysis, insertion of miniaturized electrochemical probes, and parallelization and high throughput organoid generation. Some of the other limitations of organoid technology are limited level of maturity and functions of organoids, accessibility of organoids because of short life span, heterogeneity such as variation in organoid formation efficiency, and differences in end point morphology and function.
Organoid technology also raises several ethical issues such as sources of stem cells, informed consent and privacy of cell donors, moral and legal conditions associated with organoids use, gene editing and chimera formation, commercialization, equity in results of treatment and commercialization of the organoids. Misuse and dual use of stored organoids can also have ethical implications as current guidelines are not very clear about organoid uses.
-
Human organoids have tremendous potential in translational applications and their genetic manipulation, and co-culture with microbes and parasites have opened new models to study disease mechanisms. Human organoids also provide a great opportunity to validate the results generated from the studies on animal models. The fast developments in the field of organoid research have transformed the ways experiments are done in the lab. This survey article demonstrates the recent trends in organoid research and highlights the important growth conditions and determining factors that help the in vitro development of organoids and the possible uses of organoids in disease characterization. Finally, the computational frameworks (viz., machine learning, deep learning, artificial intelligence, and soft computing approaches) related to this study are also amended to this survey paper for better exploration of related diseases.
doi: 10.3967/bes2023.083
A Comprehensive View on the Progress of Organoid Research with an Emphasis on its Relevance to Disease Characterization
-
Wrote the manuscript: Chandra Kishore, Vaishali Ji, Ayan Mukherji, and Sinthia Roy Banerjee; Searched and read papers: Chandra Kishore, Vaishali Ji, and Ayan Mukherji; Revised the manuscript: Chandra Kishore, Saurav Mallik, Soumen Kumar Pati, Namrata Tomar, and Soumadip Ghosh; Supervised the manuscript: Saurav Mallik, Aimin Li, and Raza Ali Naqvi.
None of the authors have competing interests to declare.
注释:1) AUTHOR CONTRIBUTIONS: 2) COMPETING INTERESTS: -
Figure 1. Fundamental chart of various categories of human tissue-specific organoids along with relevant factors. Inducers and/or differentiation factors might vary depending upon on the starting material (viz., iPSC/Adult stem cells/iPSC cell line), while iPSCs refers to “induced pluripotent stem cells”.
Table 1. Computational approaches of ML and DL for detecting and diagnosing disease
Employed ML approaches Associated organ Advantages Future challenges Reference Neural Networks, Logistic Regression Head and Neck Effective diagnosis of head and neck cancer-affected patients. Producing better diagnostic accuracy considering smoking history along with the perineural invasion of patients. [126] DT, RF Heart Heart disease detection with high accuracy. A diverse mixture of ML techniques to predict heart diseases with better accuracy. [127] KNN Liver Early and effective prediction of chronic liver infections. Development of a model for obtaining more accuracy to detect chronic liver infections. [128] DL Pancreas DL-based Nucleus Classification of images for predicting pancreas cancer with high accuracy. Employment of a larger dataset to produce better accuracy for predicting pancreas cancer. [129] Bayes classifiers, SVM Stomach Creation of a model by dint of ML approaches to initially detect stomach cancer. Avoid the statistical assumptions and consideration of larger datasets to detect stomach cancer with better accuracy. [130] LR, RF Kidney Effective diagnosis of chronic kidney diseases (CKD) with superior accuracy. Improvement in diagnosis of CKD with better accuracy considering more categories of severe CKD and more complex data samples of patients. [131] CNN Lung Lung cancer prediction from data of CT images. Consideration of the smoking history of patients. [132] Shallow convolutional
neural networkBreast Breast cancer identification with higher accuracy. This works compared with only limited CNN and deep learning methods, while these number could be extended in future for better prospective and better efficiency. [133] DL Brain Effective diagnosis of brain cancer by highly efficient deep learning model depending upon CNN for glioblastoma multiforme (GBM) subtype detection
with superior accuracy.This work could be extended to utilize in other human cancers for designing DL-oriented diagnostic methods through more high-throughput experimental data profile. [134] DL/CNN Brain Explanation-driven DL model through the use of CNN, local interpretable model-agnostic explanation (LIME) as well as Shapley additive explanation (SHAP) to predict discrete subtypes of brain tumours from MRI image dataset. Classification techniques with higher accuracy and better optimizer could be applied and superimposed on proposed technique. [135] -
[1] Xu ZY, Huang JJ, Liu Y, et al. Current knowledge on the multiform reconstitution of intestinal stem cell niche. World J Stem Cells, 2021; 13, 1564−79. doi: 10.4252/wjsc.v13.i10.1564 [2] Ji V, Kishore C. The emerging roles of srGAPs in cancer. Mol Biol Rep, 2022; 49, 755−9. doi: 10.1007/s11033-021-06872-2 [3] McCauley HA, Wells JM. Pluripotent stem cell-derived organoids: using principles of developmental biology to grow human tissues in a dish. Development, 2017; 144, 958−62. doi: 10.1242/dev.140731 [4] Li J, Li J Jr, Zhang SY, et al. Culture and characterization of chicken small intestinal crypts. Poult Sci, 2018; 97, 1536−43. doi: 10.3382/ps/pey010 [5] Gao Y, Chen XY, Tian TR, et al. A lysosome-activated tetrahedral nanobox for encapsulated siRNA delivery. Adv Mater, 2022; 34, e2201731. doi: 10.1002/adma.202201731 [6] Zhang TX, Zhou M, Xiao DX, et al. Myelosuppression alleviation and hematopoietic regeneration by tetrahedral-framework nucleic-acid nanostructures functionalized with osteogenic growth peptide. Adv Sci (Weinh), 2022; 9, e2202058. doi: 10.1002/advs.202202058 [7] Zhang T, Tian TR, Zhou RH, et al. Design, fabrication and applications of tetrahedral DNA nanostructure-based multifunctional complexes in drug delivery and biomedical treatment. Nat Protoc, 2020; 15, 2728−57. doi: 10.1038/s41596-020-0355-z [8] Zhou J, Li C, Liu XJ, et al. Infection of bat and human intestinal organoids by SARS-CoV-2. Nat Med, 2020; 26, 1077−83. doi: 10.1038/s41591-020-0912-6 [9] Lamers MM, Beumer J, van der Vaart J, et al. SARS-CoV-2 productively infects human gut enterocytes. Science, 2020; 369, 50−4. doi: 10.1126/science.abc1669 [10] Zhao XY, Li C, Liu XJ, et al. Human intestinal organoids recapitulate enteric infections of enterovirus and coronavirus. Stem Cell Rep, 2021; 16, 493−504. doi: 10.1016/j.stemcr.2021.02.009 [11] Takayama K. In vitro and animal models for SARS-CoV-2 research. Trends Pharmacol Sci, 2020; 41, 513−7. doi: 10.1016/j.tips.2020.05.005 [12] Chen D, Tan YW, Li ZC, et al. Organoid cultures derived from patients with papillary thyroid cancer. J Clin Endocrinol Metab, 2021; 106, 1410−26. doi: 10.1210/clinem/dgab020 [13] Allison SJ. SARS-CoV-2 infection of kidney organoids prevented with soluble human ACE2. Nat Rev Nephrol, 2020; 16, 316. [14] Ramani A, Müller L, Ostermann PN, et al. SARS-CoV-2 targets neurons of 3D human brain organoids. EMBO J, 2020; 39, e106230. doi: 10.15252/embj.2020106230 [15] Mills RJ, Humphrey SJ, Fortuna PRJ, et al. BET inhibition blocks inflammation-induced cardiac dysfunction and SARS-CoV-2 infection. Cell, 2021; 184, 2167−82.e22. doi: 10.1016/j.cell.2021.03.026 [16] Peng LY, Zhou YH, Xu WT, et al. Generation of stable induced pluripotent stem-like cells from adult zebra fish fibroblasts. Int J Biol Sci, 2019; 15, 2340−9. doi: 10.7150/ijbs.34010 [17] Ibrahim M, Xie B, Richardson MK. The growth of endothelial-like cells in zebrafish embryoid body culture. Exp Cell Res, 2020; 392, 112032. doi: 10.1016/j.yexcr.2020.112032 [18] Schauer A, Pinheiro D, Hauschild R, et al. Zebrafish embryonic explants undergo genetically encoded self-assembly. Elife, 2020; 9, e55190. doi: 10.7554/eLife.55190 [19] Ibrahim M, Richardson MK. In vitro development of zebrafish vascular networks. Reprod Toxicol, 2017; 70, 102−15. doi: 10.1016/j.reprotox.2017.02.008 [20] Post Y, Puschhof J, Beumer J, et al. Snake venom gland organoids. Cell, 2020; 180, 233−47. doi: 10.1016/j.cell.2019.11.038 [21] Angus HCK, Butt AG, Schultz M, et al. Intestinal organoids as a tool for inflammatory bowel disease research. Front Med (Lausanne), 2020; 6, 334. [22] Kishore C. Epigenetic regulation and promising therapies in colorectal cancer. Curr Mol Pharmacol, 2021; 14, 838−52. doi: 10.2174/1874467214666210126105345 [23] Kishore C, Bhadra P. Current advancements and future perspectives of immunotherapy in colorectal cancer research. Eur J Pharmacol, 2021; 893, 173819. doi: 10.1016/j.ejphar.2020.173819 [24] Fujii M, Matano M, Toshimitsu K, et al. Human intestinal organoids maintain self-renewal capacity and cellular diversity in niche-inspired culture condition. Cell Stem Cell, 2018; 23, 787−93.e6. doi: 10.1016/j.stem.2018.11.016 [25] Kishore C, Sundaram S, Karunagaran D. Vitamin K3 (menadione) suppresses epithelial-mesenchymal-transition and Wnt signaling pathway in human colorectal cancer cells. Chem Biol Interact, 2019; 309, 108725. doi: 10.1016/j.cbi.2019.108725 [26] Lindemans CA, Calafiore M, Mertelsmann AM, et al. Interleukin-22 promotes intestinal-stem-cell-mediated epithelial regeneration. Nature, 2015; 528, 560−4. doi: 10.1038/nature16460 [27] Lukonin I, Serra D, Challet Meylan L, et al. Phenotypic landscape of intestinal organoid regeneration. Nature, 2020; 586, 275−80. doi: 10.1038/s41586-020-2776-9 [28] Holmberg FE, Seidelin JB, Yin XL, et al. Culturing human intestinal stem cells for regenerative applications in the treatment of inflammatory bowel disease. EMBO Mol Med, 2017; 9, 558−70. doi: 10.15252/emmm.201607260 [29] Kishore C, Karunagaran D. Non-coding RNAs as emerging regulators and biomarkers in colorectal cancer. Mol Cell Biochem, 2022; 477, 1817−28. doi: 10.1007/s11010-022-04412-5 [30] Gheblawi M, Wang K, Viveiros A, et al. Angiotensin-converting enzyme 2: SARS-CoV-2 receptor and regulator of the renin-angiotensin system. Circ Res, 2020; 126, 1456−74. doi: 10.1161/CIRCRESAHA.120.317015 [31] Bas T, Augenlicht LH. Real time analysis of metabolic profile in ex vivo mouse intestinal crypt organoid cultures. J Vis Exp, 2014; e52026. [32] Pierzchalska M, Panek M, Grabacka M. The migration and fusion events related to ROCK activity strongly influence the morphology of chicken embryo intestinal organoids. Protoplasma, 2019; 256, 575−81. doi: 10.1007/s00709-018-1312-3 [33] Ahmad AA, Wang Y, Gracz AD, et al. Optimization of 3-D organotypic primary colonic cultures for organ-on-chip applications. 2014; J Biol Eng. [34] Acharya M, Arsi K, Donoghue AM, et al. Production and characterization of avian crypt-villus enteroids and the effect of chemicals. BMC Vet Res, 2020; 16, 179. doi: 10.1186/s12917-020-02397-1 [35] Pierzchalska M, Panek M, Czyrnek M, et al. The three-dimensional culture of epithelial organoids derived from embryonic chicken intestine. In: Turksen K. Organoids. Humana. 2019, 135-44. [36] McCracken KW, Catá EM, Crawford CM, et al. Modelling human development and disease in pluripotent stem-cell-derived gastric organoids. Nature, 2014; 516, 400−4. doi: 10.1038/nature13863 [37] Arioka Y, Ito H, Hirata A, et al. Behavior of leucine-rich repeat-containing G-protein coupled receptor 5-expressing cells in the reprogramming process. Stem Cell Res, 2017; 20, 1−9. doi: 10.1016/j.scr.2017.01.012 [38] Barlow LA. Progress and renewal in gustation: new insights into taste bud development. Development, 2015; 142, 3620−9. doi: 10.1242/dev.120394 [39] Ren WW, Lewandowski BC, Watson J, et al. Single Lgr5- or Lgr6-expressing taste stem/progenitor cells generate taste bud cells ex vivo. Proc Natl Acad Sci USA, 2014; 111, 16401−6. doi: 10.1073/pnas.1409064111 [40] Sui Y, Zhang SQ, Li YL, et al. Generation of functional salivary gland tissue from human submandibular gland stem/progenitor cells. Stem Cell Res Ther, 2020; 11, 127. doi: 10.1186/s13287-020-01628-4 [41] Miller AJ, Dye BR, Ferrer-Torres D, et al. Generation of lung organoids from human pluripotent stem cells in vitro. Nat Protoc, 2019; 14, 518−40. doi: 10.1038/s41596-018-0104-8 [42] Sachs N, Papaspyropoulos A, Zomer-van Ommen DD, et al. Long-term expanding human airway organoids for disease modeling. EMBO J, 2019; 38, e100300. doi: 10.15252/embj.2018100300 [43] Rock JR, Onaitis MW, Rawlins EL, et al. Basal cells as stem cells of the mouse trachea and human airway epithelium. Proc Natl Acad Sci USA, 2009; 106, 12771−5. doi: 10.1073/pnas.0906850106 [44] McQualter JL, Yuen K, Williams B, et al. Evidence of an epithelial stem/progenitor cell hierarchy in the adult mouse lung. Proc Natl Acad Sci USA, 2010; 107, 1414−9. doi: 10.1073/pnas.0909207107 [45] Chapman HA, Li XP, Alexander JP, et al. Integrin α6β4 identifies an adult distal lung epithelial population with regenerative potential in mice. J Clin Invest, 2011; 121, 2855−62. doi: 10.1172/JCI57673 [46] Kim M, Mun H, Sung CO, et al. Patient-derived lung cancer organoids as in vitro cancer models for therapeutic screening. Nat Commun, 2019; 10, 3991. doi: 10.1038/s41467-019-11867-6 [47] Li YS, Chan JWY, Lau RWH, et al. Organoids in Lung Cancer Management. Front Surg, 2021; 8, 753801. doi: 10.3389/fsurg.2021.753801 [48] Tan Q, Choi KM, Sicard D, et al. Human Airway Organoid Engineering as a Step toward Lung Regeneration and Disease Modeling. Biomaterials, 2017; 113, 118−32. doi: 10.1016/j.biomaterials.2016.10.046 [49] Mun SJ, Ryu JS, Lee MO, et al. Generation of expandable human pluripotent stem cell-derived hepatocyte-like liver organoids. J Hepatol, 2019; 71, 970−85. doi: 10.1016/j.jhep.2019.06.030 [50] Olgasi C, Cucci A, Follenzi A. iPSC-derived liver organoids: a journey from drug screening, to disease modeling, arriving to regenerative medicine. Int J Mol Sci, 2020; 21, 6215. doi: 10.3390/ijms21176215 [51] Hu HL, Gehart H, Artegiani B, et al. Long-term expansion of functional mouse and human hepatocytes as 3D organoids. Cell, 2018; 175, 1591−606.e19. doi: 10.1016/j.cell.2018.11.013 [52] Sorrentino G, Rezakhani S, Yildiz E, et al. Mechano-modulatory synthetic niches for liver organoid derivation. Nat Commun, 2020; 11, 3416. doi: 10.1038/s41467-020-17161-0 [53] Wu FF, Wu D, Ren Y, et al. Generation of hepatobiliary organoids from human induced pluripotent stem cells. J Hepatol, 2019; 70, 1145−58. doi: 10.1016/j.jhep.2018.12.028 [54] Kurmann AA, Serra M, Hawkins F, et al. Regeneration of thyroid function by transplantation of differentiated pluripotent stem cells. Cell Stem Cell, 2015; 17, 527−42. doi: 10.1016/j.stem.2015.09.004 [55] Ogundipe VML, Groen AH, Hosper N, et al. Generation and differentiation of adult tissue-derived human thyroid organoids. Stem Cell Rep, 2021; 16, 913−25. doi: 10.1016/j.stemcr.2021.02.011 [56] Chen KG, Park K, Spence JR. Studying SARS-CoV-2 infectivity and therapeutic responses with complex organoids. Nat Cell Biol, 2021; 23, 822−33. doi: 10.1038/s41556-021-00721-x [57] Arufe MC, Lu M, Lin RY. Differentiation of murine embryonic stem cells to thyrocytes requires insulin and insulin-like growth factor-1. Biochem. Biophys. Res. Commun. 2009;264–70. [58] Liang JQ, Qian J, Yang L, et al. Modeling human thyroid development by fetal tissue-derived organoid culture. Adv Sci, 2022; 9 e2105568. [59] Uchimura K, Wu HJ, Yoshimura Y, et al. Human pluripotent stem cell-derived kidney organoids with improved collecting duct maturation and injury modeling. Cell Rep, 2020; 33, 108514. doi: 10.1016/j.celrep.2020.108514 [60] Shankar AS, Du ZY, Mora HT, et al. Human kidney organoids produce functional renin. Kidney Int, 2021; 99, 134−47. doi: 10.1016/j.kint.2020.08.008 [61] Garreta E, Nauryzgaliyeva Z, Montserrat N. Human induced pluripotent stem cell-derived kidney organoids toward clinical implementations. Curr Opin Biomed Eng, 2021; 20, 100346. doi: 10.1016/j.cobme.2021.100346 [62] Yousef Yengej FA, Jansen J, Rookmaaker MB, et al. Kidney organoids and tubuloids. Cells, 2020; 9, 1326. doi: 10.3390/cells9061326 [63] Clevers H. Modeling Development and Disease with Organoids. Cell, 2016; 1586–97. [64] Calandrini C, Schutgens F, Oka R, et al. An organoid biobank for childhood kidney cancers that captures disease and tissue heterogeneity. Nat Commun, 2020; 11, 1310. doi: 10.1038/s41467-020-15155-6 [65] Wang QZ, Xiong YC, Zhang S, et al. The dynamics of metabolic characterization in iPSC-derived kidney organoid differentiation via a comparative omics approach. Front Genet, 2021; 12, 632810. doi: 10.3389/fgene.2021.632810 [66] Wimmer RA, Leopoldi A, Aichinger M, et al. Human blood vessel organoids as a model of diabetic vasculopathy. Nature, 2019; 565, 505−10. doi: 10.1038/s41586-018-0858-8 [67] Rask-Madsen C, King GL. Vascular complications of diabetes: mechanisms of injury and protective factors. Cell Metab, 2013; 17, 20−33. doi: 10.1016/j.cmet.2012.11.012 [68] Wimmer RA, Leopoldi A, Aichinger M, et al. Generation of blood vessel organoids from human pluripotent stem cells. Nat Protoc, 2019; 14, 3082−100. doi: 10.1038/s41596-019-0213-z [69] Lancaster MA, Renner M, Martin CA, et al. Cerebral organoids model human brain development and microcephaly. Nature, 2013; 501, 373−9. doi: 10.1038/nature12517 [70] Pham MT, Pollock KM, Rose MD, et al. Generation of human vascularized brain organoids. NeuroReport, 2018; 29, 588−93. doi: 10.1097/WNR.0000000000001014 [71] Velasco S, Kedaigle AJ, Simmons SK, et al. Individual brain organoids reproducibly form cell diversity of the human cerebral cortex. Nature, 2019; 570, 523−7. doi: 10.1038/s41586-019-1289-x [72] Ogawa J, Pao GM, Shokhirev MN, et al. Glioblastoma model using human cerebral organoids. Cell Rep, 2018; 23, 1220−9. doi: 10.1016/j.celrep.2018.03.105 [73] Shi YC, Sun L, Wang MD, et al. Vascularized human cortical organoids (vOrganoids) model cortical development in vivo. PLoS Biol, 2020; 18, e3000705. doi: 10.1371/journal.pbio.3000705 [74] Qian XY, Jacob F, Song MM, et al. Generation of human brain region-specific organoids using a miniaturized spinning bioreactor. Nat Protoc, 2018; 13, 565−80. doi: 10.1038/nprot.2017.152 [75] Alonso AD, Di Clerico J, Li B, et al. Phosphorylation of tau at Thr212, Thr231, and Ser262 combined causes neurodegeneration. 2010; J Biol Chem. 30851–60. [76] Bagley JA, Reumann D, Bian S, et al. Fused cerebral organoids model interactions between brain regions. Nat Methods, 2017; 14, 743−51. doi: 10.1038/nmeth.4304 [77] Lancaster MA, Corsini NS, Wolfinger S, et al. Guided self-organization and cortical plate formation in human brain organoids. Nat Biotechnol, 2017; 35, 659−66. doi: 10.1038/nbt.3906 [78] Kishore C, Bhadra P. Targeting brain cancer cells by nanorobot, a promising nanovehicle: new challenges and future perspectives. CNS Neurol Disord Drug Targets, 2021; 20, 531−9. doi: 10.2174/1871527320666210526154801 [79] Trujillo CA, Gao R, Negraes PD, et al. Complex oscillatory waves emerging from cortical organoids model early human brain network development. Cell Stem Cell, 2019; 25, 558−69.e7. doi: 10.1016/j.stem.2019.08.002 [80] Muguruma K, Nishiyama A, Kawakami H, et al. Self-organization of polarized cerebellar tissue in 3D culture of human pluripotent stem cells. Cell Rep, 2015; 10, 537−50. doi: 10.1016/j.celrep.2014.12.051 [81] Bergmann S, Lawler SE, Qu Y, et al. Blood-brain-barrier organoids for investigating the permeability of CNS therapeutics. Nat Protoc, 2018; 13, 2827−43. doi: 10.1038/s41596-018-0066-x [82] Linkous A, Balamatsias D, Snuderl M, et al. Modeling patient-derived glioblastoma with cerebral organoids. Cell Rep, 2019; 26 3203-11. [83] Zecevic N, Chen Y, Filipovic R. Contributions of cortical subventricular zone to the development of the human cerebral cortex. J. Comp. Neurol. 2005; 109–22. [84] Cederquist GY, Asciolla JJ, Tchieu J, et al. Specification of positional identity in forebrain organoids. Nat Biotechnol, 2019; 37, 436−44. doi: 10.1038/s41587-019-0085-3 [85] Arzua T, Yan YS, Jiang CS, et al. Modeling alcohol-induced neurotoxicity using human induced pluripotent stem cell-derived three-dimensional cerebral organoids. Transl Psychiatry, 2020; 10, 347. doi: 10.1038/s41398-020-01029-4 [86] Kathuria A, Lopez-Lengowski K, Jagtap SS, et al. Transcriptomic landscape and functional characterization of induced pluripotent stem cell-derived cerebral organoids in schizophrenia. JAMA Psychiatry, 2020; 77, 745−54. doi: 10.1001/jamapsychiatry.2020.0196 [87] Brémond Martin C, Simon Chane C, Clouchoux C, et al. Recent trends and perspectives in cerebral organoids imaging and analysis. Front Neurosci, 2021; 15, 629067. doi: 10.3389/fnins.2021.629067 [88] Zilova L, Weinhardt V, Tavhelidse T, et al. Fish primary embryonic pluripotent cells assemble into retinal tissue mirroring in vivo early eye development. Elife, 2021; 10, e66998. doi: 10.7554/eLife.66998 [89] Eiraku M, Takata N, Ishibashi H, et al. Self-organizing optic-cup morphogenesis in three-dimensional culture. Nature, 2011; 472, 51−6. doi: 10.1038/nature09941 [90] Kuwahara A, Ozone C, Nakano T, et al. Generation of a ciliary margin-like stem cell niche from self-organizing human retinal tissue. Nat Commun, 2015; 6, 6286. doi: 10.1038/ncomms7286 [91] Nakano T, Ando S, Takata N, et al. Self-formation of optic cups and storable stratified neural retina from human ESCs. Cell Stem Cell, 2012; 10, 771−85. doi: 10.1016/j.stem.2012.05.009 [92] Regent F, Chen HY, Kelley RA, et al. A simple and efficient method for generating human retinal organoids. Mol Vis, 2020; 26, 97−105. [93] Cowan CS, Renner M, De Gennaro M, et al. Cell types of the human retina and its organoids at single-cell resolution. Cell, 2020; 182, 1623−40.e34. doi: 10.1016/j.cell.2020.08.013 [94] Kim S, Lowe A, Dharmat R, et al. Generation, transcriptome profiling, and functional validation of cone-rich human retinal organoids. Proc Natl Acad Sci USA, 2019; 116, 10824−33. doi: 10.1073/pnas.1901572116 [95] Wang DS, Wang JQ, Bai LY, et al. Long-term expansion of pancreatic islet organoids from resident Procr+ progenitors. Cell, 2020; 180, 1198−211.e19. doi: 10.1016/j.cell.2020.02.048 [96] Boj SF, Hwang CI, Baker LA, et al. Organoid models of human and mouse ductal pancreatic cancer. Cell, 2015; 160, 324−38. doi: 10.1016/j.cell.2014.12.021 [97] Boretto M, Cox B, Noben M, et al. Development of organoids from mouse and human endometrium showing endometrial epithelium physiology and long-term expandability. Development, 2017; 144, 1775−86. [98] Luddi A, Pavone V, Semplici B, et al. Organoids of human endometrium: a powerful in vitro model for the endometrium-embryo cross-talk at the implantation site. Cells, 2020; 9, 1121. doi: 10.3390/cells9051121 [99] Turco MY, Gardner L, Hughes J, et al. Long-term, hormone-responsive organoid cultures of human endometrium in a chemically defined medium. Nat Cell Biol, 2017; 19, 568−77. doi: 10.1038/ncb3516 [100] Duarte AA, Gogola E, Sachs N, et al. BRCA-deficient mouse mammary tumor organoids to study cancer-drug resistance. Nat Methods, 2018; 15, 134−40. doi: 10.1038/nmeth.4535 [101] Drost J, Karthaus WR, Gao D, et al. Organoid culture systems for prostate epithelial and cancer tissue. Nat Protoc, 2016; 11, 347−58. doi: 10.1038/nprot.2016.006 [102] Pappas KJ, Choi D, Sawyers CL, et al. Prostate organoid cultures as tools to translate genotypes and mutational profiles to pharmacological responses. J Vis Exp, 2019; 60346. [103] Guo WX, Li L, He J, et al. Single-cell transcriptomics identifies a distinct luminal progenitor cell type in distal prostate invagination tips. Nat Genet, 2020; 52, 908−18. doi: 10.1038/s41588-020-0642-1 [104] Karthaus WR, Iaquinta PJ, Drost J, et al. Identification of multipotent luminal progenitor cells in human prostate organoid cultures. Cell, 2014; 159, 163−75. doi: 10.1016/j.cell.2014.08.017 [105] Chua CW, Shibata M, Lei M, et al. Single luminal epithelial progenitors can generate prostate organoids in culture. Nat Cell Biol, 2014; 16, 951−61. doi: 10.1038/ncb3047 [106] Ma L, Li JW, Nie Q, et al. Organoid culture of human prostate cancer cell lines LNCaP and C4-2B. Am J Clin Exp Urol, 2017; 5, 25−33. [107] Chen XW, Li C, Chen Y, et al. Differentiation of human induced pluripotent stem cells into Leydig-like cells with molecular compounds. Cell Death Dis, 2019; 10, 220. doi: 10.1038/s41419-019-1461-0 [108] Mullenders J, de Jongh E, Brousali A, et al. Mouse and human urothelial cancer organoids: a tool for bladder cancer research. Proc Natl Acad Sci USA, 2019; 116, 4567−74. doi: 10.1073/pnas.1803595116 [109] Sakib S, Uchida A, Valenzuela-Leon P, et al. Formation of organotypic testicular organoids in microwell culture. Biol Reprod, 2019; 100, 1648−60. doi: 10.1093/biolre/ioz053 [110] Rodríguez Gutiérrez D, Eid W, Biason-Lauber A. A human gonadal cell model from induced pluripotent stem cells. Front Genet, 2018; 9, 498. doi: 10.3389/fgene.2018.00498 [111] Chen H, Ge RS, Zirkin BR. Leydig cells: from stem cells to aging. Mol. Cell Endocrinol, 2009; 306, 9−16. doi: 10.1016/j.mce.2009.01.023 [112] Sakib S, Goldsmith T, Voigt A, et al. Testicular organoids to study cell-cell interactions in the mammalian testis. Andrology, 2020; 8, 835−41. doi: 10.1111/andr.12680 [113] Sakib S, Yu Y, Voigt A, et al. Generation of porcine testicular organoids with testis specific architecture using microwell culture. J Vis Exp, 2019; 152. [114] Baert Y, De Kock J, Alves-Lopes JP, et al. Primary human testicular cells self-organize into organoids with testicular properties. Stem Cell Rep, 2017; 8, 30−8. doi: 10.1016/j.stemcr.2016.11.012 [115] Schulze ML, Lemoine MD, Fischer AW, et al. Dissecting hiPSC-CM pacemaker function in a cardiac organoid model. Biomaterials, 2019; 206, 133−45. doi: 10.1016/j.biomaterials.2019.03.023 [116] Drakhlis L, Biswanath S, Farr CM, et al. Human heart-forming organoids recapitulate early heart and foregut development. Nat Biotechnol, 2021; 39, 737−46. doi: 10.1038/s41587-021-00815-9 [117] Richards DJ, Li Y, Kerr CM, et al. Human cardiac organoids for the modelling of myocardial infarction and drug cardiotoxicity. Nat Biomed Eng, 2020; 4, 446−62. doi: 10.1038/s41551-020-0539-4 [118] Harrison RK. Phase II and phase III failures: 2013-2015. Nat Rev Drug Discov, 2016; 15, 817−8. doi: 10.1038/nrd.2016.184 [119] Kim J, Koo BK, Knoblich JA. Human organoids: model systems for human biology and medicine. Nat Rev Mol Cell Biol, 2020; 21, 571−84. doi: 10.1038/s41580-020-0259-3 [120] de Poel E, Lefferts JW, Beekman JM. Intestinal organoids for cystic Fibrosis research. J Cyst Fibros, 2020; 19, S60−4. [121] Berkers G, van Mourik P, Vonk AM, et al. Rectal organoids enable personalized treatment of cystic fibrosis. Cell Rep, 2019; 26, 1701−8.e3. doi: 10.1016/j.celrep.2019.01.068 [122] Driehuis E, van Hoeck A, Moore K, et al. Pancreatic cancer organoids recapitulate disease and allow personalized drug screening. Proc Natl Acad Sci USA, 2019; 116, 26580−90. doi: 10.1073/pnas.1911273116 [123] Saito Y, Muramatsu T, Kanai Y, et al. Establishment of patient-derived organoids and drug screening for biliary tract carcinoma. Cell Rep, 2019; 27, 1265−76.e4. doi: 10.1016/j.celrep.2019.03.088 [124] Ludikhuize MC, Meerlo M, Burgering BMT, et al. Protocol to profile the bioenergetics of organoids using Seahorse. STAR Protoc, 2021; 2, 100386. doi: 10.1016/j.xpro.2021.100386 [125] Boj SF, Vonk AM, Statia M, et al. Forskolin-induced swelling in intestinal organoids: an in vitro assay for assessing drug response in cystic fibrosis patients. J Vis Exp, 2017; 11, 55159. [126] Crowson MG, Ranisau J, Eskander A, et al. A contemporary review of machine learning in otolaryngology–head and neck surgery. Laryngoscope, 2020; 130, 45−51. doi: 10.1002/lary.27850 [127] Mohan S, Thirumalai C, Srivastava G. Effective heart disease prediction using hybrid machine learning techniques. IEEE Access, 2019; 7, 81542−54. doi: 10.1109/ACCESS.2019.2923707 [128] Rahman AKMS, Shamrat FMJM, Tasnim Z, et al. A comparative study on liver disease prediction using supervised machine learning algorithms. Int J Sci Technol Res, 2019; 8, 419−22. [129] Chang YH, Thibault G, Madin O, et al. Deep learning based Nucleus Classification in pancreas histological images. In: 2017 39th Annual International Conference of the IEEE Engineering in Medicine and Biology Society. IEEE. 2017, 672-5. [130] Štĕpánek L, Habarta F, Malá I, et al. A machine-learning approach to survival time-event predicting: initial analyses using stomach cancer data. In: 2020 International Conference on e-Health and Bioengineering. IEEE. 2020, 1-4. [131] Qin JM, Chen L, Liu YH, et al. A machine learning methodology for diagnosing chronic kidney disease. IEEE Access, 2019; 8, 20991−1002. [132] Kadir T, Fergus G. Lung cancer prediction using machine learning and advanced imaging techniques. Transl Lung Cancer Res, 2018; 7, 304−12. doi: 10.21037/tlcr.2018.05.15 [133] Das HS, Das A, Neog A, et al. Breast cancer detection: shallow convolutional neural network against deep convolutional neural networks based approach. Front Genet, 2023; 13, 1097207. doi: 10.3389/fgene.2022.1097207 [134] Munquad S, Si T, Mallik S, et al. A deep learning-based framework for supporting clinical diagnosis of glioblastoma subtypes. Front Genet, 2022; 13, 855420. doi: 10.3389/fgene.2022.855420 [135] Gaur L, Bhandari M, Razdan T, et al. Explanation-driven deep learning model for prediction of brain tumour status using MRI image data. Front Genet, 2022; 13, 822666. doi: 10.3389/fgene.2022.822666 [136] Zhang MM, Yang KL, Cui YC, et al. Current trends and research topics regarding intestinal organoids: an overview based on bibliometrics. Front Cell Dev Biol, 2021; 9, 609452. doi: 10.3389/fcell.2021.609452 [137] Rossi G, Manfrin A, Lutolf MP. Progress and potential in organoid research. Nat Rev Genet, 2018; 19, 671−87. doi: 10.1038/s41576-018-0051-9 [138] Lehmann R, Lee CM, Shugart EC, et al. Human organoids: a new dimension in cell biology. Mol Biol Cell, 2019; 30, 1129−37. doi: 10.1091/mbc.E19-03-0135 -




 下载:
下载:



 Quick Links
Quick Links