HTML
-
Application of synthetic silica nanoparticles (SNs) has received wide research attention in a variety of industries. SNs are produced on an industrial scale as additives for cosmetics, drugs, printer toners, varnishes, and food. In addition, the SNs are being developed for different biomedical and biotechnological applications, such as cancer therapy, DNA transfection, drug delivery, and enzyme immobilization[1-3]. With growing commercialization of nanotechnology products, the chance for human exposure to SNs is increasing and many aspects related to these nanomaterials have raised concerns about their safety[4-13]. Contrary to the well-studied crystalline micron-sized silica, relatively little information exists on their toxicity. Because nanoparticles possess novel properties, like kinetics and unusual bioactivity, their potential biological effects may differ greatly from those of micron-sized bulk materials. Most of the in vitro studies on the SNs have reported results for their cellular uptake, size-and dose-dependent cytotoxicity, increased reactive oxygen species levels and pro-inflammatory stimulation[5-11, 14-16]. Evidence from a limited number of in vivo studies on the SNs demonstrates largely reversible lung inflammation, granuloma formation and focal emphysema after their exposure, with no progressive lung fibrosis[12-13, 17-19]. Reliable in vitro assays are currently not available for predicting the full effects of the nanoparticles on the lung tissue[20-22]. More research with standardized materials is therefore needed to enable comparison of experimental data for different forms of SNs, to establish which physicochemical properties are responsible for their observed toxicity. In vivo models are thus necessary to study the effect of the SNs, because high permeability through the air-blood barrier allows their fast uptake.
The aim of this present study is therefore to study and clarify biological and pathological events on intratracheally instilled SNs in the rats' lungs during sub-acute stage, using histopathological and ultrastructural evaluations. In addition, factors, such as oxidative stress and pro-inflammatory that could be important in the induction of pulmonary toxicity by the SNs are comparatively investigated using colorimetry and immunohistochemistry.
-
Two sized SiO2 powders were used in the experiments. SNs were provided by Zhejiang Hongshen Material Technology Limited Company (Zhejiang, China). Silica content in the SiO2 nanoparticles is more than 99.5% and the hydroxyl group content on the surface of the SiO2 nanoparticles is more than 45%. The surface area for the SNs is 640 ± 30 m2/g (as provided by the production company). Microscale SiO2 powder is obtained from Sigma-Aldrich (Cat. no. 5631, USA), approximately 80% of the microscale SiO2 particles has diameter between 1-5 μm, and quartz purity was 99%, according to the data sheet of the company.
Suspensions of sterilized SNs and microscale SiO2 particles were diluted to different saline concentrations and then mixed with 20, 000 U penicillium. The suspensions were then dispersed by ultrasonic vibration for 15 min and shaken vigorously by a vortex shaker prior to administration.
-
All animal experiments were performed in compliance with local ethics committee. Specific pathogen-free male Wistar rats (180-220 g weight) were purchased from Zhejiang Research Center for Experimental Animals (Zhejiang, China) at 7th week of age. They were then acclimated for 3 d during which each animal was examined to confirm suitability for study. The rats were kept in individual cages and had free access to food and water. The animals were maintained in controlled environmental temperature (25 ± 1 ℃), relative humidity (45% ± 5%) and 12 h light/dark circle. 10 rats in the trial experiments were divided randomly into two groups with five rats in each group. The rats were anesthetized lightly with ether which was instilled intratracheally with SNs and microscale SiO2 powder dissolved in saline at 50.0 mg/mL concentrations. The SNs and microscale SiO2 powder we observed via trachea under direct observation using a laryngoscope for 24 h. 50 rats were divided randomly into five groups with 10 rats in each group in subsequent experiments. The rats were administered, as trial experiment, with 1 mL saline containing 6.25, 12.5, and 25.0 mg of SNs suspensions or 25.0 mg of microscale SiO2 powder suspensions.
-
30 d after instillation of the test material into the rats, body weights were determined in the 30 d groups. Each rat was injected intraperitoneally with a lethal dose (0.1 mL) of pentobarbital sodium solution. The lungs were then removed immediately from the thorax and weighted (lung wet weight). The lung/body coefficient was then calculated using the following equation: lung / body coefficient (%) = lung wet weight (g) / body weight (g) ×/100%. A small piece of lung was fixed by 10% formalin for at least 7 d before further processing. The formalin-fixed mouse lungs were embedded in paraffin from which thin coronal sections were mounted on glass microscope slides using standard histopathological techniques. Sections were stained with hematoxylin-eosin and examined by light microscopy.
-
1 mm3 samples of lung tissues were fixed in 2.5% glutaraldehyde in 0.1 mol/L cacodylate buffer at pH 7.2, followed by post fixing in 1% OsO4 in 0.1 mol/L cacodylate buffer. The samples were dehydrated in graded alcohol series and embedded in agar 100 resin. Thin sections, contrasted with uranyl acetate and lead citrate, were examined with a JEM 1011 transmission electron microscope (JEOl, Tokyo, Japan). Meanwhile, the chemical components in the lungs were analyzed by Inca X-sight energy spectrometer (Oxford Instruments).
-
Urine was collected and centrifuged at 4000 rpm for 10 min at 4 ℃ within 24 h after rats were instilled intratracheally with 1 mL of saline containing 25.0 mg of SNs and microscale SiO2 powder suspensions. The chemical components in the urine excretions were then observed and investigated by using TEM and EDS.
-
Following rats' lungs weighing, pieces of lung tissues were subsequently washed three times with saline and homogenized in ice bath using a glass homogenizer. The homogenate was centrifuged at 4, 000 rmp for 10 min at 4 ℃. Appropriate amount of the homogenate supernatants were quantified spectrophotometrically to detect the contents of MDA, NO, Hyp, and protein. This was done using thiobarbituric acid reaction (TBA) test, nitric acid reductase method, alkaline hydrolysis assay and Coomassie brilliant blue method, according to the manufacturer instructions for the commercial Kit (Nanjing Jiancheng Bioengineering Co, Ltd, China), respectively. Results were calculated as micromole of MDA per milligrams of protein, nano-mole of NO per milligrams of protein and milligrams of hydroxyproline per milligrams of protein.
-
Tissue sections from the lungs were immunostained by using avidin-biotin complex (ABC) method in which labeled Streptavidin biotin (LSAB) kit (DAKO, Glostrup, Denmark) was used. After deparaffinization of the tissue sections, they were treated with proteinase K for 30 min at 39 ℃. The sections were then incubated with 3% H2 O2 to quench endogenous peroxidase for 20 min at room temperature and then with 10% normal goat serum for 10 min twice in the microwave oven 250 w, to inhibit nonspecific reactions. The sections were thereafter reacted over night at 4 ℃ with rabbit anti-laminin monoclonal antibody diluted with 1:200 (Santa Cruz, America) of TNF-α, TGF-β1, IL-1β, and MMP-2. The peroxidase conjugated goat anti rabbit IgG diluted 1:400 (DAKO) was reacted with sections and a secondary antibody in microwave oven 200 w for 7 min. The positive reactions resulted in brown staining with substrate 3, 3'-diaminobenzidine tetrahydrochloride (Zhongshan Godenbridge Biotechnology Co. Ltd., China). The sections were counterstained with hematoxylin for 30 s. Brown particles in the cytoplasm or cellular membrane were considered positive and the TNF-α, TGF-β1, IL-1β, and MMP-2 proteins were quantitatively analyzed by using IMS cell image analysis system (UIC/OLYMPUS, US/JP). Three fields were randomly selected for every section in 10 × 40 fields of vision and three sections for every group. Positive area ratio and integrated optical density (OD) value were determined using the following formula: positive index = positive area ratio×optical density value × 100, representing the quantity of expression for the TNF-α, TGF-β1, IL-1β, and MMP-2.
-
Data, which included lung/body coefficient, oxidative stress, Hyp, and cytokine values, are presented as mean ± SD. SPSS for Windows 11.0 software package was used for statistical analysis. Differences between the groups were examined using Dunnett's t-test, standard one-way analysis of variance (ANOVA), Student-Newman-Keuls (SNK) test and Student's t-test. P < 0.05 indicates statistical significance.
Materials
Animal Treatment
Lung Collection and Histopathological Examination
Ultrastructural Examination of Lungs by Transmission Electron Microscope (TEM) and Energy Dispersive Spectrometer (EDS)
Urinary Excretion Analysis by EDS
Determination of Malondialdehyde (MDA), Nitric Oxide (NO), and Hydroxyproline (Hyp) in Lung Homogenates
Immunohistochemistry Staining of TNF-α, TGF-β1, IL-1β, and MMP-2
Statistical Analysis
-
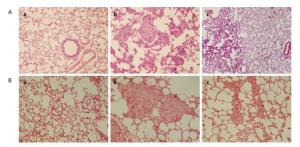
Five rats showed passive behavior, hypopnea, and hair erection in the trial experiment. Moreover, the rats also showed secretion around their eyes and moist rales of lungs after intratracheal instillation of 50 mg/mL SNs at 2-3 h. The rats were then successively killed within 24 h. An intact bronchial wall, far-ranging lung solidification, severe lung congestion and increasing lung volume and weight were observed in the rats' autopsies (Figure 1A). Pictures in Figure 1B show pathological changes in the fluid-filled and hemorrhagic alveolar cavity. Fortunately, five rats that were intratracheally instilled with 50.0 mg microscale SiO2 particles showed no overt abnormal signs after 24 h no rat died within 7 d, which is an observation period for acute toxicity. 25.0 mg per rat was confirmed to be the highest dosage for intratracheal instillation, as referred in relevant literature report[13, 23-24].
-
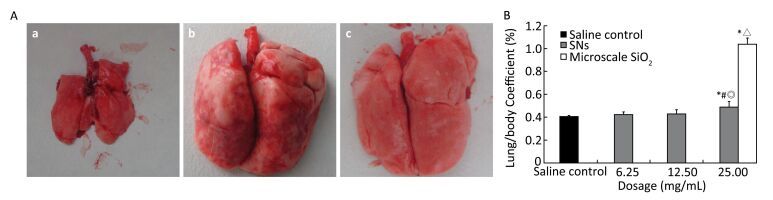
Pictures in Figure 2A show bilateral lungs of rats instilled saline with light pink color, with soft, smooth and spongy morphology. No abnormality was observed in the lungs from the saline control rats at 30 d. The lungs were intumescent in the microscale SiO2 particles group at 25 mg/mL level. Petechial and focal hemorrhage and diffuse greyish white nodules were diffusely distributed on the edges of lungs. The lungs texture was hard and sandy on palpation (Figure 2Ab). The abnormalities from the above lesion were slight in the 25 mg/mL SNs group (Figure 2 Ac).
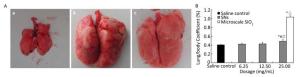
Figure 2. (A) Morphological changes of lung from rats instilled with 25 mg SNs and microscale SiO2 particles; (a) saline control group; (b) 25 mg/mL microscale SiO2 particles group; (c) 25 mg/mL SNs group. (B) Changes of lung/body coefficient from rats instilled with SNs and microscale SiO2 particles Significance indicated by: *P < 0.05 versus saline control group by the Dunnett's t-test. #P < 0.05 versus 6.25 mg/mL SNs group by the SNK test. ◎P < 0.05 versus 12.5 mg/mL SNs group by the SNK test. △P < 0.05 versus 25 mg/mL SNs group by the Student's t-test.
As shown in Figure 2B the lung/body coefficients for the 25 mg/mL microscale SiO2 particles and SNs groups increase and are significantly higher than those for the saline control groups (P < 0.05). The lung/body coefficients for the 25 mg/mL SNs group are significantly higher than those for the 6.25 mg/mL SNs group (P < 0.05) and the lung/body coefficient for the microscale SiO2 particles group is significantly higher than those for the SNs group at 25 mg/mL instillation concentration (P < 0.05).
-
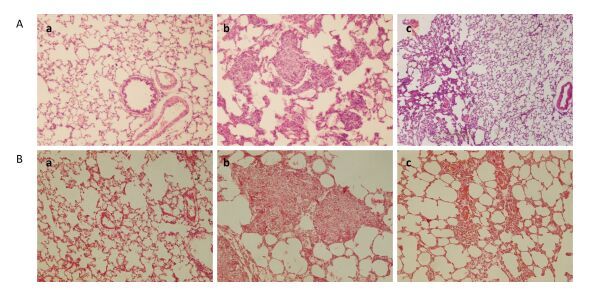
No obvious abnormalities in the bronchi, alveolus, and pulmonary interstitial structure could be observed in the saline control rats (Figure 3Aa). There are also no damaged lung tissue structure of partial alveolar cavity, atrophy, alveolar wall thickening or fracturing observed in the microscale SiO2 particles group 30 d after instillation. There is infiltration of neutrophils in the alveolar wall and septum infiltrated and also conglomeration of the macrophages in the alveolar cavity. Differently sized granulomas are scattered in the bronchioles. The granulomas are typical cellular nodules composed of particle-laden alveolar macrophages (Ams), epithelioid cells, multinucleated giant cells, lymphocytes and few Neutrophils (Figure 3Ab). However, partial lung tissue structure is destroyed in the SNs group. Neutrophils and few macrophages in the alveolar wall and septum infiltrate mildly and there is no distinct granuloma in the lungs (Figure 3Ac). Moreover, there are thick, pyknomorphic collagen fibers in the lungs from the rats instilled with the microscale SiO2 particles and some nodules are ranked closely and tended to amalgamate by VG stain (Figure 3Bb). Meanwhile, a small quantity of loose collagen fibers rank in the lungs from the rats instilled with the SNs (Figure 3Bc).
-
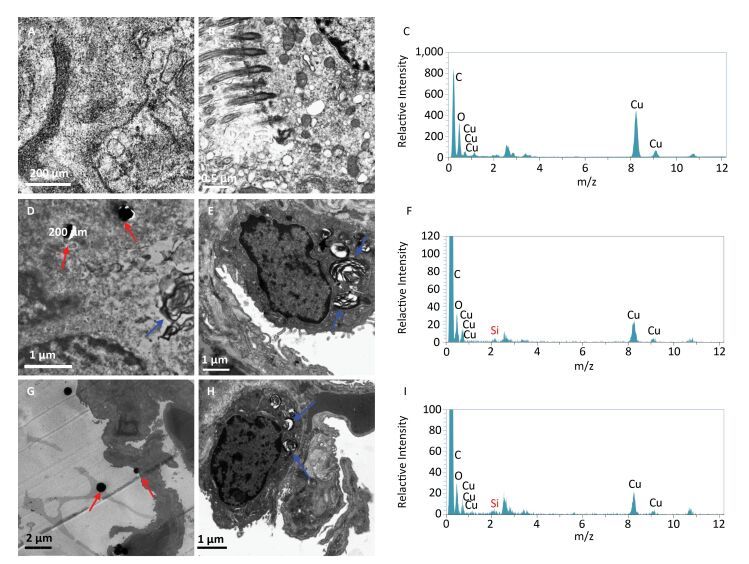
The TEM demonstrates opening alveolar cavity, alveolar walls lined with alveolar type Ⅰ cells, a few alveolar type Ⅱ cells and structural integrity of alveolar type Ⅰ and Ⅱ cells in the saline control group (Figure 4A-4B). Elements from Cu, C, and O were detected by EDS (Figure 4C). Pulmonary blood vessel cavity and basement membrane between epithelial cells are obviously incrassated and loosened, and basement membrane is indistinct in the microscale SiO2 particles group. Also, there is hyperplasia of collagen fibers and elastic fibers in the pulmonary interstitial. Mild hyperplastic type Ⅱ alveolar epithelial cells contain large amount of osmiophilic lamellar bodies (blue arrowhead). The macrophages increase and phagocytize numbers of osmiophilic lamellar bodies and powder particles (red arrowhead) (Figure 4D-4E). Si, Cu, C, and O element were also detected by EDS (Figure 4F). Moreover, the above changes of incrassation and loosening pulmonary blood vessel cavity and basement membrane between the epithelial cells, hyperplasic collagen fibers and elastic fibers in the pulmonary interstitial are inconspicuous in the SNs group. There are powder particles in the alveolar cavity (red arrowhead) and macrophages phagocytized osmiophilic lamellar bodies (blue arrowhead) (Figure 4G-4H). The Si, Cu, C, and O elements were also detected by EDS (Figure 4I).
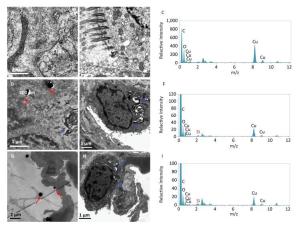
Figure 4. TEM (105 ×) and EDS images of lungs from SNs and microscale SiO2 particles treated rats for 30 d. (A, B) TEM image of saline control group; (C) EDS image of saline control group; (D, E) TEM image of 25 mg/mL microscale SiO2 particles group; (F) EDS image of 25 mg/mL microscale SiO2 particles group; (G, H) TEM image of 25 mg/mL SNs group; (I) EDS image of 25 mg/mL SNs group.
-
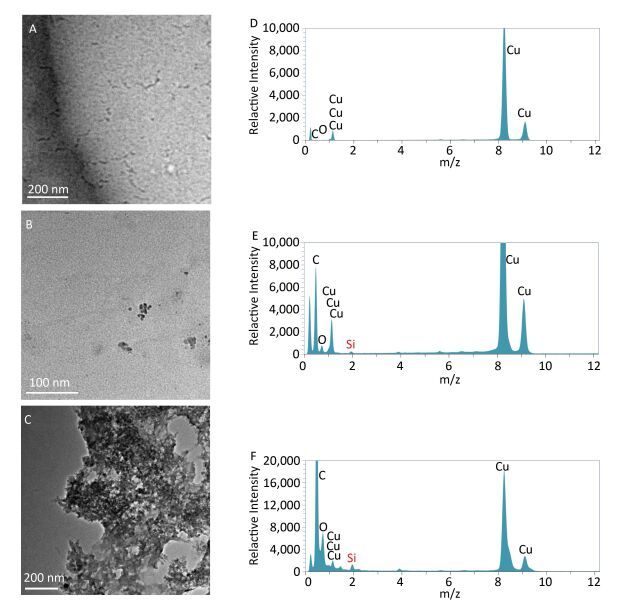
No residual proteins traces and particles were observed in the urine from the saline control group, by TEM (Figure 5A). The Si element was also not found by EDS (Figure 5D). There are small amount of particles in the urine from the rats instilled with microscale SiO2 particles (Figure 5B). The particles containing the Si element were analyzed by EDS (Figure 5E). A large amount of aggregated particles appear in the urine from the rats instilled with SNs (Figure 5C). The particles containing large amount of Si elements were analyzed by EDS (Figure 5F). The results from this part of experiment show that partial small particle sized microscale SiO2 particles can be excreted via kidney and large amounts of SNs are excreted via the kidney, due to their ultrafine particle size compared to microsized particles after instillation of the SNs and microscale SiO2 particles for 24 h.
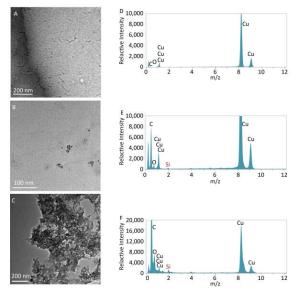
Figure 5. TEM (105 ×) and EDS images of urine from rats treated with SNs and microscale SiO2 for 24 h. (A) TEM image of saline control group; (B) TEM image of 25 mg/mL microscale SiO2 particles group; (C) TEM image of 25 mg/mL SNs group; (D) EDS image of saline control group; (E) EDS image of 25 mg/mL microscale SiO2 particles group; (F) EDS image of 25 mg/mL SNs group.
-
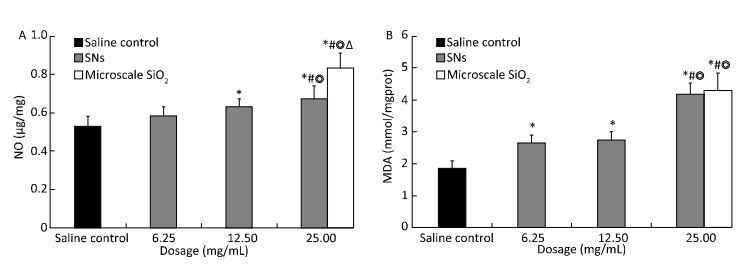
The level of MDA and NO production in the lung homogenate changed after the rats were instilled with SNs and microscale SiO2 particles for 30 d. MDA production increase in the SNs group at 6.25, 12.5, and 25 mg/mL concentration and in the microscale SiO2 particles group at 25 mg/mL concentration. NO production increase in the SNs group at 12.5 and 25 mg/mL concentrations and microscale SiO2 particles group at 25 mg/mL concentration. There are statistically significant differences between the two groups when compared with the saline control group (P < 0.05). The MDA and NO production in the SNs group at 25 mg/mL concentration is higher than those in the SNs group at 6.25 and 12.5 mg/mL concentrations (P < 0.05). Moreover, the NO production in the microscale SiO2 particles group at 25 mg/mL concentration is higher than that in the SNs group at same concentration (P < 0.05). There is no statistically significant MDA difference between the SNs group and microscale SiO2 particles group at 25 mg/mL concentration (P > 0.05) (Figure 6).

Figure 6. Content of NO (A) and MDA (B) in lungs homogenate from rats treated with SNs and microscale SiO2 particles for 30 d. Significance indicated by: *P < 0.05 versus saline control group by the Dunnett's t-test. #P < 0.05 versus 6.25 mg/mL SNs group by the SNK test. ◎P < 0.05 versus 12.5 mg/mL SNs group by the SNK test. △P < 0.05 versus 25 mg/mL SNs group by the Student's t-test.
-
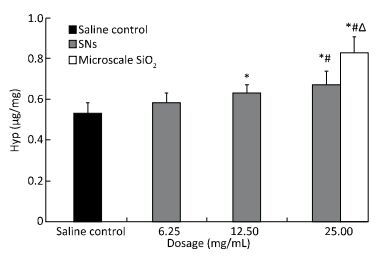
The level of Hyp production in the lung homogenate increase after the rats were instilled with the SNs and microscale SiO2 particles at 30 d. The Hyp production increase in the SNs group at 12.5 and 25 mg/mL concentrations of SNs and in the microscale SiO2 particles group at 25 mg/mL concentration. There are statistically significant differences between groups when they compared with the saline control group (P < 0.05). The Hyp production in the SNs group at 25 mg/mL concentration is higher than that in the SNs group at 6.25 mg/mL concentration (P < 0.05). Moreover, the Hyp production in the microscale SiO2 particles group at 25 mg/mL concentration is higher than that in the SNs group at same dosage (P < 0.05) (Figure 7).
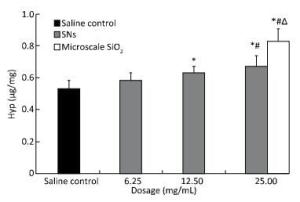
Figure 7. Content of Hyp in lung homogenate from rats treated with SNs and microscale SiO2 particles for 30 d. Significance indicated by: *P < 0.05 versus saline control group by the Dunnett's t-test. #P < 0.05 versus 6.25 mg/mL SNs group by the SNK test. △P < 0.05 versus 25 mg/mL SNs group by the Student's t-test.
-
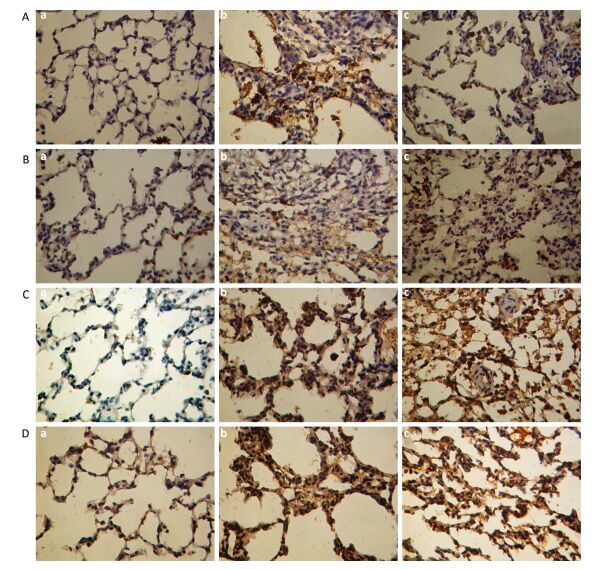
Immunohistological images for the TNF-α, TGF-β1, IL-1β, and MMP-2 expression in the lung tissue are shown in Figure 8A to 8D, respectively, at 30 d post instillation of the SNs and microscale SiO2 particles. Positive expressions of cytokines are presented with brown or dark brown. A few cells for positive cytokines expression are localized in the area of alveolar cavity with exudative inflammation in saline group. The macrophages and some inflammatory cells such as neutrophils, lymphocytes and plasma cells in silicotic nodules demonstrate positive expression in the microscale SiO2 particles group. Brown positive stains are observed in the cytoplasm of macrophages and inflammatory cells located in the area of inflammation reaction and silicotic nodules. Positive expressions of cytokines in the lung tissue are reduced outside the silicotic nodules. Moreover, the particle-laden AMs, lymphocytes and plasma cells display positive expressions of cytokines in the SNs group. Besides, some inflammatory cells with exudative inflammation in the alveolar cavity show positive expressions. Higher quantity of positive expressions appeared in the microscale SiO2 particles group compared with SNs group.
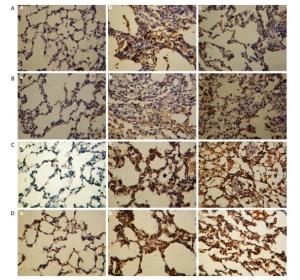
Figure 8. Immunohistochemical stain for TNF-α, TGF-β1, IL-1β, and MMP-2 in lungs from SNs and microscale SiO2 particles treated rats for 30 d (250 ×). (A) TNF-α; (B) TGF-β1; (C) IL-1β; (D) MMP-2. Images of (a-c) show (a) saline control group; (b) 25 mg/mL microscale SiO2 particles group; (c) 25 mg/mL SNs group.
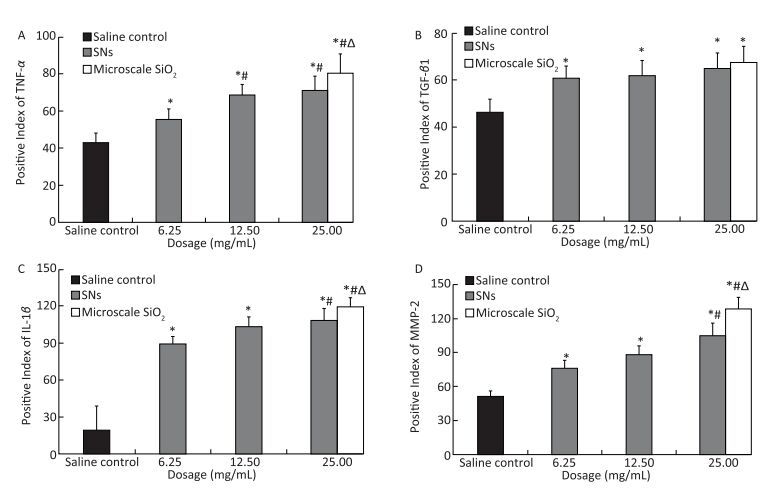
The comparison of positive expression levels of TNF-α, TGF-β1, IL-1β, and MMP-2 in the lung tissue from the SNs and microscale SiO2 particles groups is shown in Figure 9A to 9D, respectively. TNF-α, TGF-β1, IL-1β, and MMP-2 expressions increase in the SNs group at 6.25, 12.5, and 25 mg/mL SNs concentrations, and also in the microscale SiO2 particles group at 25 mg/mL concentration. By the Dunnett's t-test, there are statistically significant differences between groups when compared with the saline control group (P < 0.05). TNF-α expression in the SNs group at 12.5 and 25 mg/mL concentrations is higher than that in the SNs group at 6.25 mg/mL concentration (P < 0.05), by the SNK test. Moreover, the TNF-α, IL-1β, and MMP-2 expressions in the microscale SiO2 particles group at 25 mg/mL concentration is higher than that in the SNs group at same dosage (P < 0.05), by the Student's t-test. There is no statistically significant difference of TGF-β1 expression between the SNs and microscale SiO2 particles groups at 25 mg/mL concentration (P > 0.05).
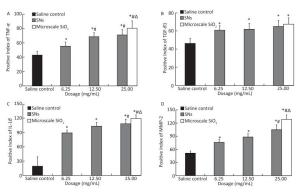
Figure 9. Positive expression levels of TNF-α (A), TGF-β1 (B), IL-1β (C), and MMP-2 (D) in lungs from SNs and microscale SiO2 particles treated rats for 30 d. Significance indicated by: *P < 0.05 versus saline control group by the Dunnett's t-test. #P < 0.05 versus 6.25 mg/mL SNs group by the SNK test. △P < 0.05 versus 25 mg/mL SNs group by the Student's t-test.
Acute Pulmonary Lesions in Rats Induced by Instilled SNs
Morphological Changes and Influence of Lung/body Coefficient from Rats Instilled with SNs
Pathological Changes in Lungs from SNs Instilled Rats
Ultrastructural Changes and EDS of Lungs from Rats Instilled with SNs
EDS of Urinary Excretions from Rats Instilled with SNs
Oxidative Injury of Lungs from Rats Induced by Instilled SNs
Content of Hyp in Lung Homogenate from Rats Induced by Instilled SNs
Effect on TNF-α, TGF-β1, IL-1β, and MMP-2 in the Lungs from Rats Induced by Instilled SNs
-
The direct instillation of silica into the lungs via the trachea has been employed in many studies as an animal model for silicosis. Inhalation provides a natural route of entry into the host and as such, is preferable for the introduction of silica into the lungs. An intratracheal instillation cannot be further from the physiological condition. The distribution of an instilled material within the respiratory tract will likely differ from the distribution of an inhaled material. The upper respiratory tract (i.e., the nasal passages, oral passages, pharynx, and larynx) can be a potentially important target site for an inhaled test material passed by intratracheal instillation. Especially, another disadvantage of instillation is the introduction of the toxicant being non-physiological and involving invasive delivery usually at a dose and/or dose rate substantially greater than that which would have occurred during inhalation. However, instillation has certain advantages over inhalation, as discussed in detail by Brain et al.[25]. Briefly, with instillation, the actual dose delivered to the lungs of each animal can be essentially assured. The technique is simpler than inhalation exposure procedures and minimizes risks to laboratory workers from highly toxic, carcinogenic, or radioactive materials. Intratracheal instillation permits the introduction of a range of doses to the lungs within a short time, and avoids exposure to the skin and pelt that can occur with inhalation exposure. Furthermore, intratracheal instillation has become sufficiently widely used as a screening tool for determining the approximate dose range that may be appropriate for later inhalation studies, or to determine the ranking of toxicity for a series of structurally similar chemical agents[25-26]. Respiration toxicological information for various particles has reviewed that some biological end points, such as pulmonary inflammation, fibrosis and susceptibility to infection, hypersusceptibility and development of lung cancer, were similar after the rats were exposed to low-solubility by two kind manners of inhalation and instillation. Some researchers have therefore considered that selectively applying intratracheal instillation by investigating toxicological character of dust according to different aims is likely to provide valuable information consistent with inhalation on the base of predominating application rules[27].
In the pulmonary toxicological study of dust, particle loading into the lungs is an important factor. A number of studies have investigated the relationship that may exist between the pulmonary silica burden and subsequent development of silicosis. The particles exceeding loading do not directly influence pulmonary pathological end point. Over-load of dusts is enough to damage the ability of macrophages to clean particles, then inducing nonspecific inflammation, particles deposition in the pulmonary interstitial or alveolus cavity and epithelia hyperplasia. By contrary, the particles are normally eliminated by all kinds of cleaning mechanisms[28]. A large amount of silica in the lung tissue is closely related to silicosis development. Nagelschmidt summarized the literatures on pathological response of silicosis to multiple silica loads. He considered that there was relationship between increased silica amounts and silicosis pathological classification[29]. In classical silicosis of gold-miners and molder, 1 to 3 g of silica particles was found in their lungs to result in pulmonary fibrosis. This current study referred to a variety of dust dosages/concentrations of experimental silicosis in the animal model[13, 23]. In the trial experiment, every rat was intratracheally instilled with SNs dissolved in saline at 50 mg/mL concentrations via trachea on the premise of established pulmonary fibrosis model induced by the SNs. The rats were successively died at 24 h and they were expected to die of acute pulmonary edema by gross and histopathological inspection of their lungs. Fortunately, five rats that were instilled intratracheally with 50.0 mg microscale SiO2 particles showed no overt abnormal signs at 24 h and no rat died in 7 d, which is an observation period for acute toxicity. These results suggest that the acute pulmonary lesions for the rats exposed to high dosage of SNs are more serious than those from the microscale SiO2 particles group rats at same dosage. These results are in agreement with findings from other investigators. Kaewamatawong reported that mice were intratracheally instilled with 3 mg of 14 nm and 230 nm colloidal silica particles (CSs) and pathologically examined from 30 min to 24 h post exposure[30]. They histopathologically found that the lungs exposed to both sizes of particles showed bronchiolar degeneration and necrosis, neutrophilic inflammation in alveoli with alveolar type Ⅱ cell swelling and particle-laden alveolar macrophages (AMs) accumulation. The 14-nm CSs, however, induces extensive alveolar hemorrhage compared to 230-nm CSs from 30 minutes onwards. The 14-nm CSs also causes more severe bronchiolar epithelial cell necrosis and neutrophil influx in alveoli than 230-nm CSs at 12 and 24 h post exposure. These findings are perhaps in connection with small size effect from the nanoparticles and liability to aggregation. High concentration nanoparticles instilled intratracheally for a comparatively short time aggregate to larger mass compared with macroscale particles, and they also obstruct the airway to a different extent along the bronchus, bronchiole, and terminal bronchioles entering into alveoli. In addition, pulmonary reactivity to SNs is obviously increased because of the large surface area and high bioactivity of the SNs. Therefore, in this study, 25 mg SNs in 1 mL of saline per rat is confirmed highest exposure dose for intratracheal instillation, based on our trial experiment and literature reports[13].
Sayes et al. reported that rats were exposed to 1 or 5 mg/kg of crystalline silica (Min-U-Sil 5, α-quartz) and precipitated amorphous silica by intratracheal instillation, and BAL fluids from the rats were analyzed at 24 h, 1 week, 1 month, and 3 months post instillation exposure[20]. They found that the crystalline silica particles produced sustained inflammation and cytotoxicity. Precipitated amorphous silica particles produced reversible and transient inflammatory responses. Moreover, results from in vivo pulmonary toxicity studies by Chen et al.demonstrated that the Wistar rats were instilled intratracheally with 20 mg of nanosized SiO2 or 20 mg of microsized SiO2 . The rats were sacrificed at 1 and 2 months after instillation. The lung/body coefficient of nanosized SiO2 groups are significantly lower than those of the microsized SiO2 groups at both 1 and 2 months after instillation (P < 0.05 or P < 0.01), but without significant differences from those of saline control groups[17]. The results from our in vivo pulmonary toxicity studies indicate that the instilled SNs produce little toxicity and the microscale SiO2 particles exposures produced pulmonary fibrosis.
The lung ultrastructural change is the same an intuitionistic index for histomorphological changes. Transmission electron microscopy (TEM) reveals hyperplastic alveolar type Ⅱ cells lining the alveoli[31], and the occasional clusters of alveolar type Ⅱ cells contained lots of osmiophilic lamellar bodies dissociated in the alveoli cavity[32]. The hypertrophic AMs are activated by silica contained vesicle of phagocytosing silica, lamellar bodies, and lipoproteinosis[33]. Some researchers pointed out hyperplastic alveolar type Ⅱ cells being initial events and hypertrophic AMs being key link to pulmonary fibrosis development in experimental silicosis[34-35]. The ultrastructural changes in the lungs as induced by the SNs and microscale SiO2 particles in this study are in accordance with given literature. The result from the lungs EDS analysis indicate the macrophage engulfed SNs and microscale SiO2 particles.
Bio-distribution and clearance of the nanoparticles are not likely due to the microscale SiO2 particles on account of their small size. Cho et al.prepared ultrafine amorphous silica particles (UFASs) in phosphate buffered saline (PBS) and intratracheally administered to A/J mice at 0, 2, 10, and 50 mg/kg doses. The animals were sacrificed at 24 h, 1, 4, and 14 weeks following exposures. The intratracheal instillation of the UFASs at 2 and 10 mg/kg doses has no effects on the lung/body coefficients gains. In contrast, the instillation of 50 mg/kg UFASs significantly increase the absolute and relative lung weights, while 24 h and 1 week instillation have no effects on the lung/body coefficients gains at 4 or 14 weeks following exposures compared to controls. The histopathological examination reveals that the UFASs induce severe inflammation with neutrophils at an early stage and chronic granulomatous inflammation at the later stage. However, the lung lesions are milder at 4 weeks and have almost recovered at the final time point[17]. These findings perhaps explain that the ultrafine particles may translocate from the site of deposition into the lungs and extrapulmonary organs through the systemic circulation, which may result in the rapid elimination of lung inflammation and injury. However, during the early event after instillation, the severity of injury due to ultrafine particles is more severe than that of fine particles. He et al. investigated the biodistribution and urinary excretion of three types of surface-modified silica nanoparticles with 45 nm size in mice injected intravenously (iv) using an optical imaging method, taking advantage of RuBPY dye doped in the silica matrix as a synchronous fluorescence signal. Results from the in vivo imaging studies show that three types of surface-modified silica nanoparticles can all be cleared from the circulation and present inside organs and partly excreted through the renal route[36]. Several studies corroborat that quartz dust is difficult to eliminate outside the lungs at long exposure periods by organism own clearance system[37-38]. It is an important reason for silicosis development and progress. The current study reveals that the SNs are partly excreted through the renal excretion route. However, this needs further study to assess whether the SNs produce the same pulmonary toxicity as the microscale SiO2 particles. TEM and EDS analysis of urine samples in this study confirm that there is relatively little excretion of small size microscale SiO2 particles through the renal route. In contrast, a large amount of granular substances containe Si in the urine from rats post exposure to SNs at 24 h. The results from the excretion studies confirm that the SNs are in fact excreted through the urine as intact SNs through the renal route. Hence, these findings perhaps explain that the SNs deposition in the lungs are less than microscale SiO2 , and result in slight pulmonary lesion.
Oxidative stress is the result of an imbalance in the pro-oxidant/antioxidant homeostasis. Many reports have been released on the relationship between oxidative stress and inflammation. Oxidative stress may trigger activation of transcription factor, induce mRNA expression of inflammation media, and finally bring up inflammation reaction and related disease[39]. Nitric oxide is an omnipresent signaling molecule produced by a variety of mammalian cells, including vascular endothelium, neurons, smooth muscle cells, macrophages, neutrophils, platelets, and pulmonary epithelium. In pathological condition, nitric oxide formation is increased and it induces inflammation development[40]. Park et al. treated mice with 50 mg/kg silica nanoparticles through intraperitoneal injection. The mice were sacrificed at 12, 24, 48, and 72 h after treatments, respectively. Activated macrophages from the peritoneal cavity of the mice and increased NO release from the cells to the supernatant in a time-dependent manner were observed as a result. At the same time, the reactive oxygen species (ROS) and NO generation were increased and intracellular glutathione (GSH) was decreased in a dose-dependent manner in RAW264.7 cell line which originated from the mouse peritoneal macrophage[41]. Study suggested that silica nanoparticles induced pro-inflammatory responses in vivo/in vitro and pro-inflammatory responses may be triggered by ROS and NO generation. MDA is an end product of lipid peroxidation and superoxide dismutase (SOD) is a cleaner of free radicals. There is negative correlation between MDA and SOD. Yang et al. reported that SiO2 nanoparticles induced inhibition of primary mouse embryo fibroblasts cell viabilities and elevation of ROS and MDA levels in cell medium (at 20-100 μg/mL) in an explicit dose-dependent manner[42]. These results proved that the oxidative stress was probably a key route by which the nanoparticles induced cytotoxicity. Some in vivo experiments directly demonstrated that silicosis was a state of oxidative stress and that increased generation of ROS was associated with enhanced levels of oxidative enzymes and lipid peroxidation[43-44]. Similarly, in our current research study, the level of MDA and NO in the lung homogenate is increased in the SNs group at 12.5 and 25 mg/mL concentration and in the microscale SiO2 particles group at 25 mg/mL concentration. These experimental results reveal that the SNs may result in pulmonary lesion-induced oxidative stress.
Free radical chain reaction was initiated by free crystalline silica inhalation and various bioactive substances, such as TNF-α, TGF-β1, PDGF, and FGF were released by AMs in another study[45]. Besides the AMs, the above cytokines are produced by neutrophils, lymphocytes, and alveolar epithelial cells (type Ⅰ and type Ⅱ) stimulated by the dust. These cytokines promote fibroblasts proliferation, collagen metabolism, eventually leading to pulmonary fibrosis[46]. Park et al.reported in another study that the mRNA expressions of inflammatory-related genes, such as IL-1β, TNF-α, IL-6, iNOS, and COX-2, were upregulated in a time-dependent manner in the macrophages harvested from the mice treated with silica 50 mg/kg nanoparticles. The levels of pro-inflammatory cytokines (IL-1β and TNF-α) released to serum in the treated mice were also elevated after i. p. injection of silica nanoparticles 50 mg/kg. IL-1β reached maximum at 12-24 h after treatment and the level was decreased gradually in a time-dependent manner at 72 h. Also, TNF-α reached maximum at 24 h after treatment and its level decreased gradually in a time-dependent manner[41]. In studies by Choi et al., the histopathological examination revealed that 14nm ultrafine amorphous silica (UFAS), intratracheally administered to A/J mice, induced severe inflammation with neutrophils at an early stage and chronic granulomatous inflammation at the later stage. The mRNA and protein levels of IL-1β, IL-6, IL-8, TNF-α, MCP-1, and MIP-2 in the lung tissues are significantly increased during the early stages, but there are no changes after weeks 1 (TNF-α) or 4 (IL-1β, IL-6, IL-8, MCP-1, and MIP-2). Instillation of UFASs induced transient, but very severe lung inflammation. These results show that the cytokines (IL-1β, IL-6, IL-8, and TNF-α) and chemokines (MCP-1 and MIP-2) play important roles in the inflammation induced by the intratracheal instillation of UFASs[17]. Another study showed that the UFAS induced severe alveolar epithelial thickening and pulmonary fibrosis at 1 week by Gomori's trichrome staining, and almost recovered at 4 and 14 weeks. The mRNA and protein levels of cytokines (IL-4, IL-10, IL-13, and IFN-γ), matrix metalloproteinases (MMP-2, MMP-9, and MMP-10) and tissue inhibitor of matrix metalloproteinase-1 (TIMP-1) in lung tissues are significantly elevated at 24 h and 1 week post-treatment. These levels decrease to near control range at 4 and 14 weeks except for the IFN-γ and MMP-2. These results demonstrate that the UFAS induce pulmonary fibrosis in the same way as crystalline silica. However, the degree of fibrosis observed is transient. This study show that cytokines (IL-4, IL-10, IL-13, and IFN-γ), MMPs (MMP-2, MMP-9, and MMP-10) and TIMP-1 play important roles in the fibrosis induced by the intratracheal instillation of UFAS[47].
Specific substrate of MMP-2 is Type IV collagen and the later is important component of alveolar wall basement membrane. High expression of MMP-2 induced by nosogenesis plays roles in destruction of alveolar epithelial cells basement membrane and invasion of alveolar fibroblasts to the alveolar space in pulmonary fibrosis. Animal experiments and clinical studies demonstrated elevation of MMP-2 expression in the pulmonary fibrosis[48-49]. The MMP-2 in the pulmonary tissue is mainly from the AMs. Pulmonary interstitial fibroblasts are major cells source for the MMP-2 in early and mid-term pulmonary fibrosis. Therefore, pulmonary interstitial fibroblasts are not simply effective cells in pulmonary fibrosis, as they are concerned with structural damage of the lung basement membrane and initiate the process of pulmonary fibrosis by MMP-2 expression. The increase in collagen synthesis and secretion is an important link to pulmonary fibrosis development under the regulation of AMs. In vitro experiments in another study indicated that the supernatant medium from the AMs exposed to quartz promoted proliferation, collagen synthesis and secretion of fibroblasts[50-51]. According to in vivo study, the lung/body coefficient and hydroxyproline content for the nanosized SiO2 groups are significantly lower than those of the microsized SiO2 groups at both 1 and 2 months after instillation of nanosized SiO2 . The expressions of IL-4 and TGF-β1 in the nanosized SiO2 groups are significantly lower than those in the microsized SiO2 groups, and the effect of fibrogenesis induced by nanosized SiO2 might be milder than that induced by microsized SiO2 at 2 months after instillation[13].
The Hyp production in the lung tissue from the rats increase in 12.5 and 25 mg/mL SNs in this study, and also in 25 mg/mL microscale SiO2 particles instilled group. The Hyp production in the microscale SiO2 particles group at 25 mg/mL concentration is higher than that in the SNs group at same dosage. The TNF-α, TGF-β1, IL-1β, and MMP-2 expressions are increased in the SNs group at 6.25, 12.5, and 25 mg/mL concentrations and also in the microscale SiO2 particles group at 25 mg/mL concentration when compared with the saline control group. The TNF-α, IL-1β, and MMP-2 expressions in the SNs group and microscale SiO2 particles group at 25 mg/mL concentration are higher than those in the SNs group at 6.25 and 12.5 mg/mL concentrations. Moreover, the TNF-α, IL-1β, and MMP-2 expressions in the microscale SiO2 particles group at 25 mg/mL concentrations are higher than those in the SNs group at same dosage. Cytokine expression in the SNs and microscale SiO2 particles instilled group is mainly located in macrophages and some inflammatory cells, such as neutrophils, lymphocytes and plasma cells. These results demonstrate that the SNs could induce pulmonary fibrosis in the same way as microscale SiO2 particles by stimulating cytokine expression increase in the lung tissue.
The results from the in vivo study are different from that from our previously reported in vitro study. The in vitro investigations reveal that the SNs are more cytotoxic and damaged RAW264.7 cells more severely than the microscale SiO2 particles in same dosage[52]. The study of single 7 mg/kg intravenous infusions of 13 nm SNs in rats, Zhuravskii et al. found mast cell (MC) abundance in the liver and recruitment in the liver preceded fibrosis[53]. The in vivo investigations display that the degree of fibrosis induced by the SNs is milder than that induced by the microsized SiO2 in the rats. On the one hand, these findings are perhaps attributed to discrepancy in the physicochemical properties and biological activity between the SNs and microscale SiO2 particles. Shamsi et al reported that SNs incubated buffalo kidney cystatin (BKC) changed its conformation, reduced cell viability clearly suggesting toxicity of SNs. SNs have a deteriorating effect on BKC thereby causing a decrease in its ability to inhibit papain and hence less functionality. BKC activity decrease can certainly be an implication in many renal diseases highlighting the importance of this important thiol protease inhibitor[54]. Although chemical components of the SNs and microscale SiO2 particles are the same, biological activity for the SNs appear to depend on the size effect due to small size and large surface area of the SNs. The size of the nanoparticles is similar to DNA, proteins, viruses and biological molecules, hence they are likely able to pass through the pulmonary blood barrier and skin barrier, and get into the body by means of simple diffusion and penetration form. These nanoparticles are prone to permeate the pores on the membrane and enter into the cells or organelles like mitochondria, endoplasmic reticulum, lysosomes, Golgi apparatus, cell nucleus, etc, combined with biological macromolecules or catalytic reactions occurring at the same time as well. The normal biological macromolecules and three-dimensional structure of the biological membrane is thus altered, resulting in loss of some important hormones and enzymes activity[55-56]. Some scholars believe therefore that size is an important factor for determining the toxicity of nanoparticles besides dose. Even if they are the same material with same shape and dose, as long as their sizes change, their biological toxicity must be retested and reevaluated. Some experimental results reveal that there is difference of toxicity between microscale and nanoscale materials[5, 7-8, 10, 20]. Lin et al. investigated the cytotoxicity of amorphous (colloidal) SNs (15 and 46 nm) in cultured human alveolar epithelial cells (A549 cells) in another study. Nanoparticles with both sizes of SNs are more cytotoxic than fine quartz particles (Min-USil 5), and there are no significant difference in toxicity for same sized SNs[57]. On the other hand, the effect on fibrogenesis by the SNs may be milder than that of microscale SiO2 particles in rats, potentially resulting from nanoparticles tending to be diffused to pulmonary interstitial, and easily translocating blood circulation and eliminated from the body in the urine, thereby reducing the SNs preservation in the lungs, due to their ultrafine particle size compared to microsized particles. This current study reveal that there are a great deal SNs in the urine of rats instilled intratracheally with the SNs at 24 h. These result support the above discussed view. Research on the SNs has just started and lot of further research needs to be done for the sake of clarifying their toxicity and health effects.
-
Results from the present in vivo experiment have shown that the rats died from acute edema in succession after an intratracheal instillation with 50 mg/mL of SNs. The low dosage of the SNs could bring about pulmonary lesions, such as rise in lung/body coefficients, lung inflammation, damaged alveolar, granuloma nodules formation, collagen metabolite perturbation in the lung tissue and extracellular matrix aggradation by inducing increase of lipid peroxidation and high expression of cytokines (TNF-α, TGF-β1, IL-1β, and MMP-2), all resulting in pulmonary fibrosis. The experimental results further revealed that the effect on fibrogenesis by the SNs may be milder than that of the microscale SiO2 particles in the rats, potentially resulting from nanoparticles tending to diffuse through the pulmonary interstitial, and easily translocating blood circulation and eliminated from the body in the urine, hence reducing the SNs preservation in the lungs due to their ultrafine particle size compared to microsized particles. However, the acute lung injury induced by the SNs cannot be ignored and the pulmonary lesion effects induced by SNs instillation times need further investigation.







 Quick Links
Quick Links
 DownLoad:
DownLoad: