-
The aim of the present study is to evaluate the ability and mechanism by which grape seed procyanidin extract (GSPE) relieves arsenic trioxide (As2O3)-induced renal inflammatory injury. Therefore, male Kunming mice were treated with As2O3and/or GSPE by gavage for 5 weeks. Mice were then sacrificed and inflammatory cytokines of kidneys were examined by ELISA, whereas the expression levels of molecules involved in the nuclear factor (NF)-κB signaling pathway were evaluated by both qRT-PCR and Western blot. Our results indicate that GSPE prevents As2O3-mediated renal inflammatory injury by inhibiting activation of the NF-κB signaling pathway and inflammatory cytokine production, while promoting expression of anti-inflammatory cytokines.
Arsenic, a heavy metal that was identified as a carcinogen, is ubiquitous in nature. One of the primary routes of exposure to this metal is via consumption of contaminated drinking water. Indeed, it has been reported that this way can result in the development of injury of the liver, kidney, and other viscera[1]. In a previous study, consumption of sodium arsenite enhanced tumor necrosis factor (TNF)-α expression in rats. It demonstrated that arsenic consumption can cause inflammatory injury in kidney tissues[2]. Therefore, nuclear transcription factor kappa B (NF-κB), a pathway has an ability to regulate arsenic-mediated inflammatory response, plays an indispensable role. Indeed, arsenic was shown to promote NF-κB activation, as well as to enhance inflammatory cytokine and decrease anti-inflammatory cytokine production. Moreover, toxic substances can promote the degradation of IκB by enhancing IκB kinase activity, resulting in the formation of NF-κBp65 and NF-κBp50 dimers, which subsequently translocate to the nucleus and induce the expression of pro-inflammatory cytokines[3]. Thus, the development of methods for counteracting arsenic-mediated toxicity is of great significance.
Procyanidins (PCs) are polyphenol compounds that exert strong anti-inflammatory effects[4]. For example, treatment with PC was shown to reverse the increased production of TNF-α and IL-1β observed in rats treated with carrageenin[5]. Notably, PCs comprise up to 90% of grape seed procyanidin extract (GSPE)[6]. However, the effects of these extracts on renal inflammatory damage induced by As2O3have yet to be tested. In this study, we therefore aimed to explore the effects and mechanism by which GSPE suppresses arsenic-induced inflammatory injury, and to provide a theoretical basis for the development of methods to prevent and/or control arsenic toxicity via the application of GSPE.
Healthy, specific pathogen-free, male Kunming mice (n = 40), weighing between 18 g and 22 g, were obtained from the laboratory center of Xinjiang Medical University (Urumqi, China) (license numbers: SYXK 2011-0004). After one week of acclimation, animals were divided into four groups of 10 mice each: Group 1, control group receiving normal saline solution (0.9% NS); Group 2, arsenic-treated group (5 mg/kg As2O3); Group 3, dual treatment group (400 mg/kg GSPE and 5 mg/kg As2O3); Group 4, GSPE-treated group (400 mg/kg). All mice were administered their respective treatments daily by gavage for a total of 5 weeks. At the end of the treatment period, mice were sacrificed and the kidneys were then collected. All experiments involving animals were conducted with the approval of the Ethics Committee of Shihezi University (NO. 2015-076-01).
The ELISA kits used to detect IL-1β (NO. L151214485), IL-6 (NO. L151210437), IL-10 (NO. L151221646), CRP (NO. L151110355), and TNF-α (NO. 151102081) were purchased from Cloud-Clone Corp (Katy, TX, USA). All other chemicals were of analytical grade and were obtained commercially.
Kidney tissue homogenates were generated by suspending tissues in total protein extraction lysis buffer containing phenylmethane sulfonyl fluoride (PMSF), protease inhibitors, and a phosphatase inhibitor (Sigma Aldrich, St. Louis, MO, USA), incubating on ice for 30 min, and centrifuging at 12, 000 rpm at 4 ℃ for 10 min. Analysis of proteins of phosphorylated (P)-IκB-α, IκB-α, IKKα/β, NF-κBp65, NF-κBp50 were then conducted by Western blotting techniques, with primary antibodies (all purchased from Abcam, Cambridge, UK) and the appropriate secondary antibodies (Zsbio, Beijing, China). Immunocomplexes were visualized using an enhanced chemiluminescence (ECL) Plus kit (GE, Connecticut, USA) and film. All experiments were repeated three times.
Total RNA was extracted using TRizol reagent (Invitrogen, Grand Island, NY, USA). RNA (5 μL) samples were reverse-transcribed into cDNA using a Transcriptor First-Strand cDNA Synthesis Kit (Roche, Indianapolis, IN, USA). The relative expression levels of genes of interest were then determined via qPCR using the reverse primer (Supplemental Table 1, available in www.besjournal.com). All samples were tested in triplicate. Relative expression levels were calculated using β-actin.
Name Gene Sequence (5' to 3') MACTBF TTCCTTCTTGGGTATGGAAT MACTBR GAGCAATGATCTTGATCTTC IκBαF CCAGGTAGCCGTGGATAGAG IκBαR AGCAGTCTTGACGCAGACCT IKKbetaF CCAGGTAGCCGTGGATAGAG IKKbetaR AGCAGTCTTGACGCAGACCT IkkaF TACAAGAGAGCGGGCAGAAC IkkaR GGAAGTGGCCCTCAGTAACA p65F CACTGTCACCTGGAAGCAGA p65R GACCTGGAGCAAGCCATTAG NF-kappaB P50F AGCCCCTAATACACGCCTCT NF-kappaB P50R TAACAGCAGGACCCAAGGAC Table Supplemental Table 1. Sequences of Primers Used in this Study
Data are presented as means ± standard deviations. A general linear model was used to analyze the effects of the combination of As2O3 and GSPE in a two-factor, two-level factorial design. Analysis of variance (ANOVA) was used to evaluate differences among the experimental groups. P values < 0.05 were considered statistically significant.
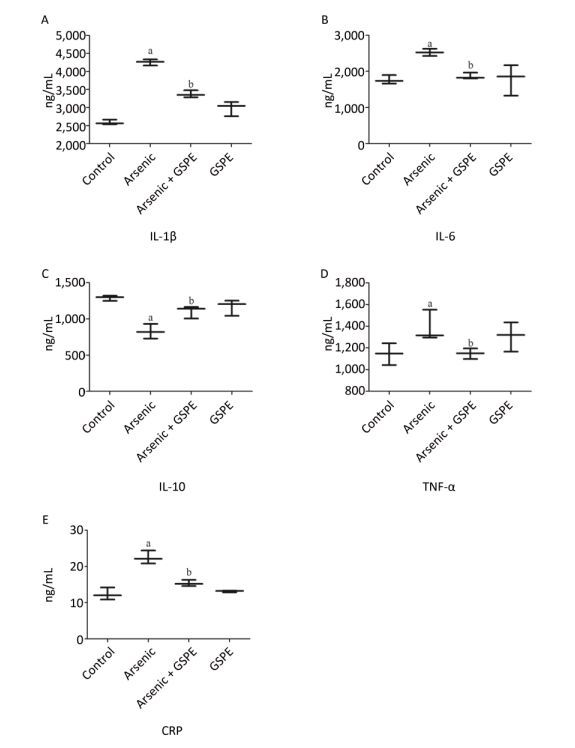
Compared to the NS control group, treatment with GSPE alone had no significant effect on cytokine production. Conversely, treatment with As2O3alone caused a marked increase in the production of the pro-inflammatory cytokines IL-1β, IL-6, TNF-α, and CRP (P < 0.05 for each), and a significant decrease in the production of the anti-inflammatory cytokine IL-10 (P < 0.05). However, each of these effects was abrogated by treatment with As2O3in the presence of GSPE (P < 0.05, compared to the As2O3 group for each) (Supplemental Figure 1, available in www.besjournal.com). These results, consistent with previous study[7], suggested that consumption of As2O3caused inflammation-dependent kidney damage in mice. In addition, we demonstrated that GSPE abolished the previous effects of As2O3. Likewise, Terra X et al. found that procyanidins reduced the mRNA expression of CRP and of TNF-α and IL-6 in the livers and adipose tissues, respectively, of rats exposed to high-fat diets, suggesting procyanidins prevented inflammation via diminishing proinflammatory molecules and enhancing anti-inflammatory ones[8]. Nevertheless, there was no further study of the anti-inflammatory mechanism of procyanidins. Thus, the identification of the mechanism of GSPE could provide an optimal method for treating arsenic-related disease.
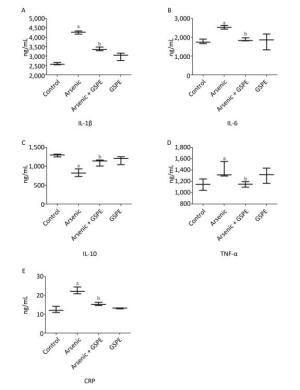
Figure Supplemental Figure 1. Evaluation of the effects of grape seed procyanidin extract (GSPE) treatment on arsenic-induced inflammation. Mice (n = 10 per group) were provided normal saline (control), arsenic trioxide (As2O3) alone (5 mg/kg), GSPE alone (400 mg/kg), or As2O3 and GSPE (5 and 400 mg/kg, respectively) for 5 weeks. At the end of the experimental period the mice were sacrificed, and their kidneys were harvested, homogenized, and subjected to ELISA analysis for the quantification of (A) interleukin (IL)-1β, (B) IL-6, (C) IL-10, (D) tumor necrosis factor (TNF)-α, and (E) C-reactive protein (CRP) production levels. aP < 0.05, compared to the normal saline (NS) control group; bP < 0.05, compared to the As2O3 group.
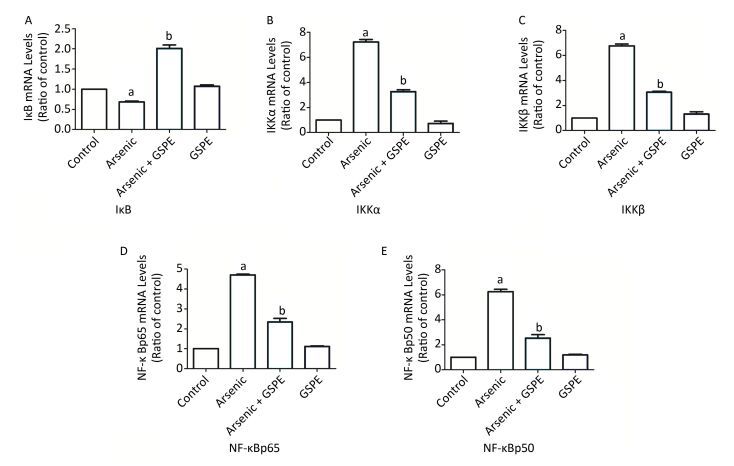
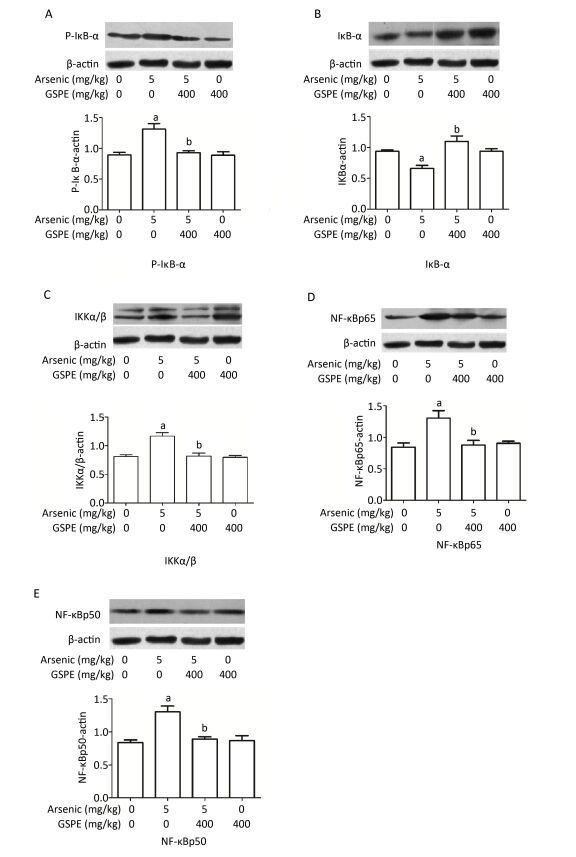
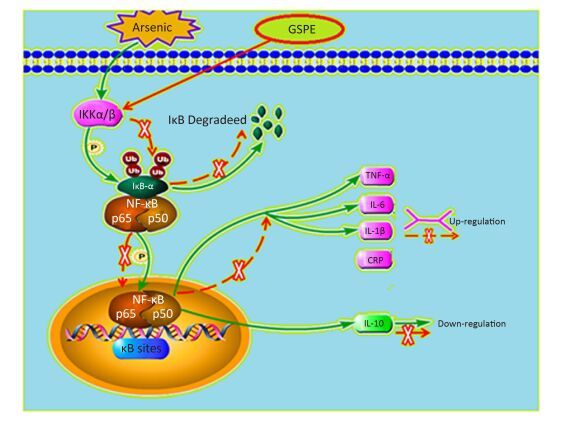
Therefore, we next characterize the effects of arsenic and GSPE on the activation status of the NF-κB pathway, kidney tissues from mice in each group were subjected to qRT-PCR and Western blot analysis to evaluate the expression levels of factors involved in this pathway. While treatment with As2O3 alone promoted the expression of IKKα, IKKβ, NF-κBp65, and NF-κBp50, and inhibited IκB expression, compared to the control population (P < 0.05 for each), the mice treated with As2O3 and GSPE exhibited decreased mRNA expression of IKKα, IKKβ, NF-κBp65, and NF-κBp50, and increased IκB expression (P < 0.05 for each). Meanwhile, treatment with GSPE alone had no effect on the expression levels of these factors, compared to the control group (Figure 1). Similar results were obtained by Western blot analysis (Figure 2). Consistent with these findings, in a previous study, Roy et al.[9]demonstrated that exposure to 3 mg/kg sodium arsenite (NaAsO2) for 2 weeks promoted renal inflammatory injury in rats via activation of the NF-κB pathway. On the contrary, treatment with 100 mg/kg and 250 mg/kg GSPE for 22 weeks significantly relieved inflammation in human proximal renal tubular epithelial cells in vitro by inhibiting the nuclear translocation of NF-κBp65 and the expression of IL-1β[10]. Together, combined with the present sduty, these data suggest a mechanism by which As2O3promotes the phosphorylation and consequent degradation of IκB protein by triggering the expression of IκB kinase (IKKα/β), thereby activating NF-κBp65 and NF-κBp50. These NF-κB dimers subsequently translocate to the nucleus where they interact with and activate genes harboring the specific κB binding site, including those encoding inflammatory cytokines (Figure 3). Whereas, GSPE may restrain the As2O3-mediated inflammatory damage by overturning the As2O3-dependent phosphorylation and degradation of IκB by obstructing the activation of IKKα/β expression, thereby inhibiting the expression of NF-κBp65 and NF-κBp50, and consequently, the expression of pro-inflammatory cytokines (Figure 3).
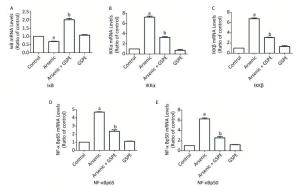
Figure 1. Evaluation of the effects of grape seed procyanidin extract (GSPE) treatment on the mRNA expression levels of components of the nuclear factor (NF)-κB pathway. Mice (n = 10 per group) were provided normal saline (control), arsenic trioxide (As2O3) alone (5 mg/kg), GSPE alone (400 mg/kg), or As2O3 and GSPE (5 and 400 mg/kg, respectively) for 5 weeks. At the end of the experimental period, the mice were sacrificed, and their kidneys were harvested, homogenized, and subjected to qRT-PCR analysis to evaluate the expression levels of (A) IκB, (B) IKKα, (C) IKKβ, (D) NF-κBp65, and (E) NF-κBp50. aP < 0.05, compared to the normal saline (NS) control group; bP < 0.05, compared to the As2O3 group.
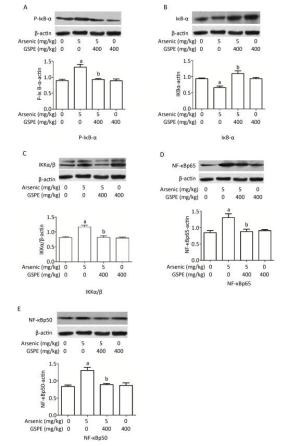
Figure 2. Examination of the effects of grape seed procyanidin extract (GSPE) treatment on arsenic-induced activation of the nuclear factor (NF)-κB pathway. Mice (n = 10 per group) were provided normal saline (control), arsenic trioxide (As2O3) alone (5 mg/kg), GSPE alone (400 mg/kg), or As2O3 and GSPE (5 and 400 mg/kg, respectively) for 5 weeks. At the end of the experimental period the mice were sacrificed, and their kidneys were harvested, homogenized, and subjected to Western blot analysis to evaluate the levels of (A) phosphorylated (P)-IκB-α, (B) IκB-α, (C) IKKα/β, (D) NF-κBp65, (E) and NF-κBp50. Top panels show images of each respective Western blot, while bottom images show graphic depictions of the quantities of each protein detected in the corresponding blot, as determined by densitometry analysis. aP < 0.05, compared to the normal saline (NS) control group; bP < 0.05, compared to the As2O3 group.
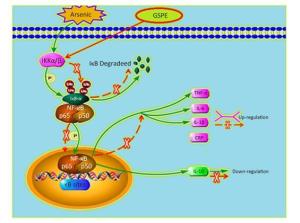
Figure 3. Model depicting the proposed mechanism by which grape seed procyanidin extract (GSPE) ameliorates arsenic-induced kidney damage in mice by inhibiting the inflammatory response (the 20% size was curtailed).
In summary, our findings suggest that GSPE comprises an effective inhibitor of arsenic-induced kidney damage in mice. Specifically, while As2O3induces inflammation-related kidney damage via activation of the NF-κB pathway, which in turn promotes the expression of inflammatory cytokines and inhibits the expression of IL-10, GSPE alleviates these effects by inactivating NF-κB signaling. Together, these data provide a theoretical basis for the development of treatments for preventing and/or controlling the toxicity of arsenic via the utilization of GSPE. However, further studies are needed to confirm the effects of GSPE on arsenic-related kidney damage in vitro and in humans.
WANG Cheng, LI Jun, LI Shu Gang, and LI Feng conceived of and designed the experiments. SONG Guan Ling, NIU Qiang, XU Shang Zhi, FENG Gang Ling, WANG Hai Xia, and LI Yu performed the experiments. WANG Cheng and LI Shu Gang analyzed the data. SONG Guan Ling, NIU Qiang, XU Shang Zhi, and FENG Gang Ling contributed reagents, materials, analytical tools. WANG Cheng and LI Jun wrote the paper. LI Shu Gang and LI Feng contributed to paper revisions.
Grape Seed Procyanidin Extract Reduces Arsenic-Induced Renal Inflammatory Injury in Male Mice
doi: 10.3967/bes2017.071
the Key Areas of Science and Technology Research Project of Xinjiang Production and Construction Corps 2014BA039
the International Cooperative Project of Shihezi University GJHZ201602
the Key Areas of Science and Technology Research Project of Xinjiang Production and Construction Corps 2015AG014
National Natural Science Foundation of China 81560517
- Received Date: 2017-04-10
- Accepted Date: 2017-06-15
| Citation: | WANG Cheng, LI Jun, SONG Guan Ling, NIU Qiang, XU Shang Zhi, FENG Gang Ling, WANG Hai Xia, LI Yu, LI Shu Gang, LI Feng. Grape Seed Procyanidin Extract Reduces Arsenic-Induced Renal Inflammatory Injury in Male Mice[J]. Biomedical and Environmental Sciences, 2017, 30(7): 535-539. doi: 10.3967/bes2017.071 |







 Quick Links
Quick Links
 DownLoad:
DownLoad: