-
Cilots in an accelerating aircraft or spaceship are often exposed to the force of acceleration (hypergravity), especially the high sustained positive acceleration (+Gz) force acting along the body axis from head to toe. In some extreme cases, pilots of modern high-performance fighter aircrafts have to suffer an acceleration as high as +9 Gz for 15 s to 45 s[1, 2]. Repeated exposure to high +Gz force can cause a series of physiological reactions in the body, including an increase in body weight and hydrostatic pressure, redistribution of blood to the lower body, and cerebral ischemia. These changes can damage the heart, brain, and nervous system[3-5].
Fighter pilots who had undergone flight training in a high +Gz environment had increased bone mineral density in their cervical and thoracic vertebrae[6, 7]. Some studies have reported the effects of hypergravity on the load-bearing bone in different species. For example, animal studies in a 2 G hypergravity environment showed that the femur trabecular volume was increased in mice and the cortical bone was thickened[8-10], but bone architectural changes were inconsistent in mice that lived in a 3 G hypergravity environment. Kawao et al. found that the mineral content of tibial trabecular bone in mice was increased in a 3 G hypergravity environment[11]; while Gnyubkin et al. found that the cortical bone was thinned out, and bone formation rate was decreased in mice that lived in a 3 G hypergravity environment[9]. The mechanisms that explain these changes are not clear yet, but we propose that a high hypergravity environment might have an adverse effect on bone formation.
It is believed that hypergravity environment can promote the differentiation of bone marrow mesenchymal stem cells into osteoblasts[12-14], and influence the proliferation and differentiation of osteoblasts. From previous studies, we concluded that there might be a ‘critical point’ for the effects of hypergravity on osteoblasts and osseointegration. Below this ‘critical point’ hypergravity condition may promote osteoblast proliferation and inhibit differentiation[15-18]. In contrast, above the ‘critical point’, the hypergravity condition may inhibit osteoblast proliferation and promote differentiation[19-21].
In some countries, dental implant therapy has been applied in military pilots cautiously[22]. Until now, there are little fundamental studies assessing the effect of high +Gz environment on osseointegration after tooth implant surgery. Therefore, the aims of our study are to observe the effect of high +Gz environment on dental implant osseointegration in a rabbit model and to investigate its mechanism.
-
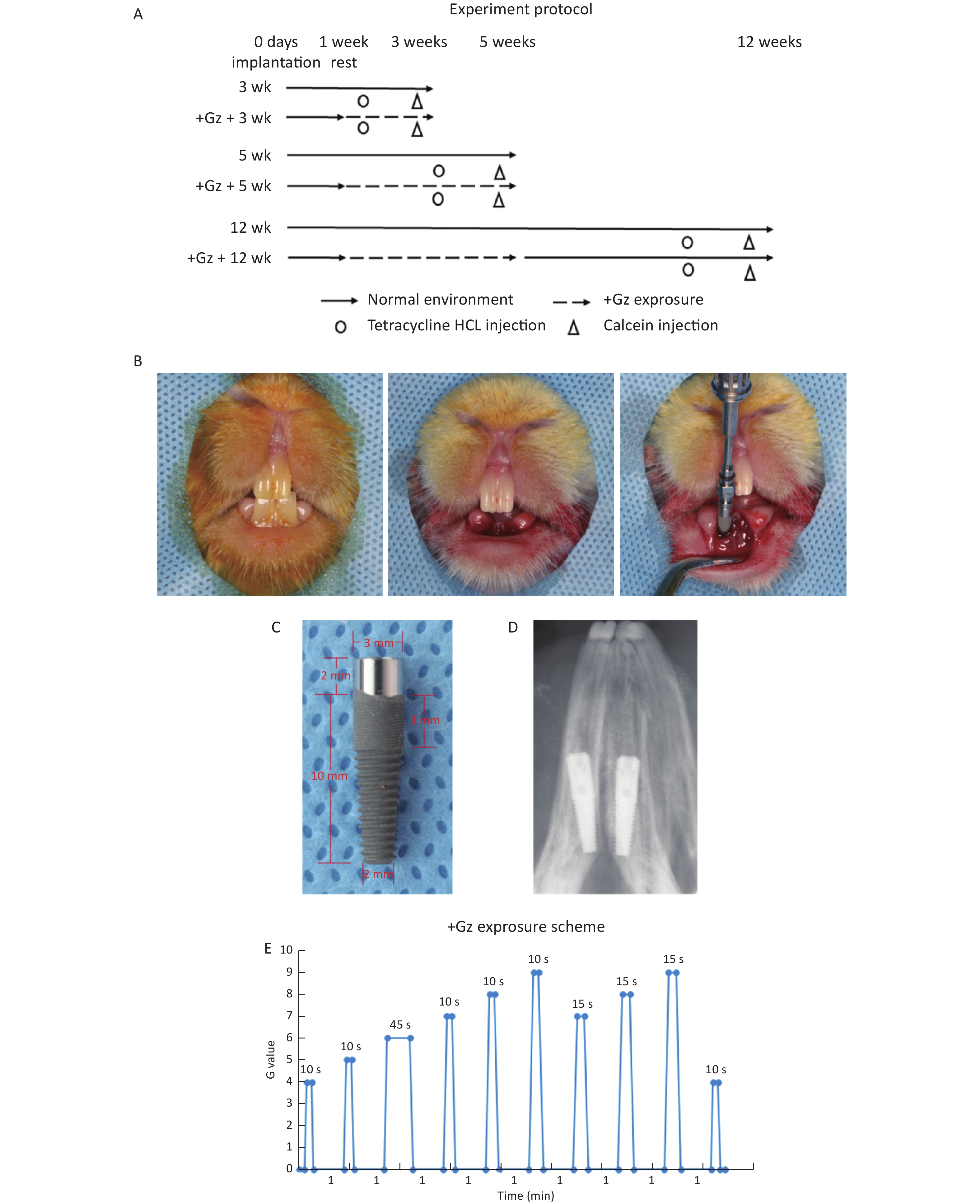
The screw-type titanium implants were manufactured by WeGo Group Co., Ltd (Yantai, Shandong, China). The implant size (3.0 mm in diameter and 12.0 mm in length) was specifically designed to match the length and diameter of the rabbit mandibular incisors. The implant was a screw-type implant with micro threads and a smooth neck of 2 mm (Figure 1C). The implant surface was treated with sand-blasting with large grit and acid-etching (SLA) technology. Afterward, the implants were sterilized by gamma radiation and double parked with a special design to ensure safe implant handling and placement, avoiding any hand contact.
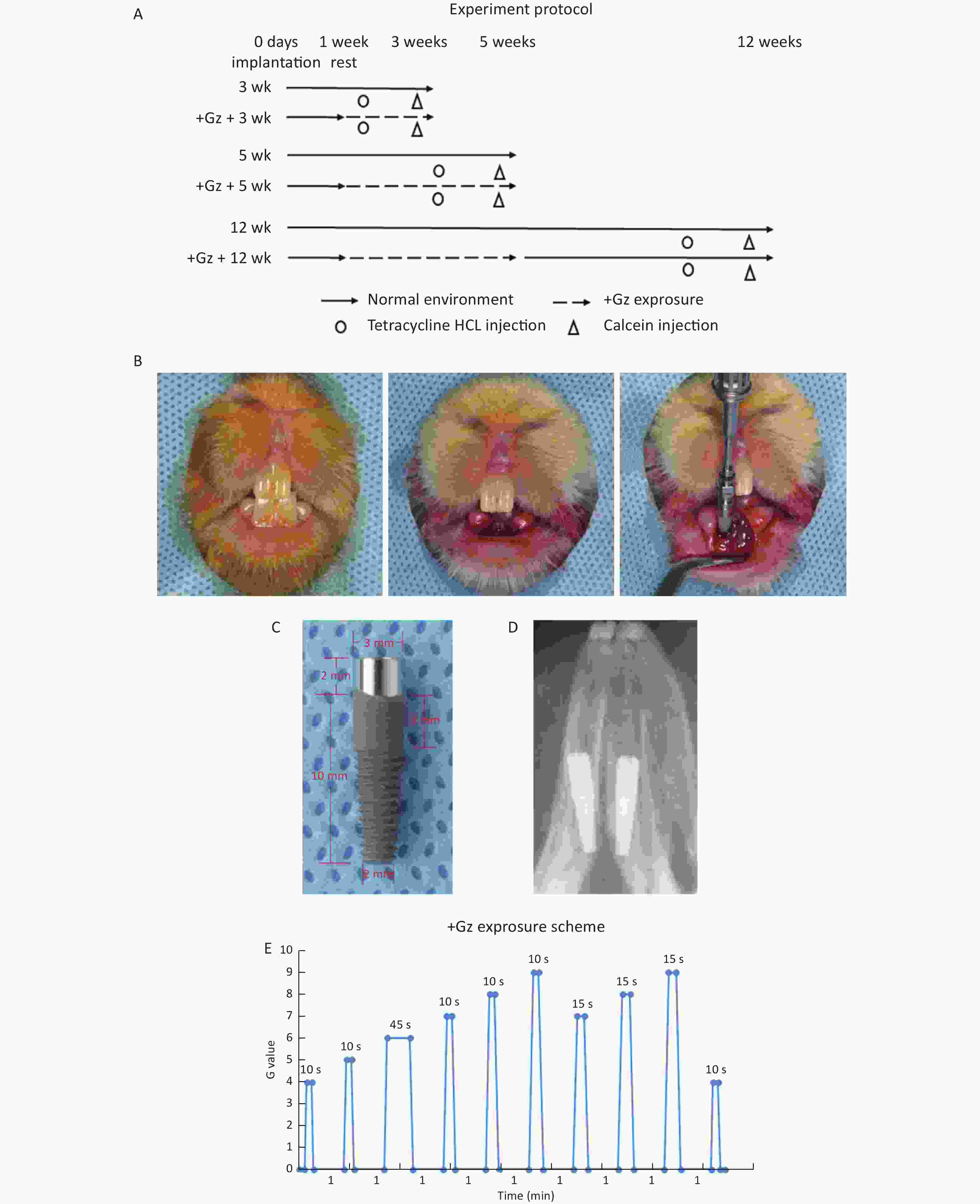
Figure 1. The effects of high +Gz environment on osseointegration after tooth implant surgery. (A) Experiment design and protocol. (B) Immediate implant placement surgery in rabbits. (C) View of the implant. (D) An X-ray film of the dental implant after immediate implantation. (E) The +Gz exposure protocol. wk, week.
-
Forty-eight adult, male New Zealand White rabbits (Oryctolagus cuniculus) weighing 2,485 (± 248) g were obtained from Beijing Vital River Laboratories. The animals were housed under appropriate environmental conditions, were exposed to 12-h light-dark cycles, and had access to standard food and water ad libitum. The protocol was approved by the Committee for Animal Experiments Ethics at the Air Force General Hospital, PLA, China (Protocol # kz2016006). All surgeries were performed under pentobarbital sodium anesthesia, and all necessary efforts to minimize animal discomfort were made.
The experimental scheme is illustrated in Figure 1A. The rabbits were randomly distributed into 6 equal groups (3 experimental groups and 3 control groups, 8 rabbits in each group). After one week of acclimation, the rabbits were anesthetized by an intraperitoneal injection of 3% pentobarbital sodium (1.2 mL/kg weight) (Sinopharm Chemical Reagent, Beijing, China) and were locally anesthetized with primacaine adrenaline (Produits Dentaires Pierre Rolland, Merignac, France). Bilateral mandibular incisors were extracted with minimal trauma to preserve the cortical bone. The implants were immediately placed into each tooth socket, and primary stability of the implants was achieved manually at about 15 N·cm. The gingiva was then closely sutured (Figure 1B, 1D). After surgery, all the rabbits had an intramuscular injection of penicillin (4 × 105 U/d) (North China Pharm, Shijiazhuang, Hebei, China) every day for 3 days.
-
One week after surgery, the experimental rabbits were exposed to high +Gz environment 3 times a week for 2 or 4 weeks in an animal centrifuge, while the control rabbits were raised in a common environment. The rabbits in one experimental group were exposed to +Gz for 2 weeks, and then sacrificed along with those in a control group. The rabbits in the other two experimental groups were exposed to +Gz for 4 weeks. Then the rabbits in one experimental group and one control group were killed at the end of 4 weeks. The rabbits in the 3rd experimental group were raised in a common environment for another 7 weeks after +Gz exposure, and then sacrificed along with those in the 3rd control group.
+Gz exposure was accomplished by using a specially designed animal centrifuge provided by the Air Force Aeromedicine Institute (Beijing, China). The animal centrifuge had an arm length of 1 m and an acceleration range of 1-15 G. Each rabbit was placed inside a cuboid restraint device, which was mounted in the centrifuge arm with the head of the rabbit facing the axis of the centrifuge for +Gz orientation. During the intervals between centrifuge runs, each rabbit was allowed to move freely in the cage.
The +Gz exposure protocol (Figure 1E) was designed according to the national[23] and international[24] military standards. The growth rate of +Gz was 1 G/s, the peak value was 4-9 G, the peak duration was 10-45 s, and the interval time was 1 min. The experimental rabbits were exposed to +Gz 3 times a week (every Monday, Wednesday, and Friday).
-
To monitor new bone formation around the implants, the rabbits were subcutaneously injected with calcein (10 mg/kg body weight) (Sigma Chemical Co., St. Louis, MO, USA) 14 days before being sacrificed and with tetracycline hydrochloride (30 mg/kg body weight) (Sigma Chemical Co., St. Louis, MO, USA) 4 days before being euthanized[25, 26].
The rabbits were sacrificed by an intravenous injection of pentobarbital sodium (Sinopharm Chemical Reagent, Beijing, China). Then, the soft tissues were stripped off and both sides of the jaw were harvested. The left-sided implants and the surrounding bone were fixed in 70% ethanol for micro-CT examination and histological observation. The right-sided implants and the surrounding bone were carefully collected using a Φ5 mm trephine bur (GC Dental, Tokyo, Japan); afterward, the surrounding 1 mm bone was scraped from the implant and frozen at −80 °C until analysis.
-
To evaluate the mass and microarchitecture of the bone surrounding the implants, micro-CT was performed using the Inveon MM system (Siemens, Munich, Germany). Images were acquired with an effective pixel size of 8.99 μm, voltage of 80 kV, current of 500 μA, and exposure time of 1,500 ms during each of the 360 rotational steps. The bone histomorphometric parameters were calculated using the Inveon Research Workplace (Siemens) as follows: bone volume fraction (BV/TV), trabecular thickness (Tb.Th), trabecular number (Tb.N), and trabecular separation (Tb.Sp) in the area of 1 mm around the middle and lower 2/3rds of the implant.
-
After micro-CT analysis, the specimens were dehydrated in an ascending series of ethanol, cleared in chloroform, embedded in methyl methacrylate, sliced into sections of approximately 200 μm along the buccal-lingual axis of the implant by using a rotary diamond saw (SP1600, Leica, Nussloch, Germany), and then ground to a final thickness of approximately 30 μm. The slices were observed under a fluorescence microscope (BX61, Olympus, Tokyo, Japan), and the mineral apposition ratio (MAR, the ratio of the mean vertical distance between the two fluorescence lines and the time interval) at different time points was quantitatively analyzed using Image-Pro Plus (Media Cybernetics, Silver Spring, MD, USA).
-
After fluorescence observation, the slices were stained with methylene blue (Sinopharm Chemical Reagent, Beijing, China) and acid magenta (Sinopharm Chemical Reagent, Beijing, China) and observed under a microscope (BX61, Olympus). The bone implant contact (BIC, the percentage of the linear surface of the implant in direct contact with the mineralized bone) was quantitatively analyzed using Image-Pro Plus. BIC was measured as a range covering the mesial and distal sides of the middle and lower 2/3rds of the long axis of the implant, excluding the bottom of the implant.
-
Total cellular RNA was extracted from the frozen bone tissue around the implants using the Ultrapure RNA Kit (CoWin Bioscience, Beijing, China) following the manufacturer’s instructions. The concentration of RNAs was analyzed by Nanodrop ND-2000 (Thermo, Wilmington, DE, USA) at a wavelength of 260 nm. The purity of RNAs was also determined by the ratio of RNA OD260/OD280. Isolated RNA (2 μg) was reverse-transcribed into cDNA in a reaction mixture containing 4 μL of 5× RT buffer, 4 μL of dNTP mixture (2.5 mmol/L), 2 μL of primer mix, and 1 μL of SuperRT (200 U/μL) in a total volume of 20 μL using the SuperRT One-Step RT-PCR Kit (CoWin Bioscience, Beijing, China). The reaction mixture was incubated at 50 °C for 50 min and at 85 °C for 5 min in a Veriti 96-well Fast Thermal Cycler (Applied Biosystems, Foster City, CA, USA).
Real-time PCR was performed on the Roche LightCyler 480 PCR system (Roche, Basel, Switzerland) using the Power SYBR green PCR Master Mix (Applied Biosystems, Foster City, CA, USA). Primer sequences (forward and reverse) of the detected genes and the control gene GAPDH (Table 1) were designed according to the published sequences by using Primer Premier 5.0 (Premier Biosoft, Palo Alto, CA, USA) and synthesized by Applygen Technologies Inc (Beijing, China). The PCR amplification reaction mixture (in a final volume of 25 μL) contained 12.5 μL of PCR Master Mix, 1 μL of cDNA, 1 μL of forward primers, and 1 μL of reverse primers for the detected genes and GAPDH. PCR conditions used were as follows: pre-denaturation for 10 min at 95 °C, denaturation for 15 s at 95 °C, and annealing for 15 s at 60 °C and extension for 35 s at 72 °C for a total of 40 cycles. Each individual cDNA sample for each gene was assayed in triplicate. A control sample without cDNA was used in each run to exclude genomic DNA contamination. After performing PCR, the Ct values of each sample were collected using MxPro-Mx3005p QPCR Software (Agilent, Santa Clara, CA, USA).
Target gene Forward primer (5’→3’) Reverse primer (5’→3’) BMP-2 TGAGGATTAGCAGGTCTTT GCTGGATTTGAGGCGTTT OPN GCTAAACCCTGACCCATCT CGTCGGATTCATTGGAGT TGF-β1 AGGACCTGGGCTGGAAGTG GCGCACGATCATGTTGGA RANKL AATGCCCGATTCATGTAGGAG AGATCGAACCATGAACCTTCC OPG GATCCAGAAACCTCTCGTCAG TGTGCCAAGTGTCTGTGTAG GAPDH GGTCGGAGTGAACGGATTT CTCGCTCCTGGAAGATGG Note. BMP-2, bone morphogenenetic protein-2; OPN, osteopontin; TGF-β1, transforming growth factor-β1; RANKL, receptor activator of nuclear factor κB ligand; OPG, osteoprotegerin; GAPDH, glyceraldehyde-3-phosphate dehydrogenase. Table 1. Primer sequence of the detected genes
-
Statistical evaluation of the data was performed using the SPSS 19.0 software (IBM, Armonk, NY, USA). Mean values and standard deviations were calculated. The differences between the two groups were analyzed using two independent samples t-test, and the differences within groups were first analyzed using one-way ANOVA, followed by the Fisher’s Least Significant Difference (LSD) tests. Differences were considered statistically significant at P < 0.05.
-
All the rabbits went through the surgery and recovered well. No signs of inflammation were noticed at the surgical sites. The experimental rabbits could endure the predetermined +Gz exposure. Mean body weight and daily food consumption between the control and experimental rabbits were not significantly different during the entire experiment.
-
The images obtained by micro-CT showed that osseointegration was observed in all implants, and the trabecular bone had closely surrounded the implants at 12 weeks (Figure 2A, 2B). BV/TV, Tb.Th, and Tb.N values were increased, and the Tb.Sp value was decreased in both groups as the observation time period increased. In the control group, the changes that occurred from 3 weeks to 5 weeks were more obvious than the changes that occurred from 5 weeks to 12 weeks, and these changes were also more obvious than the changes that occurred from 3 weeks to 5 weeks in the experimental group. BV/TV and Tb.N values were significantly lower, while the Tb.Sp value was significantly higher in the experimental group [(38.80 ± 5.10)%, (4.79 ± 0.37)/mm, and (130.03 ± 17.98) μm respectively] compared with the control group [(47.85 ± 5.03)%, (5.40 ± 0.25)/mm, and (103.31 ± 11.13) μm respectively] at 5 weeks (all P < 0.05). However, there was no significant difference between the two groups at 3 weeks and 12 weeks (Figure 2C).
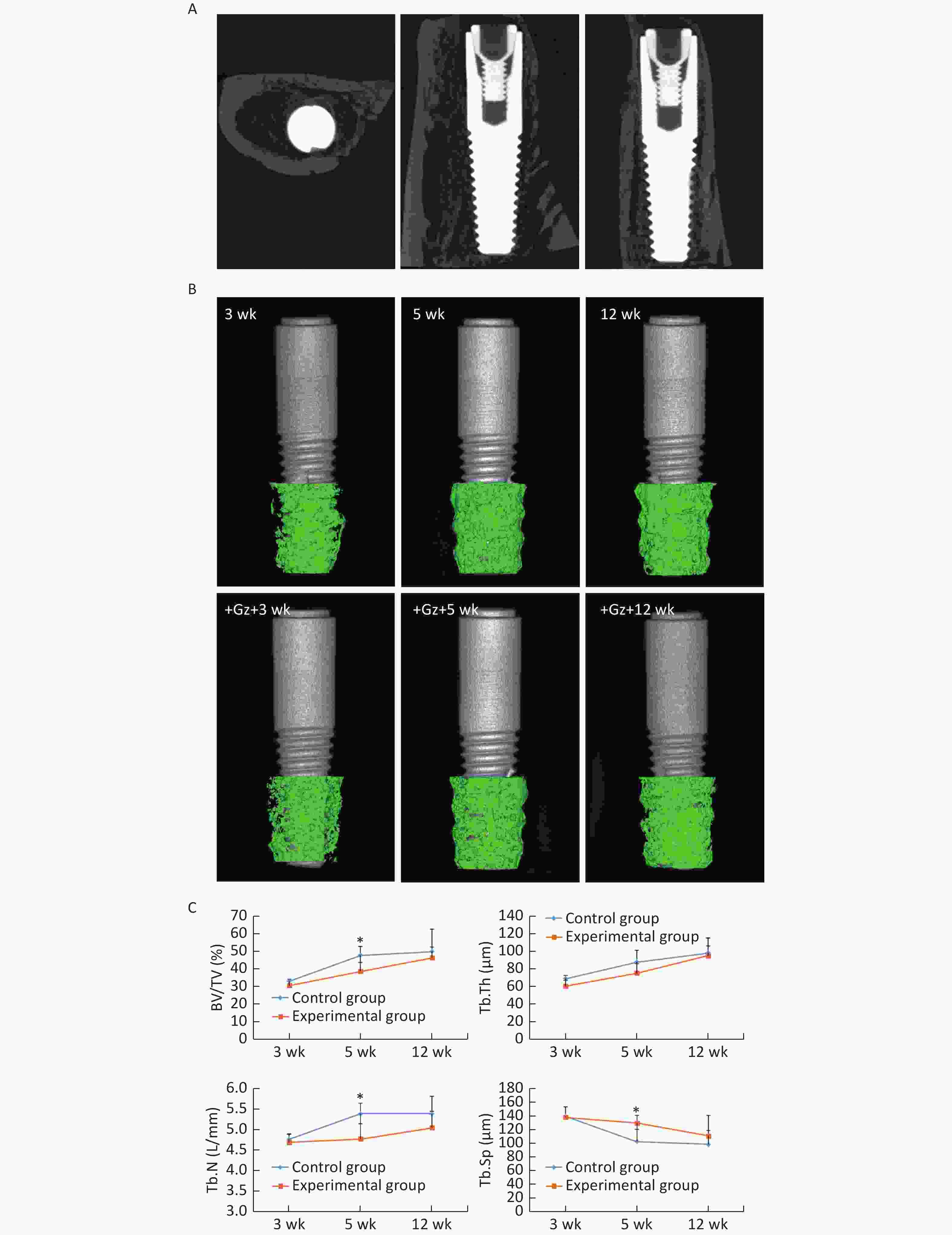
Figure 2. Micro-CT analysis of the implants. (A) Three-dimensional reconstruction of the implant in the experimental group at 12 weeks. (B) Three-dimensional reconstruction of the trabecular bone in the ROI area of the implants in each group. (C) The bone histomorphometric analysis in the ROI area of the implants. *P < 0.05 as compared to the control group at the same time point. wk, week.
-
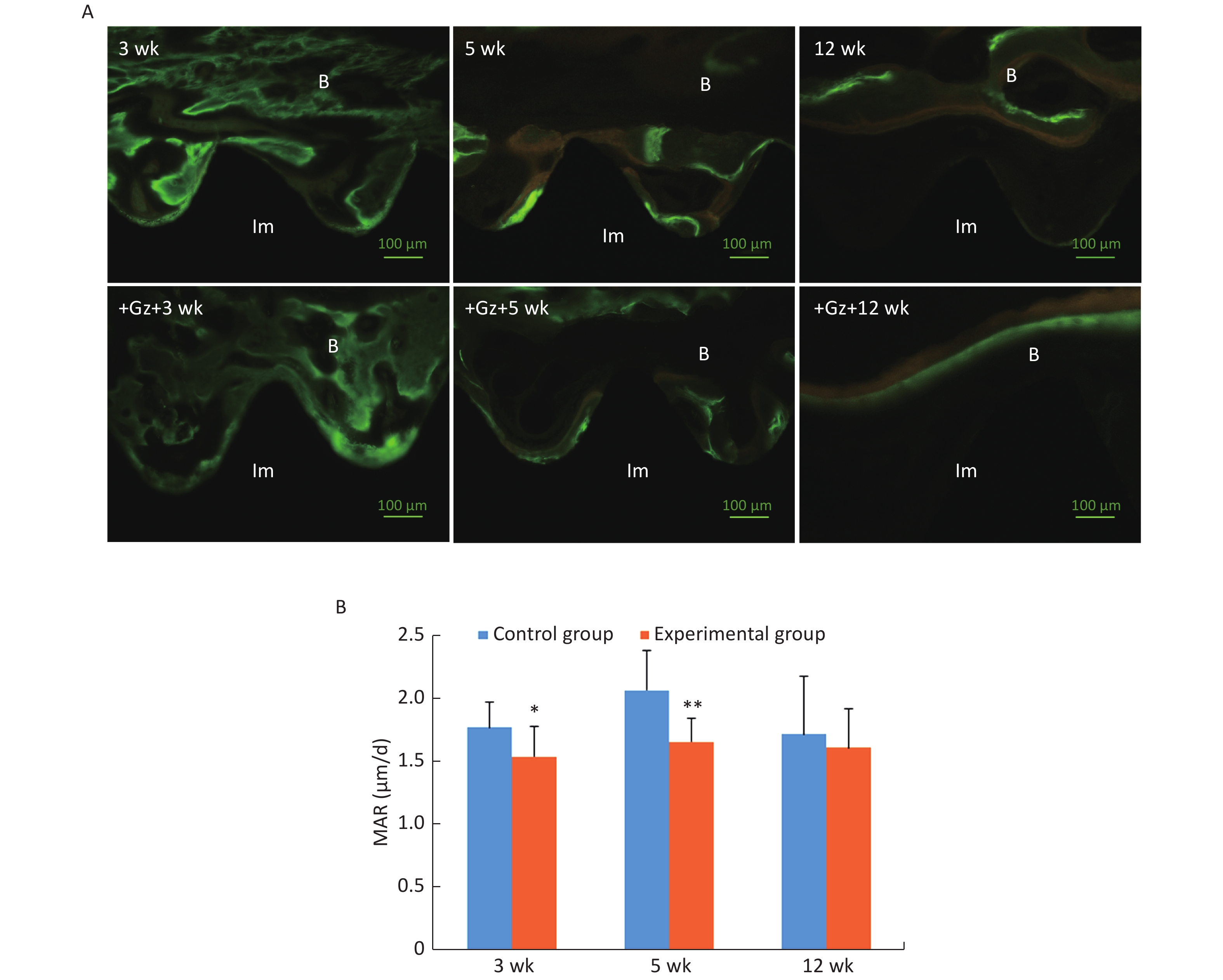
Under a fluorescence microscope, we were able to detect green fluorescence (calcein) near the implant surface and yellow fluorescence (tetracycline hydrochloride) further away from the implant surface. The interval between the outside of the green fluorescence and the outside of the yellow fluorescence was the new bone formed during double labeling.
Scattered green and yellow fluorescences were observed in the 3-week control slices, while yellow fluorescence was not very obvious in the 3-week experimental slices. Both linear green and yellow fluorescent ribbons were observed in the 5-week control slices, and the distance between the green ribbon and the yellow ribbon was obvious. The fluorescent ribbons were weak and uneven, and the distance between the green ribbon and the yellow ribbon was narrow in the 5-week experimental group. At 12 weeks after surgery, there was only a small green and yellow fluorescence overlap around the implants both in the control group and the experimental group (Figure 3A).
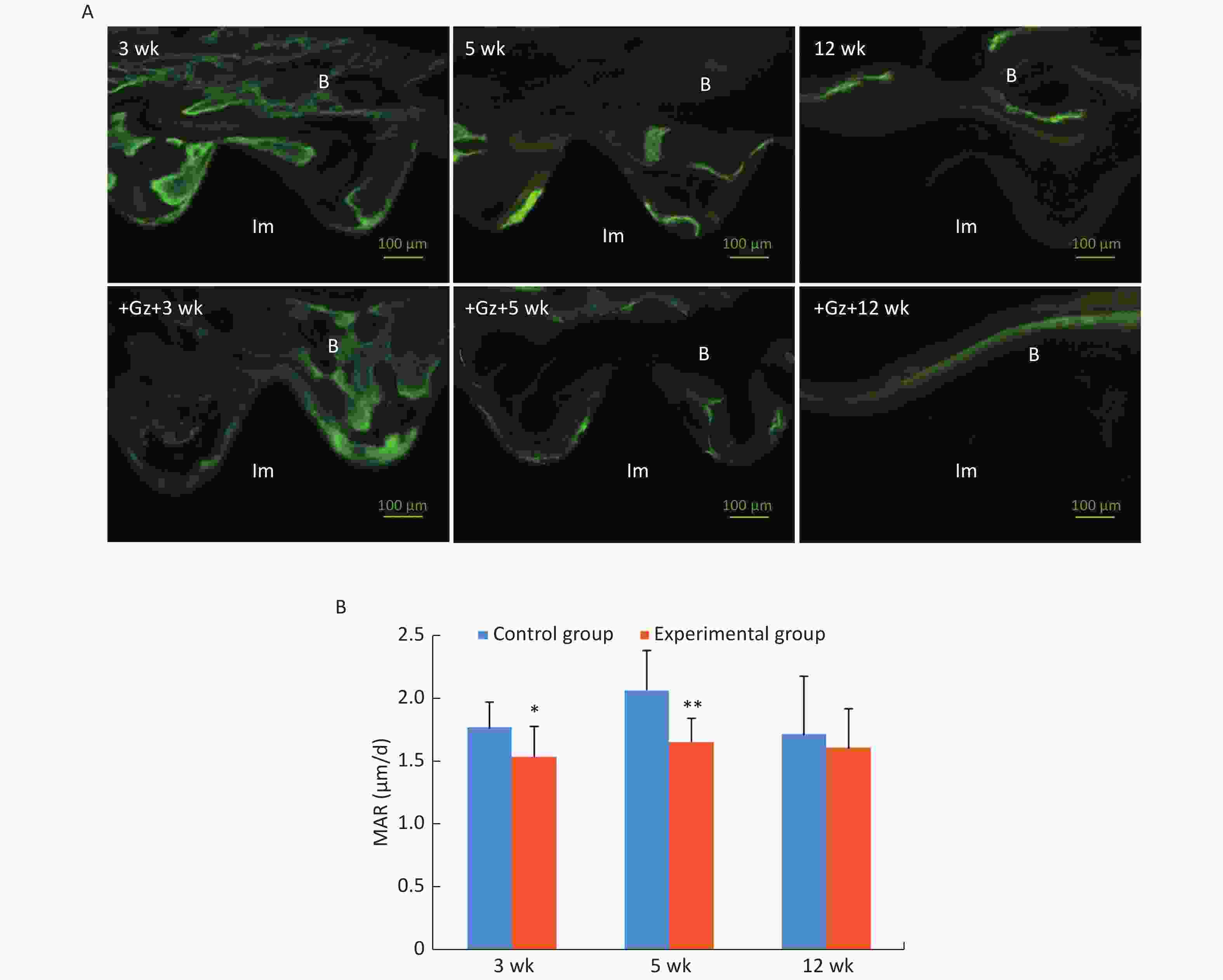
Figure 3. Fluorescence double labeling observation of the implants. (A) Fluorescence microscopic images with the double labeling of calcein and tetracycline hydrochloride in each group (200×) (Im: implant, B: bone). (B) Comparison of mineral apposition ratio (MAR) of the implants. Scale bar indicates 100 μm; *P < 0.05, **P < 0.01 as compared to the control group at the same time point. wk, week.
MAR in the experimental group at 3 weeks and 5 weeks after surgery [(1.54 ± 0.24) μm/d and (1.66 ± 0.19) μm/d, respectively] was significantly lower than that in the control group [(1.77 ± 0.20) μm/d and (2.07 ± 0.31) μm/d, respectively] (3 weeks P < 0.05, 5 weeks P < 0.01) (Figure 3B).
-
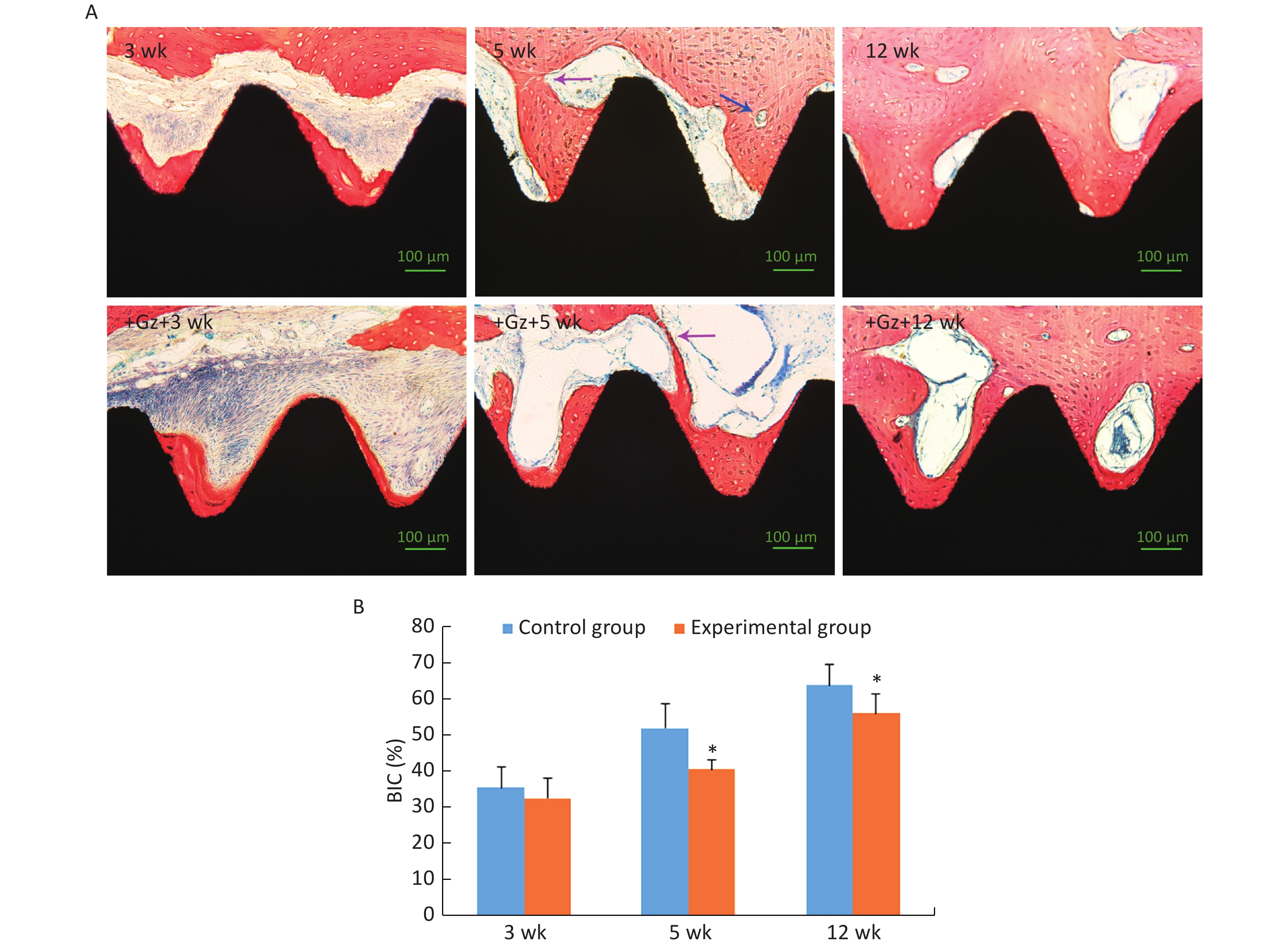
After 3 weeks of the healing period, the newly formed trabecular bone with osteoblasts at the edge was in direct contact with the partial implant surface, and it had spread over the implant surface, towards the host bone (contact osteogenesis). However, there were many inflammatory cells and loose connective tissue in some areas around the experimental implants. At 5 weeks after surgery, the newly formed peri-implant trabecular bone continued to spread over the implant surface and was connected with the new bone developed from the host bone cavity towards the implant surface (distance osteogenesis). Bone remodeling was active and mature trabecular bone was formed. At 12 weeks after surgery, all implants were surrounded by thick and dense lamellar bone (Figure 4A).
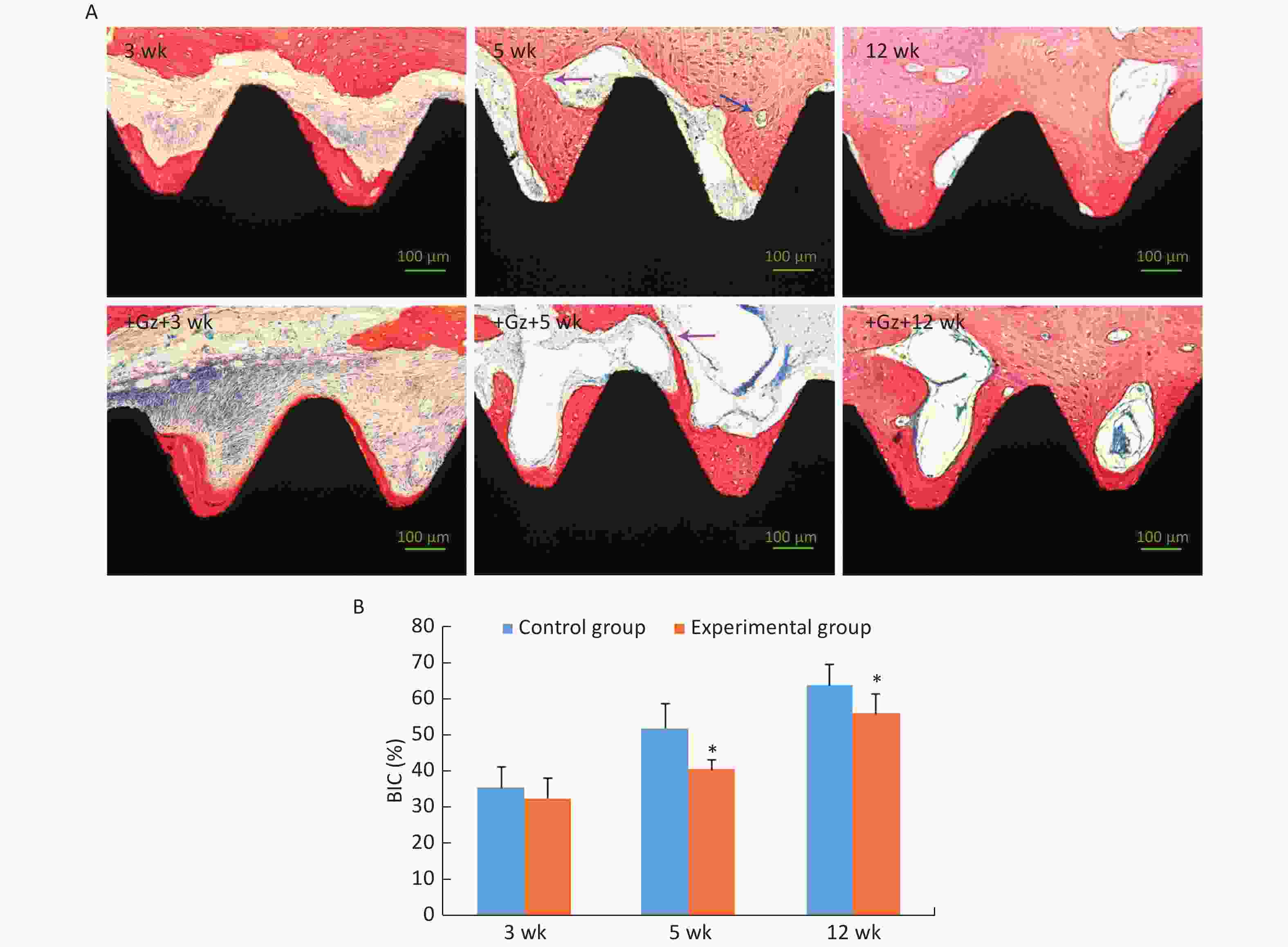
Figure 4. Histological examination of the implants. (A) Histological images of the implant-bone ground slices stained with methylene blue and acid magenta (200×). The purple arrow indicates the place where distance osteogenesis and contact osteogenesis were connected. The blue arrow indicates an osteoclast. Scale bar indicates 100 μm. (B) Comparison of bone-implant contact (BIC) of the implants. *P < 0.05 as compared to the control group at the same time point. wk, week.
The value of BIC was significantly increased in both groups as the observation time period increased, and it was significantly lower in the experimental group [(40.42 ± 2.85)% and (55.97 ± 5.27)%, respectively] than in the control group [(51.97 ± 6.74)% and (63.74 ± 5.56)%, respectively] at 5 weeks and 12 weeks (both P < 0.05) (Figure 4B). Even so, the value of BIC in the experimental group at 12 weeks was up to 55.97%, and it was adequate to ensure implant stability and long-term use[27, 28].
-
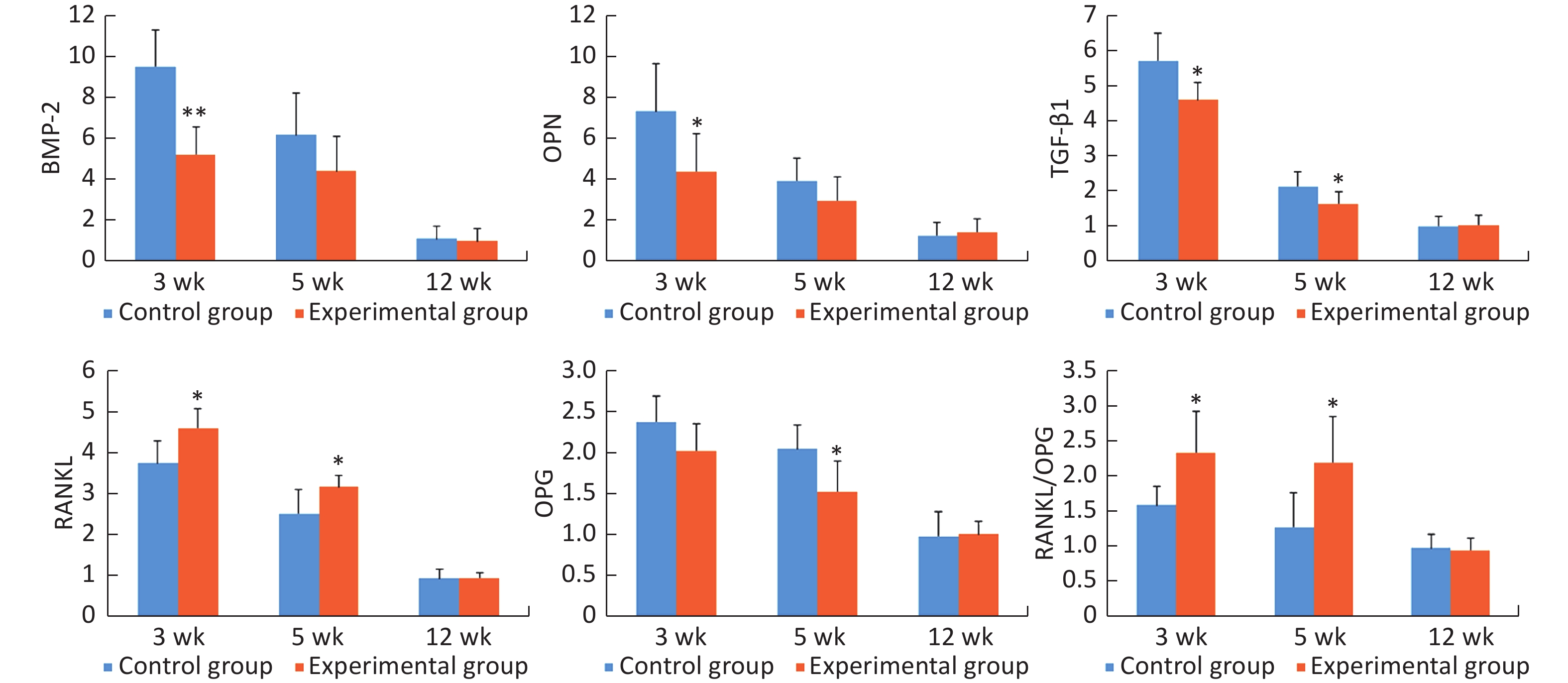
The mRNA expression levels of bone morphogenetic protein-2 (BMP-2), osteopontin (OPN), transforming growth factor-β1 (TGF-β1), receptor activator of nuclear factor κB ligand (RANKL), and osteoprotegerin (OPG) were highest at 3 weeks after surgery, and then, these levels decreased gradually at 5 weeks and 12 weeks. At 3 weeks after surgery, the mRNA expression levels of BMP-2, OPN, and TGF-β1 were significantly lower (0.55-, 0.60-, and 0.80-fold, respectively), and the mRNA expression level of RANKL and RANKL/OPG ratio were significantly higher (1.23- and 1.47-fold, respectively) in the experimental group than in the control group (all P < 0.05). At 5 weeks after surgery, the mRNA expression levels of TGF-β1 and OPG were significantly lower (0.77- and 0.75-fold, respectively), and the mRNA expression level of RANKL and RANKL/OPG ratio were significantly higher (1.26- and 1.72-fold, respectively) in the experimental group than in the control group (all P < 0.05). At 12 weeks after surgery, there was no obvious difference in the mRNA expression levels of the detected genes between the two groups (Figure 5).
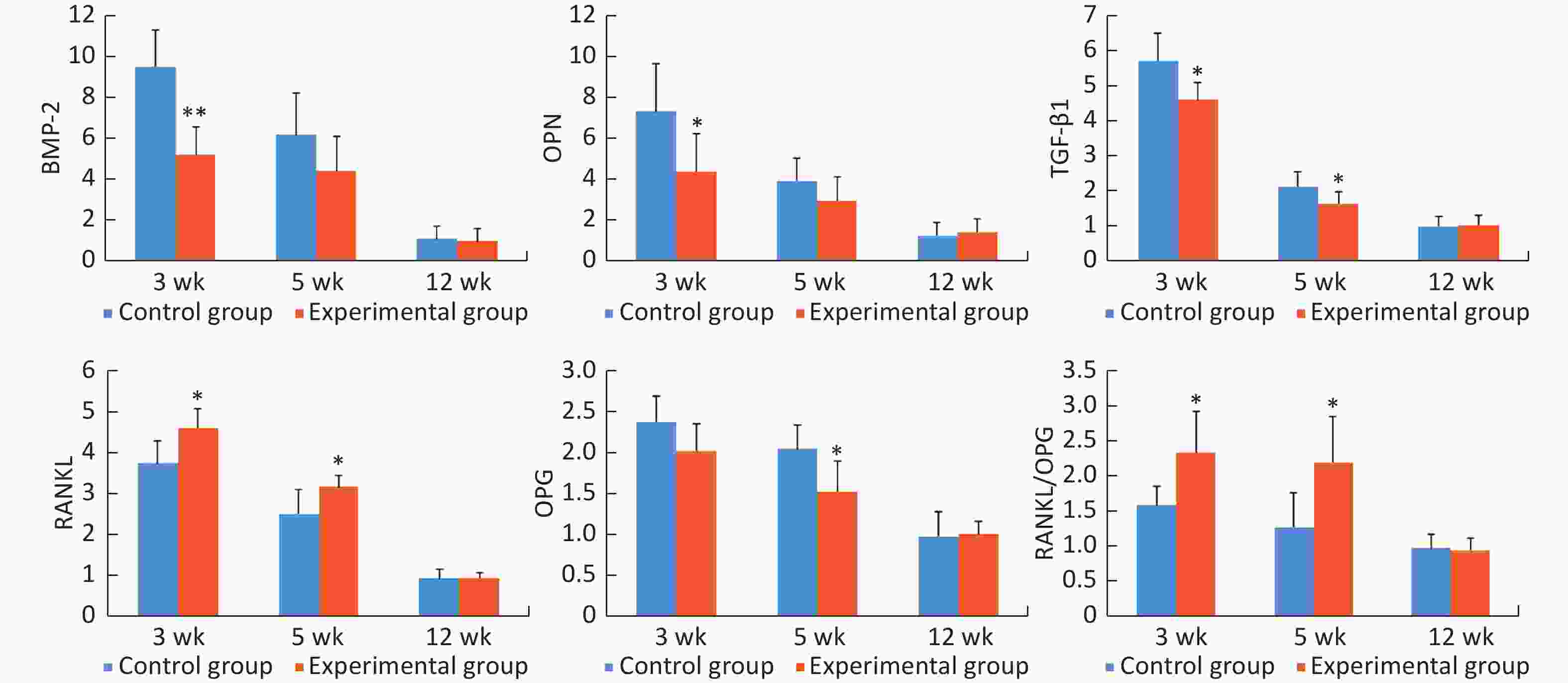
Figure 5. The relative mRNA expression levels of the detected genes in peri-implant bone tissues. *P < 0.05, **P < 0.01 as compared to the control group at the same time point. wk, week.
In summary, all implants had good osseointegration at 12 weeks. Differences in mRNA expression of osteoblast and osteoclast activity-related cytokines between the two groups were found mainly at 3 weeks and 5 weeks. Differences of the microarchitecture and osseointegration were found mainly at 5 weeks and 12 weeks.
-
In the present study, we found that early exposure to the high +Gz environment after tooth implant surgery can inhibit osseointegration of dental implants by inhibiting osteogenesis of osteoblasts and promoting bone resorption by osteoclasts. This is the first study to assess the effect of high +Gz environment on osseointegration. To elucidate the mechanisms of the effect of high +Gz environment, we further assessed the mRNA expression levels of some cytokines closely related to the function of osteoblasts and osteoclasts by using RT-PCR.
In this study, we used the rabbit incisor immediate implant model taking into account the experimental condition of the animal centrifuge and the anatomical features of the rabbit mandibular structure. Our animal centrifuge can only centrifuge small animals such as rabbits and rats, but cannot centrifuge large animals like pigs and dogs. In vivo studies that use rabbits to evaluate implant osseointegration are usually carried out in the tibia or the femur[29, 30]. However, the stress states of the jaw and the tibia or the femur are different when exposed to +Gz environment. Due to the rabbit's short mouth opening range, it is difficult to extract the teeth and place implants in the rabbit molar area. However, the alveolar bone in the anterior region of the rabbit is thin and a remarkable reduction in the height and width of the alveolar ridge often occurs following tooth extraction[31, 32]. Implant placement immediately after tooth extraction may prevent the occurrence of this atrophy. Indeed, as previously demonstrated by Weber et al.[33], the rabbit incisor immediate implant model is a feasible and successful in vivo model for the investigation of osseointegration.
We choose the middle and lower 2/3rds of the implant as the region of interest (ROI) both in micro-CT observation and histological evaluation because the middle and lower 2/3rds of the implant was more closely contacted by the surrounding alveolar bone after immediate implant placement.
We observed in this study that the values of BV/TV, Tb.Th, Tb.N, and BIC were gradually increased, and the Tb.Sp value was gradually decreased during the observation time period in the two groups. The changes that occurred from 3 weeks to 5 weeks were more obvious than the changes that occurred from 5 weeks to 12 weeks in the control group, indicating that new bone was formed mainly in the first 5 weeks after surgery. After 5 weeks, bone remodeling occupied the first place, mineralization of the new bone tissue was increased, and mature lamellar bone was formed. This process was similar to that described in previous studies[34-36]. Compared with the values in the control group, the values of BV/TV, Tb.N and BIC in the experimental group were significantly lower and the Tb.Sp value was significantly higher at 5 weeks. MAR in the experimental group was significantly lower than that in the control group both at 3 weeks and 5 weeks. These results indicated that exposure to the +Gz environment may slow down new bone formation and mineralization, and it might have an adverse effect on osseointegration. Nevertheless, both the micro-CT observation and histological evaluation of the specimens in this study showed that initial osseointegration was achieved in all implants after a healing period of 12 weeks. Some experts believe that the value of BIC in a clinically successful implant should be at least 50%[27, 28]. In our study, although the value of BIC in the experimental group was significantly lower than that in the control group at 12 weeks, both these values were above 50%. Good osseointegration was achieved to ensure the stability of the implants and their long-term use. This result indicated that the adverse effect of high +Gz environment on osseointegration was reversible if the exposure time was not very long.
The placement of an implant into a bone activates a wide range of biological reactions, including new bone formation and bone remodeling[37-39]. Therefore, osseointegration is a complex process involving a number of cytokines.
BMP-2 can promote the transformation of the surrounding mesenchymal stem cells into osteoprogenitors, and it plays an important role in the induction of osteogenesis[40, 41]. OPN is a characteristic marker of osteoblasts in the matrix formation and maturation stage, and it also plays an important role in bone formation and bone remodeling[42, 43]. TGF-β1 is essential for osteoblastic differentiation of mesenchymal precursors. It can induce the synthesis of BMPs and promote the production of osteoid and extracellular proteins, such as collagen, OPN, and osteonectin[44]. At 3 weeks, the mRNA expression levels of BMP-2, OPN, and TGF-β1 in the experimental group were significantly lower than those in the control group. At 5 weeks, the mRNA expression level of TGF-β1 in the experimental group was still significantly lower than that in the control group. These findings indicated that high +Gz environment may suppress the osteogenic activity of osteoblasts.
RANKL can bind to the receptor activator of nuclear factor κB (RANK) on osteoclast precursors, promote osteoclast precursor differentiation, increase osteoclast production, and regulate osteoclast-mediated resorption. OPG can also bind to RANK and block the RANKL/RANK interaction, and thus, it can inhibit osteoclastogenesis and bone resorption[45]. The balance of RANKL and OPG controls osteoclast activity; therefore, it is usually preferable to evaluate the RANKL/OPG ratio rather than the RANKL and OPG absolute values[46]. Our study showed that the expression level of RANKL mRNA and RANKL/OPG ratio in the experimental group were significantly higher than those in the control group both at 3 weeks and at 5 weeks, indicating that high +Gz environment can increase the activity of osteoclasts and promote bone resorption.
There were some limitations of the present study. The initial stability of implants is a key factor in osseointegration. Several studies have shown that micro-mobility greater than 150 μm may affect osseointegration of the implants, resulting in the implants being enwrapped by fibrous tissue[47-49]. As the implant site was the cancellous bone in the rabbit mandible, the implant torsional moment in our study was 15 N·cm, which is lower than the normal requirement of 30 N·cm[50]. This may have slightly reduced the initial stability of implants and increased the micro-mobility caused by high +Gz.
-
In conclusion, within the limitations of this study, we assessed the effect of high +Gz environment on dental implant osseointegration. We found that early exposure to the high +Gz environment after tooth implant surgery might have an adverse effect on osseointegration, but the effect of short time exposure on osseointegration was reversible. Furthermore, we demonstrated that high +Gz environment may inhibit osteogenesis of osteoblasts by decreasing the mRNA expression levels of BMP-2, OPN, TGF-β1, and OPG, and it may promote bone resorption by osteoclasts by increasing the mRNA expression level of RANKL and RANKL/OPG ratio. The outcome of this study is of great interest, and it has implications for the application of dental implants in fighter pilots and astronauts. Based on these results, we recommend that fighter pilots need longer recovery time after tooth implantation, and the exact recovery time should be studied further in future work.
-
ZHU Xiao Ru and KE Jie designed the study together. ZHU Xiao Ru and DENG Tian Zheng performed the experiments. PANG Jian Liang and LIU Bing analyzed the data. ZHU Xiao Ru wrote the manuscript. KE Jie reviewed the manuscript.
-
The authors declare no conflict of interest.
Effect of High Positive Acceleration (+Gz) Environment on Dental Implant Osseointegration:A Preliminary Animal Study
doi: 10.3967/bes2019.087
- Received Date: 2019-02-09
- Accepted Date: 2019-07-12
-
Key words:
- Positive acceleration /
- Osseointegration /
- Dental implant /
- Micro-CT /
- Real-time polymerase chain reaction /
- Osteoblast
Abstract:
| Citation: | ZHU Xiao Ru, DENG Tian Zheng, PANG Jian Liang, LIU Bing, KE Jie. Effect of High Positive Acceleration (+Gz) Environment on Dental Implant Osseointegration:A Preliminary Animal Study[J]. Biomedical and Environmental Sciences, 2019, 32(9): 687-698. doi: 10.3967/bes2019.087 |







 Quick Links
Quick Links
 DownLoad:
DownLoad: