-
HIV infection is usually characterized by high viral replication and progressive CD4+ T lymphocyte (CD4) depletion. However, some patients demonstrate more moderate disease progression. A subset of HIV-infected individuals that maintain CD4 counts exceed 500 cells/μL for more than 7 years without antiretroviral therapy (ART) are referred to as long-term nonprogressors (LTNPs). In some this load can last for 20-25 years[1]. With promotion of VL detection methods, another patient subgroup referred to as viremia controllers (VCs) emerged, these patients can maintain low viremia (VL < 2.7 or 3.3 log 10 copies/mL) without ART. Some VCs maintain VLs below the test line (1.7 log 10 copies/mL) are known as elite controllers (ECs). Various studies report on LTNPs and VCs, but the prevalence estimates vary among studies because of methodological differences in definition and population sampling. In a US HIV natural history study cohort, LTNPs comprise 1.52%-4.01% of all members in the cohort and long-term VCs account for 2.83%-3.91%[2]. Long-term ECs are fewer than 1% of all patients with HIV[3]. To date, there are no epidemiological studies performed on these subgroups in China.
In Henan and neighbor provinces, HIV-1 infections among paid blood donation (PBD) and transfusion recipients occurred from 1992 to 1996 until the Chinese government banned paid blood donations. During this period, blood was collected from multiple paid donors, plasma was separated, and red blood cells, which were often mixed with those from other donors, were re-infused to the donors, and the blood and plasma production were used by transfusions recipients, these illegal practices resulted in the HIV-1 pandemic in Henan[4]. Former researches confirmed that the HIV-1 strains present in PBD and transfusion recipients in Henan are likely to have a single origin of B' subtype, possibly arriving from Yunnan, where the B' sequence was predominant in the early 1990s[5-6]. Facts pertaining to infection were revealed to the public in 2000[4], because of a large-scale anti-HIV-1 screening initiated from 2003 in Henan, lots of infections could be diagnosed, and the definitive counts emerged in 2005. At present, almost 20 years have been passed after the infection, still exist a few patients remain healthy, despite never receiving ART. These individuals are most likely LTNPs/VCs. So we used follow-up information and laboratory testing to identify and describe the features of patients who have controlled their disease progression immunologically or virologically for > 20 years.
All patients of this historical prospective cohort study resided in Henan and were recorded in the 'National HIV/AIDS Integrated Information Management System' (HIV/AIDS System). All identified patients with HIV/AIDS in China undergo confirmatory HIV western blot testing. These cases are then entered into the 'HIV/AIDS System'. Patient-specific data collected included demographic information; disease status (HIV or AIDS); risk behavior information (injecting drug use, experience of heterosexual or homosexual sex, plasma donation, blood transfusion/received blood products, surgical history, mother HIV-positive, spouse/sexual partner HIV positive, occupational exposure to HIV) and laboratory test results. All patients in the 'HIV/AIDS System' are followed by public health professionals, either via phone calls or face-to-face interviews, every six months. Data collected in this study includes demographic, clinical information and laboratory test results, up to December 31, 2014.
We adopted definitions of LTNP/VC from previous studies and the actual state of HIV-1 infections and patient follow-up policies in Henan to establish a study definition of LTNP/VC. All patients were: 1) Being diagnosed as HIV-1 infection before December 31, 2005; 2) Antiretroviral-naïve since the date of HIV-1 seroconversion; 3) Asymptomatic by the end of December 31, 2014; and 4) Recorded adequate CD4 and VL results between 2011 and 2014 (CD4 count test usually carry out once every half year and VL test usually once a year, some patients exhibited little desire to be followed because of their good health, so not enough results for these patients). Patients were defined as LTNPs if they had only one CD4 count result and the value was > 800 cells/μL, only two CD4 count results and both values > 600 cells/μL, only three CD4 results and all values > 500 cells/μL, or four and more than four CD4 count results with only one count < 500 cells/μL. VCs were defined as patients only having two VL results and both values < 3.3 log 10 copies/mL, or three or more results with at least three values < 3.3 log 10 copies/mL. ECs were patients who only had two VL results and both values < 1.7 log 10 copies/mL, or three or more results with at least three values < 1.7 log 10 copies/mL. LTNPs+VCs-were the cases belong to LTNPs but not VCs. LTNPs were defined regardless of VL and VCs regardless of CD4 count.
We completed specialized follow-up and sampling of all patients identified as LTNPs, VCs and ECs every 6 months from January 1, 2015 to June 1, 2016. Serologic evidence of infection was checked by Western blot (MP, Singapore). Anti-HCV was tested by ELISA (InTec, China). HIV-1 DNA quantitative detection (DNA) was completed by a fluorescence real-time quantitative PCR method using HIV-1 DNA Detection Kit (Supbio, China). The demographic variables were analyzed using descriptive statistics, continuous variables were expressed as the mean (SD) and/or interquartile ranges [IQR], and qualitative variables as the frequency (n) and percentage (%). We compared CD4 counts, VL, and HIV-1 DNA using independent-samples t-tests. The tests were two-sided, and a P-value < 0.05 indicated statistical significance.
A total of 29, 294 patients were confirmed as having acquired HIV/AIDS through contaminated blood donation or transfusion performed prior to December 31, 2005. Of these patients, 526 were ART-naïve and asymptomatic until December 31, 2014. Three hundred eighteen patients were followed perennially and demonstrated sufficient CD4 and VL results, with 92 patients fulfilling our definition of LTNP or VC. There were 70 cases of LTNPs (0.24%, 70/29, 294), 43 of VCs (0.15%, 43/29, 294), 48 of LTNPs + non-VCs (LTNPs+VCs-, 0.16%, 48/29, 294), and 8 cases of ECs (0.03%, 8/2, 9294) (Figure 1). All participants who followed and sampling provided written informed consent. This study was reviewed and approved by the Institutional Review Board of the Henan CDC.
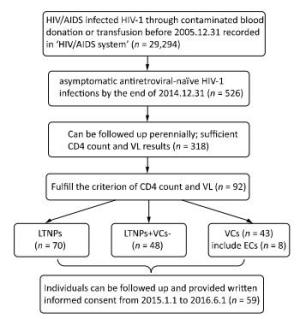
Figure 1. Selection algorithm for the patients included in the study. HIV/AIDS system, National HIV/AIDS integrated information management system; VL, virus load; LTNPs, long-term nonprogressors; VCs, viremia controllers; ECs, elite controllers; LTNPs+VCs-, the cases belong to LTNPs but not VCs.
The demographic information was summarized in Table 1, including age, sex, occupation, marital status and education level. One patient died due to reasons unrelated to AIDS, in March 2015. Three patients received antiretroviral therapy in August 2015 and were therefore excluded. Of the remaining 88 patients, 59 could be followed. Of the 59 patients, 87.9% were anti-HCV positive. The interquartile range of the CD4 count nadir was 526.5 (451.8-733.5) cells/μL, the highest VL was 3.3 (2.3-4.0) log 10 copies/mL, and the HIV-1 DNA value was 207.0 (74.3-506.5) copies/106cells. We divided these 59 patients into three groups: LTNPs, LTNPs+VCs-, and VCs. The CD4 count nadir of the VC group was 489.0 (429.3-594.5) cells/μL, significantly lower than the LTNPs (t = 2.713, P = 0.008) and the LTNPs+VCs-(t = -2.536, P = 0.014). In the VC group, the highest VL was 3.0 (2.4-3.4) log 10 copies/mL and HIV-1 DNA was 283.0 (82.0-457.0) copies/106cells; however, significant differences in the highest VL and HIV-1 DNA were not found between the groups (Table 1).
Table 1. Demographic Data of the Study Groups
Characteristic All (n = 92) LTNPs (n = 70) VCs (n = 43) LTNPs+VCs-(n = 48) Age (1994) Mean ± SD 27.7 ± 8.2 27.3 ± 8.4 28.1 ± 7.8 27.5 ± 8.8 IQR 26.4 (18.7-33.4) 25.8 (20.8-33.0) 26.0 (22.6-31.1) 27.9 (20.9-34.2) Age (2016) Mean ± SD 49.2 ± 8.3 48.9 ± 8.4 49.6 ± 7.8 49.0 ± 8.8 IQR 47.9 (40.2-54.9) 47.4 (42.4-54.5) 47.5 (44.1-52.6) 49.4 (42.4-55.7) Sex, n(%) Male 59 (63.1) 45 (64.3) 29 (67.4) 30 (62.5) Female 33 (35.9) 26 (35.7) 14 (32.6) 18 (37.5) Occupation (anti-HIV-1 confirmation), n (%) Farmer 85 (92.4) 65 (92.9) 40 (93.2) 44 (91.7) Others 7 (7.6) 6 (7.1) 3 (7.0) 4 (8.3) Marital Status (anti-HIV-1 confirmation), n (%) Unmarried 5 (5.4) 4 (5.7) 2 (4.7) 3 (6.3) Married 57 (62.0) 42 (58.6) 26 (60.5) 30 (62.5) Divorced/Widowed 30 (32.6) 25 (35.7) 15 (34.9) 15 (31.3) Education (anti-HIV-1 confirmation), n (%) Illiterate 5 (5.4) 4 (5.7) 3 (7.0) 2 (4.2) Primary 40 (43.5) 30 (42.9) 16 (37.2) 23 (47.9) Junior high school 45 (48.9) 34 (48.6) 24 (55.8) 21 (43.8) High school and above 2 (2.2) 2 (2.9) 0 (0.0) 2 (4.2) CD4 nadir cells/μLa 526.5 (451.8-733.5) 602.0 (501.0-775.0) 489.0 (429.3-594.5) 628.0 (502.5-787.5) (N = 58) (N = 43) (N = 32) (N = 25) Highest VL (log copies/mL)b 3.3 (2.3-4.0) 3.4 (2.1-4.1) 3.0 (2.4-3.4) 3.7 (2.0-4.3) (N = 59) (N = 44) (N = 32) (N = 26) Highest HIV-1 DNA (copies/106cell)c 207.0 (74.3-506.5) 158.5 (59.5-484.0) 283.0 (82.0-457.0) 158.5 (60.5-524.0) (N = 58) (N = 44) (N = 31) (N = 26) Anti-HCV positive 51 (87.9) 37 (86.0) 28 (87.5) 22 (88.0) (N = 58) (N = 43) (N = 32) (N = 25) Distribution in county, n 34 33 19 27 Note. IQR: interquartile range; LTNPs+VCs-: the cases belong to LTNPs but not VCs. aSignificant difference between LTNPs and VCs (t = 2.713, P = 0.008); Significant difference between LTNPs+VCs-and VCs (t = -2.536, P = 0.01); No significant difference between LTNPs+VCs-and LTNPs (t = -0.231, P = 0.82). bNo significant difference between them. LTNPs vs. VCs (t = 0.770, P = 0.44); LTNPs+VCs-vs. VCs (t = -1.180, P = 0.24); LTNPs vs. LTNPs+VCs-(t = -0.489, P = 0.63). cNo significant difference between them. LTNPs vs. VCs (t = 0.228, P = 0.77); LTNPs+VCs-vs. VCs (t = -0.820, P = 0.42); LTNPs vs. LTNPs+VCs-(t = -0.538, P = 0.59). Each of these 59 patients had maintained different CD4 and VL status since 2011; the three different types are exhibited in Figure 2. The cases P1, P2, and P3 were typical LTNPs and VCs. Almost every CD4 count was > 600 cells/μL and the corresponding VLs never exceeded 1, 000 copies/mL. Meanwhile, P4 and P5 exhibited excellent CD4 counts, but inferior VL. Their CD4 results were > 750 cells/μL, while the corresponding VL was approximately 4.3 log 10 copies/mL. Although patient P6 was VC, this patient's CD4 was consistently < 500 cells/μL. T1 and T2 were twins with the same infection time and route. T1 was both LTNP and VC, but T2 was only VC, and received ART in August 2015.
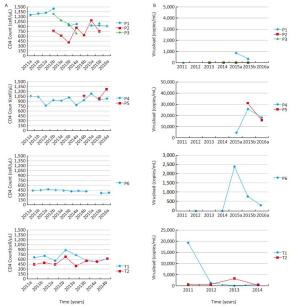
Figure 2. CD4+ T-cell count (A) and HIV-1 virus load (B) in eight individuals who possessed the representative patterns of long-term nonprogressors and viremia controllers in this study. In each case, CD4+ T-cell counts (cells/µL) and VL (copies/mL) are represented in the Y-axis (different value scale of virus load in Y axis) against time expressed as years in the X-axis. 2011a refers to the first half of 2011 and 2011b refers to the second half of 2011, and so on. A virus load value of 0 corresponds to a value less than assay sensitivity.
Previously the definition of LTNP/VC varied among studies[7]. In this study, the definition of LTNP/VC was established according to definitions used previously, the actual state of HIV-1 infections, and follow-up policies particular to Henan. While identifying LTNPs/VCs, we excluded many quasi cases that did not fit our study definition, despite having complete laboratory test results that were close to the criterion required for LTNP/VC. Meanwhile, in this LTNPs/VCs cohort, CD4 counts, VL, and HIV-1 DNA measures were independent and varied without fixed patterns. P4 and P5 simultaneously had higher CD4 counts, VL, and HIV-1 DNA values at all times, whereas P6 always had lower CD4, VL, and HIV-1 DNA values. The other cases maintained intermediate values. One study also found heterogeneous VLs in patients with > 20 years as LTNPs[8]. Thus, we determined that no precise standard exists among these cases. Continuous follow-up is necessary given the individualized and seemingly unpredictable nature of this disease in LTNPs/VCs. Past studies identified a LTNPs/VCs cohort without CD4 count, VL, and HIV-1 DNA results and patient-specific factors appear to exert a sizable effect on disease outcomes in this population. HIV-1 infection in humans is a dynamic process and may not be adequately characterized by strict standards and definitions. Therefore, clear and accurate definitions, with a measure of flexibility, may be more beneficial for disease progression research efforts.
Past studies have often overestimated the prevalence of LTNPs/VCs because of uncertain seroconversion dates[8]. All patients included in this study had to be actively followed; however, many LTNPs/VCs were asymptomatic, did not present for regular medical visits, and exhibited little desire to be followed. Consequently, these patients were excluded; therefore, we suspect that our study underestimated the prevalence of LTNPs/VCs.
The HIV-1 infection source of this LTNPs/VCs cohort was monophyletic B, 'subtype, so it is clear that the LTNPs/VCs did not originate from viral factors, at least not the patients included in this study. Many studies maintain that the viral factor is not necessarily important for forming different clinical outcomes. For example, one study had isolated replication-competent strains from VCs, with subsequent genotypic and phenotypic analyses overwhelming suggesting that these isolates were all toxic[9]. Additional evidence suggests that persistent replication and genetic evolution occurs in VCs[10]. Moreover, a case of a 10-year VC patient, whose HIV-1 was transmitted from another patient who exhibited normal progression, was also reported[11]. Generally speaking, these studies suggest that viral suppression cannot be simply interpreted as infection with a defective or attenuated strain, even if viral differences between normal progressors and LTNPs/VCs are evident[11]. The findings above were based on the study of a small number of cases with different clinical histories. In this study, the LTNPs/VCs cohort all had monophyletic B' subtype HIV-1, and additional cases further consolidate this opinion.
One theory is that loss of viral control and CD4 depletion occur independently[3]. In this study, CD4 was significantly lower in the VCs group than in the LTNPs or the LTNPs+VCs-. The differences between the highest VL and HIV-1 DNA were not significant. The mechanism of viremia control was unclear, but it appears that the VCs exhibited different control mechanisms[12]. VL was not stable and many VCs experienced transient increases in VL called 'blips'[3]. Although it is easy to understand the actions of the two indicators showed in this study, it seems that CD4 count was more helpful for identifying these special cases.
Retroviruses require chromosomal integration for successful lifecycle completion. After long-term natural selection, the integrated virus gradually becomes part of the human genome in a process called 'endogenization'. These account for approximately 8% of our genome[13]. In a study focused on two ECs, endogenization was believed to be the cause of viremia control and long-term non-progression. The researchers even suggested that without therapeutic and prophylactic strategies, after several decades of HIV-1/host interactions and millions of deaths, it is likely that a few individuals will have endogenized and neutralized the virus, transmitting it to their progeny[14]. In view of this LTNPs/VCs cohort, we also determined that these cases demonstrate a balance between HIV-1 and host, achieved after years of interaction. The state of symbiosis was individualized, so the physical descriptions of these cases were different; in addition, the prognosis of P1-P6 was excellent. Our findings seem to confirm that there is no rigorous standard that can clearly define LTNP/VC.
It is worth mentioning that, although the twin brothers had identical genetic backgrounds and monophyletic infection sources, their CD4 counts and VL changes were significantly different. By definition, they belong to the LTNPs+VCs+ and LTNPs-VCs+ groups, respectively. It appears that the effects of the host's genetic background and viral factors are limited as the disease progress, and that other undetected factors may play a more important role. This knowledge was also alluded to in another study[15].
The LTNPs/VCs being assessed here are a distinct cohort, data from this cohort provided valuable information. All members of this cohort had the same route of transmission (blood-borne transmission), monophyletic B' subtype source of infection and survived for more than 20 years without ART. This cohort increases our understanding of the likelihood of survival without ART for some patients with HIV-1. Moreover, the non-replicability of this cohort makes it even more important. Further research will better reveal the effects of HIV-1 infection in human beings and may provide more informations for HIV/AIDS prevention and control.
Authors' Contributions JL wrote the paper. JL and ZW conceived and designed the study. JL, PF, NL, DS, and QZ performed data collection and statistical analysis. JL, XX, JY, and GS performed the experiments. JL, XX, CL, and ST carried out staff training and quality management. JL and XX conducted research at the study sites. All authors read and approved the final manuscript.
doi: 10.3967/bes2017.121
Characteristics of Long-term Nonprogressors and Viremia Controllers Infected with HIV-1 via Contaminated Blood Donations or Transfusions Conducted 20 Years Earlier
-
Abstract: To characterize long-term nonprogressors (LTNPs) and viremia controllers (VCs), infected with HIV-1 through contaminated blood donation or transfusion between 1992 and 1996 in Henan, China. LTNPs and VCs were defined by CD4+T lymphocyte (CD4) count and viral load (VL). Of 29, 294 patients infected with HIV-1 via contaminated blood donation or transfusion that had conducted for more than 20 years, 92 were LTNPs/VCs. There were 70 LTNPs (0.24%), 43 VCs (0.15%), and 48 LTNPs+VCs-(0.16%). VCs had a significantly lower CD4 nadir, compared to LTNPs and LTNPs+VCs-, and no significant differences for the highest VL and HIV-1 DNA. Cases P4 and P5 were LTNPs, while their VL reached approximately 4.3 log copies/mL. P6 was a VC, but with CD4 < 500 cells/μL constantly. Data from the LTNPs/VCs cohort provided valuable information, future research is needed.
-
Figure 2. CD4+ T-cell count (A) and HIV-1 virus load (B) in eight individuals who possessed the representative patterns of long-term nonprogressors and viremia controllers in this study. In each case, CD4+ T-cell counts (cells/µL) and VL (copies/mL) are represented in the Y-axis (different value scale of virus load in Y axis) against time expressed as years in the X-axis. 2011a refers to the first half of 2011 and 2011b refers to the second half of 2011, and so on. A virus load value of 0 corresponds to a value less than assay sensitivity.
Table 1. Demographic Data of the Study Groups
Characteristic All (n = 92) LTNPs (n = 70) VCs (n = 43) LTNPs+VCs-(n = 48) Age (1994) Mean ± SD 27.7 ± 8.2 27.3 ± 8.4 28.1 ± 7.8 27.5 ± 8.8 IQR 26.4 (18.7-33.4) 25.8 (20.8-33.0) 26.0 (22.6-31.1) 27.9 (20.9-34.2) Age (2016) Mean ± SD 49.2 ± 8.3 48.9 ± 8.4 49.6 ± 7.8 49.0 ± 8.8 IQR 47.9 (40.2-54.9) 47.4 (42.4-54.5) 47.5 (44.1-52.6) 49.4 (42.4-55.7) Sex, n(%) Male 59 (63.1) 45 (64.3) 29 (67.4) 30 (62.5) Female 33 (35.9) 26 (35.7) 14 (32.6) 18 (37.5) Occupation (anti-HIV-1 confirmation), n (%) Farmer 85 (92.4) 65 (92.9) 40 (93.2) 44 (91.7) Others 7 (7.6) 6 (7.1) 3 (7.0) 4 (8.3) Marital Status (anti-HIV-1 confirmation), n (%) Unmarried 5 (5.4) 4 (5.7) 2 (4.7) 3 (6.3) Married 57 (62.0) 42 (58.6) 26 (60.5) 30 (62.5) Divorced/Widowed 30 (32.6) 25 (35.7) 15 (34.9) 15 (31.3) Education (anti-HIV-1 confirmation), n (%) Illiterate 5 (5.4) 4 (5.7) 3 (7.0) 2 (4.2) Primary 40 (43.5) 30 (42.9) 16 (37.2) 23 (47.9) Junior high school 45 (48.9) 34 (48.6) 24 (55.8) 21 (43.8) High school and above 2 (2.2) 2 (2.9) 0 (0.0) 2 (4.2) CD4 nadir cells/μLa 526.5 (451.8-733.5) 602.0 (501.0-775.0) 489.0 (429.3-594.5) 628.0 (502.5-787.5) (N = 58) (N = 43) (N = 32) (N = 25) Highest VL (log copies/mL)b 3.3 (2.3-4.0) 3.4 (2.1-4.1) 3.0 (2.4-3.4) 3.7 (2.0-4.3) (N = 59) (N = 44) (N = 32) (N = 26) Highest HIV-1 DNA (copies/106cell)c 207.0 (74.3-506.5) 158.5 (59.5-484.0) 283.0 (82.0-457.0) 158.5 (60.5-524.0) (N = 58) (N = 44) (N = 31) (N = 26) Anti-HCV positive 51 (87.9) 37 (86.0) 28 (87.5) 22 (88.0) (N = 58) (N = 43) (N = 32) (N = 25) Distribution in county, n 34 33 19 27 Note. IQR: interquartile range; LTNPs+VCs-: the cases belong to LTNPs but not VCs. aSignificant difference between LTNPs and VCs (t = 2.713, P = 0.008); Significant difference between LTNPs+VCs-and VCs (t = -2.536, P = 0.01); No significant difference between LTNPs+VCs-and LTNPs (t = -0.231, P = 0.82). bNo significant difference between them. LTNPs vs. VCs (t = 0.770, P = 0.44); LTNPs+VCs-vs. VCs (t = -1.180, P = 0.24); LTNPs vs. LTNPs+VCs-(t = -0.489, P = 0.63). cNo significant difference between them. LTNPs vs. VCs (t = 0.228, P = 0.77); LTNPs+VCs-vs. VCs (t = -0.820, P = 0.42); LTNPs vs. LTNPs+VCs-(t = -0.538, P = 0.59). -
[1] Mikhail M, Wang B, Saksena NK. Mechanisms involved in non-progressive HIV disease. AIDS Rev, 2003; 5, 230-44. [2] Okulicz JF, Marconi VC, Landrum ML, et al. Clinical outcomes of elite controllers, viremic controllers, and long-term nonprogressors in the US Department of Defense HIV natural history study. J Infect Dis, 2009; 200, 1714-23. doi: 10.1086/599183 [3] Okulicz JF, Lambotte O. Epidemiology and clinical characteristics of elite controllers. Curr Opin HIV AIDS, 2011; 6, 163-8. doi: 10.1097/COH.0b013e328344f35e [4] Kaufman J, Jing J. China and AIDS--the time to act is now. Science, 2002; 296, 2339-40. doi: 10.1126/science.1074479 [5] Zhang L, Chen Z, Cao Y, et al. Molecular characterization of human immunodeficiency virus type 1 and hepatitis C virus in paid blood donors and injection drug users in China. J Virol, 2004; 78, 13591-9. doi: 10.1128/JVI.78.24.13591-13599.2004 [6] Li Z, He X, Wang Z, et al. Tracing the origin and history of HIV-1 subtype B' epidemic by near full-length genome analyses. AIDS, 2012; 26, 877-84. doi: 10.1097/QAD.0b013e328351430d [7] Gurdasani D, Iles L, Dillon DG, et al. A systematic review of definitions of extreme phenotypes of HIV control and progression. AIDS, 2014; 28, 149-62. doi: 10.1097/QAD.0000000000000049 [8] van der Helm JJ, Geskus R, Lodi S, et al. Characterisation of long-term non-progression of HIV-1 infection after seroconversion: a cohort study. Lancet HIV, 2014; 1, e41-8. doi: 10.1016/S2352-3018(14)70016-5 [9] Blankson JN, Bailey JR, Thayil S, et al. Isolation and characterization of replication-competent human immunodeficiency virus type 1 from a subset of elite suppressors. J Virol, 2007; 81, 2508-18. doi: 10.1128/JVI.02165-06 [10] O'Connell KA, Brennan TP, Bailey JR, et al. Control of HIV-1 in elite suppressors despite ongoing replication and evolution in plasma virus. J Virol, 2010; 84, 7018-28. doi: 10.1128/JVI.00548-10 [11] Bailey JR, O'Connell K, Yang HC, et al. Transmission of human immunodeficiency virus type 1 from a patient who developed AIDS to an elite suppressor. J Virol, 2008; 82, 7395-410. doi: 10.1128/JVI.00800-08 [12] Cockerham LR, Hatano H. Elite control of HIV: is this the right model for a functional cure? Trends Microbiol, 2015; 23, 71-5. doi: 10.1016/j.tim.2014.11.003 [13] Feschotte C, Gilbert C. Endogenous viruses: insights into viral evolution and impact on host biology. Nat Rev Genet, 2012; 13, 283-96. https://hal.archives-ouvertes.fr/hal-00679842/document [14] Colson P, Ravaux I, Tamalet C, et al. HIV infection en route to endogenization: two cases. Clin Microbiol Infect, 2014; 20, 1280-8. doi: 10.1111/1469-0691.12807 [15] Bartha I, McLaren PJ, Brumme C, et al. Estimating the Respective Contributions of Human and Viral Genetic Variation to HIV Control. PLoS Comput Biol, 2017; 13, e1005339. doi: 10.1371/journal.pcbi.1005339 -




 下载:
下载:



 Quick Links
Quick Links