-
Aluminum (Al) is the most abundant metal in the Earth’s crust and is widely used in everyday life and industry. Al is neurotoxic to mammals. Epidemiological studies have confirmed the positive relationship between an increased environmental Al concentration and cognitive impairment[1]. Hippocampal long-term potentiation (LTP) has been widely used as a neuronal model of synaptic plasticity in experiments designed to elucidate the synaptic mechanisms involved in the loss of learning and memory[2]. The alpha-amino-3-hydroxy-5-methylisoxazole-4-propionic acid (AMPA)-type glutamate receptor (AMPAR), a major glutamate-gated ion channel in the mammalian central nervous system, plays important roles in synaptic transmission, synaptic plasticity such as LTP, and learning and memory cognitive function of the brain. LTP typically results from an increased concentration of AMPARs at the postsynaptic membrane surface[3].
A reversible post-translational modification by the addition of a long-chain fatty acid to a cysteine residue of the AMPAR, called S-palmitoylation, is critical for the modulation of AMPAR function, which dynamically regulates protein stability, transport, and activity and is the key element of LTP[4]. This process is a distinctive, reversible lipid modification and potentially regulates the function of AMPAR via cycles of palmitoylation and depalmitoylation, which are catalyzed by protein palmitoyltransferases and thioesterases, respectively[5]. However, the role of AMPA palmitoylation in the impairment of LTP induced by Al exposure is still poorly understood.
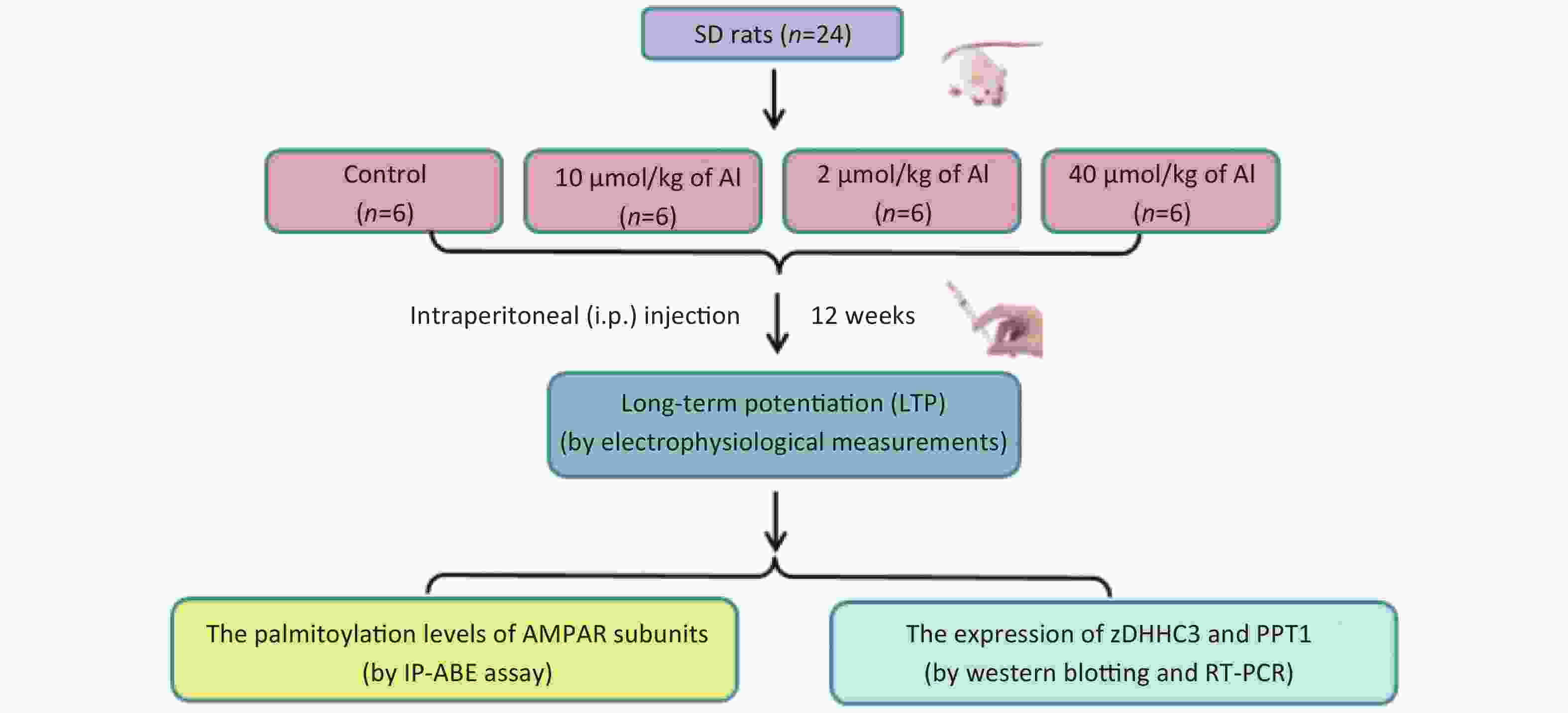
Here, we aimed to investigate the possible link between AMPA palmitoylation and the impairment of LTP induced by Al in vivo and to clarify the underlying mechanisms. First, to explore the involvement of palmitoylation in Al-induced LTP suppression, we administered different doses of Al to rats and determined the palmitoylation levels of glutamate receptor 1 (GluR1) and GluR2 after LTP measurement. Second, the expression of palmitoyltransferase (zDHHC3) and palmitoyl protein thioesterase-1 (PPT1) in the hippocampi of rats were tested at the same time to confirm the changes in palmitoylation and depalmitoylation.
In accordance with the methods of Al(mal)3 preparation described in our previous publication[5], AlCl3·6H2O (Sigma-Aldrich, St. Louis, MO, USA) was dissolved in distilled water to concentrations of 20, 40, and 80 mmol/L. Maltolate was dissolved in phosphate-buffered saline to concentrations of 60, 120, and 240 mmol/L. Al(mal)3 solution is the mixed solution of AlCl3·6H2O and maltol solutions. Al(mal)3 was freshly prepared for each experiment by mixing the solutions in equal volumes, adjusting the pH to 7.4 with NaOH, and filtering the solutions through 0.22 µm syringe filters. The total Al concentration after filtration were 10, 20, and 40 mmol/L.
The experiments were performed on 2-month-old male Sprague-Dawley rats (SPF grade, the Laboratory Animal Center, Shanxi Medical University, Taiyuan, China) kept under constant temperature and humidity conditions with free access to food and water. The rats (weight 180–200 g) were randomly divided into four groups (n = 6 per group), namely, control, low-dose, medium-dose, and a high-dose group, in the subchronic Al treatment procedure and received saline (control group) or Al(mal)3 (10, 20, and 40 µmol/(kg·d) bw via an intraperitoneal injection every two days for 12 weeks. After electrophysiological measurements were performed, the rats were decapitated, and the hippocampi were rapidly removed for biochemical analyses and stored at −80 °C until use. The present study was approved by the Ethics Committee for Animal Studies of Shanxi Medical University. All efforts were made to minimize the number of animals used and their suffering. SPSS 22.0 software (SPSS Co. Ltd., Chicago, IL, USA) was used to analyze all results. Experimental data are expressed as the means ± standard deviation. Differences among groups were analyzed using one-way analysis of variance, followed by the least significance difference test for multiple comparisons when F was significant. Correlation analysis was used to analyze the correlation between the amplitude of the standardized field excitatory postsynaptic potential (fEPSP) and the palmitoylation of GluR1 and GluR2 in the hippocampus and the correlation between GluR1 and GluR2 palmitoylation and zDHHC3 and PPT1 protein expression. P-values < 0.05 were considered significant.
Except for the 40 µmol/kg Al(mal)3 group, the remaining rats developed normally and were lively and active over the study period. The rats in the 40 µmol/kg Al(mal)3 group showed poor response and had dull fur. No spontaneous animal mortality was observed during the course of the study. As shown in Supplementary Table S1 (available in www.besjournal.com), Al exposure resulted in reduced net increase of body weight (F = 25.522, P < 0.05). Decreases were observed in brain indices compared with the control group at the end of Al exposure, but the difference was not statistically significant (F = 3.521, P = 0.085).
Table S1. Net increase in body weight and brain indices of rats after Al(mal)3 treatment for 12 weeks
Items Groups Control 10 μmol 20 μmol 40 μmol Net increase of body weight (g) 198 ± 16 190 ± 13 191 ± 14 177 ± 11a Brain indices (g/1,000g) 6.19 ± 0.23 6.11± 0.16 6.13 ± 0.12 5.95 ± 0.26 Note. Values are represented as mean ± SEM (n = 6). aP < 0.05 compared with the control group. The LTP in the hippocampal CA1 region was measured by electrophysiological measurements in vivo. The surgical procedure and electrophysiological recordings were performed in accordance with the methods described in our previous publication[5]. With the increase in Al(mal)3 concentrations, the average value of the fEPSP amplitude gradually decreased after high frequency stimulation (HFS) (Figure 1A). The fEPSP amplitudes at four time points were selected for statistical analysis (Figure 1B). No significant difference was recorded in the basic fEPSP amplitude in the control and Al-exposed rats. The fEPSP amplitudes of the control group (n = 5) were (2.20 ± 0.17), (1.95 ± 0.09), and (1.82 ± 0.02) at 1, 30, and 60 min after HFS, respectively. The fEPSP amplitudes of the 10 µmol/kg Al(mal)3 group (n = 5) were (1.99 ± 0.07) at 1 min, (1.70 ± 0.09) at 30 min, and (1.57 ± 0.10) at 60 min after HFS, presenting a slight but significant decrease compared with the control group (P < 0.05). These values dropped to (1.76 ± 0.05), (1.41 ± 0.12), and (1.35 ± 0.11) in the 20 µmol/kg Al(mal)3 group (n = 5, P < 0.05) and further decreased to (1.50 ± 0.10), (1.22 ± 0.11), and (1.05 ± 0.04) in the 40 µmol/kg Al(mal)3 group (n = 5, P < 0.05). The difference in LTP was evident between the Al(mal)3 exposure groups and control group, and further suppression became notable with the increase Al(mal)3 concentrations. These findings were consistent with those of a previous report[5]. The findings indicate that the animal model was established successfully and was suitable to study the mechanism of LTP induced by Al in detail.

Figure 1. Effects of subchronic Al(mal)3 exposure on LTP in rats. (A) Scatter plots of the standardized fEPSP amplitude in different dose groups. (B) Statistical analysis of the standardized fEPSP amplitude at four time points (basic, 1 min, 30 min, and 60 min). Each point represents the mean ± SD; n = 5; a: P < 0.05 vs. the control group, b: P < 0.05 vs. 10 μmol/kg group, c: P < 0.05 vs. 20 μmol/kg group.
The palmitoylation levels of AMPAR subunits (GluR1 and GluR2) in the hippocampus were detected by immunoprecipitation and acyl-biotin exchange assay, which was performed in accordance with the methods described in previous publication[6]. Two dosages (20 and 40 µmol/kg of Al) of Al(mal)3 induced significant changes in the palmitoylation levels of GluR1 (F = 3.3348, P < 0.05) and GluR2 (F = 23.07, P < 0.05) compared with those in the control group (Figure 2). Significantly, the level of palmitoylation of GluR2 in 40 µmol/kg Al(mal)3 rats decreased compared with that in control group and 10 and 20 µmol/kg Al(mal)3 groups (P < 0.05). As shown in Supplementary Figures S1A and S1B (available in www.besjournal.com), the correlation analysis between the standardized fEPSP amplitudes and the palmitoylation levels of GluR1 and GluR2 in the hippocampus showed that the standardized fEPSP amplitude at 60 min after HFS in the rat hippocampus positively correlated with the palmitoylation levels of GluR1 (r = 0.660, P < 0.05) and GluR2 (r = 0.895, P < 0.05). The results showed that with the increase in Al concentrations, the palmitoylation levels of GluR1 and GluR2 gradually decreased. We found a positive correlation between LTP and the palmitoylation of GluR1 and GluR2. Therefore, we hypothesized that LTP damage may be related to the decrease in the palmitoylation level of AMPAR. However, the mechanisms of this effect are unclear.
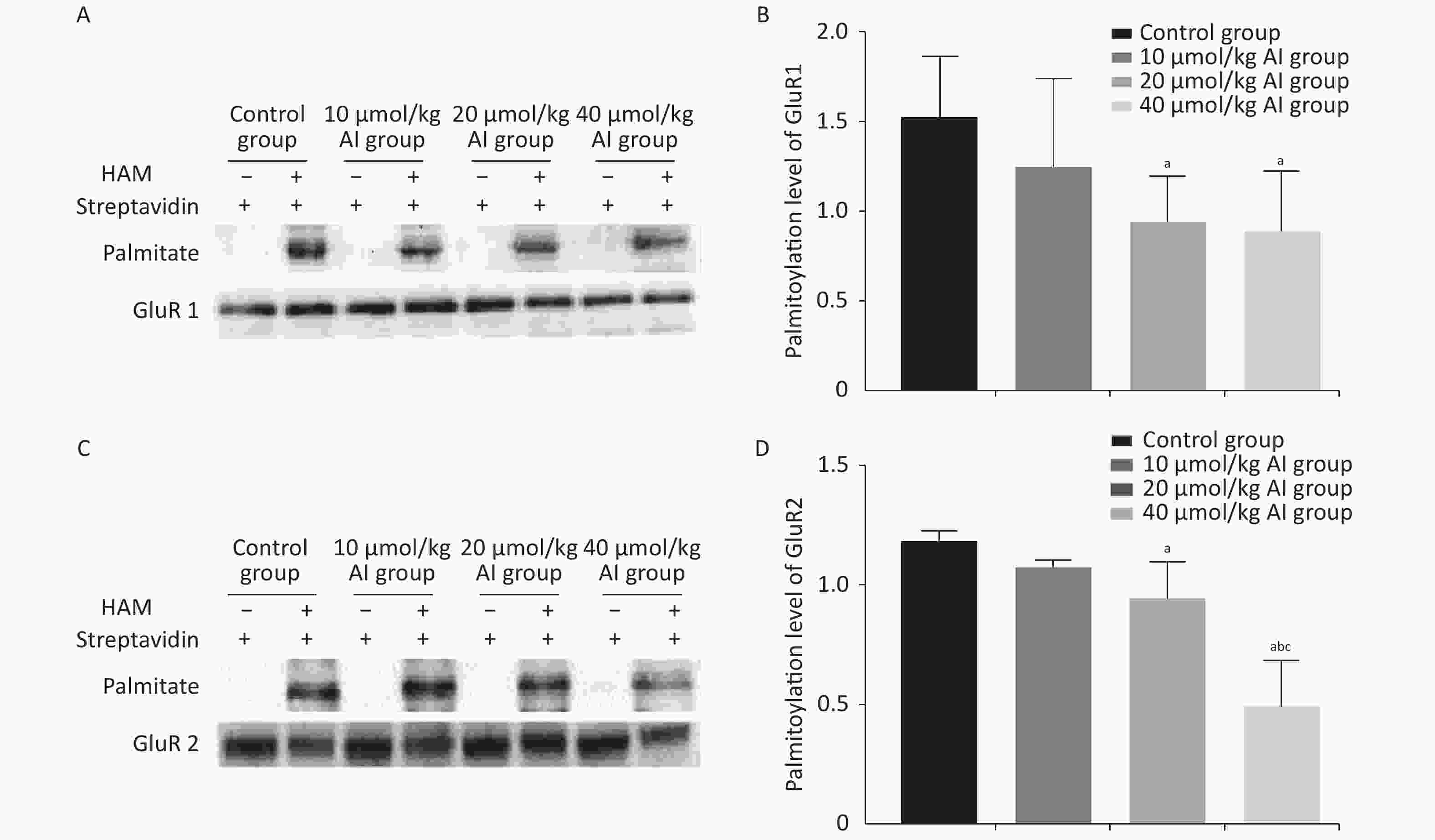
Figure 2. Effects of subchronic Al(mal)3 on the palmitoylation of AMPAR Subunits (GluR1 and GluR2) in the rat hippocampus in vivo. The level of palmitoylation of each protein was quantified as the ratio obtained by normalizing the band in the pulldown blot representing the palmitoylated protein to the corresponding input band representing the total protein for each condition. (A, B): Palmitoylation level of GluR1 in the hippocampus. (C, D): Palmitoylation level of GluR2 in the hippocampus. Values are presented as the mean ± SD (n = 5); a: P < 0.05 vs. the control group, b: P < 0.05 vs. 10 μmol/kg group, c: P < 0.05 vs. 20 μmol/kg group.
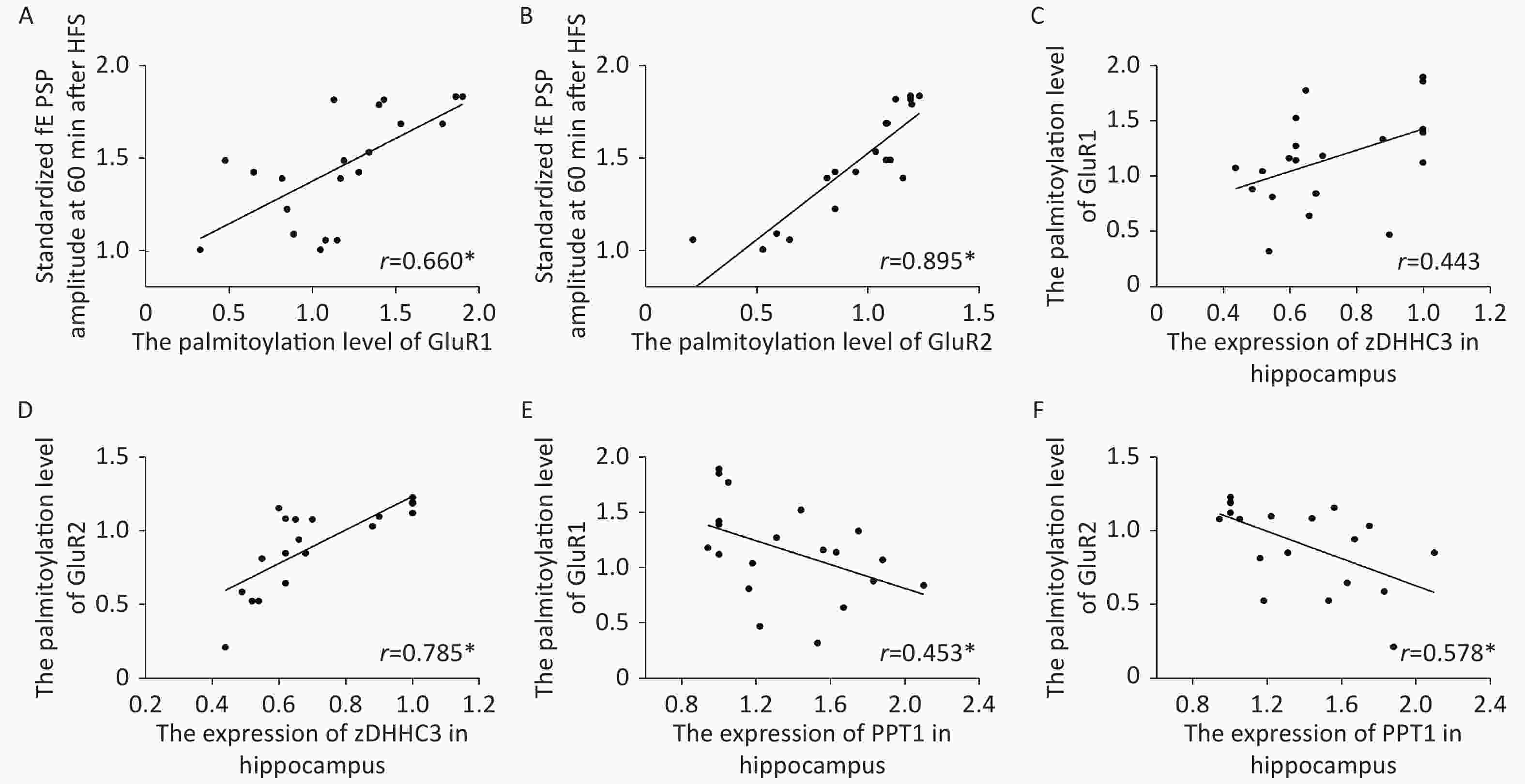
Figure S1. Scatter plots of the standardized fEPSP amplitude, the palmitoylation levels of AMPA receptors and the zDHHC3 and PPT1 protein expression in hippocampi exposed to Al(mal)3, (A) There was a positive correlation between the standardized fEPSP amplitude and the palmitoylation level of GluR1 (r = 0.660, *: P < 0.01). (B) There was a positive correlation between the standardized fEPSP amplitude and the palmitoylation level of GluR2 (r = 0.895, *: P < 0.01). (C, D) There was a positive correlation between the expression level of zDHHC3 and the palmitoylation levels of GluR1 (r = 0.785) and GluR2 (r = 0.785, *: P < 0.01). (E, F): There was a negative correlation between the expression level of PPT1 and the palmitoylation levels of GluR1 (r = −0.453, *: P < 0.01) and GluR2 (r = −0.578, *: P < 0.01).
The protein and gene concentrations of zDHHC3 and PPT1 in the hippocampus were measured by Western blotting and reverse transcription polymerase chain reaction, respectively. We observed that with the increase in the Al concentration, the expression of zDHHC3 gradually decreased, whereas the expression of PPT1 gradually increased. The results regarding the gene and protein are consistent. As shown in Figure 3A, the increase in Al(mal)3 concentration decreased the relative intensities of zDHHC3 (F = 21.34, P < 0.05) in a dose-dependent manner compared with that in the control group. The relative intensity of zDHHC3 in the 40 mmol/kg Al(mal)3 group was significantly lower than those in the control and 10 mmol/kg Al(mal)3 groups. With the increase in Al(mal)3 concentrations, the gene levels of zDHHC3 gradually decreased (F = 17.00, P < 0.05) compared with that in the control group (Figure 3B). In the 40 µmol/kg Al(mal)3 group, the gene level of zDHHC3 substantially decreased compared with those in the control and 10 µmol/kg Al(mal)3 groups (P < 0.05). Increasing the Al(mal)3 concentration increased the protein expression of PPT1 compared with that in the control group (F = 5.10, P < 0.05) (Figure 3C). As shown in Figure 3D, Al-induced dose-dependent increases of PPT1 gene expression also occurred (F = 75.60, P < 0.05), and PPT1 gene expression in the 20 and 40 µmol/kg Al(mal)3 groups remarkably increased compared with that in the 10 µmol/kg Al(mal)3 group (P < 0.05). Thus, Al may influence the expression of zDHHC3 and PPT1 in the rat hippocampus and further influence the palmitoylation of AMPAR. Correlation analysis (Supplementary Figure S2C–S2F available in www.besjournal.com) showed that the expression level of zDHHC3 was positively correlated with the palmitoylation levels of GluR1 and GluR2, and that the expression level of PPT1 was negatively correlated with the palmitoylation of GluR1 and GluR2, which supports this view.
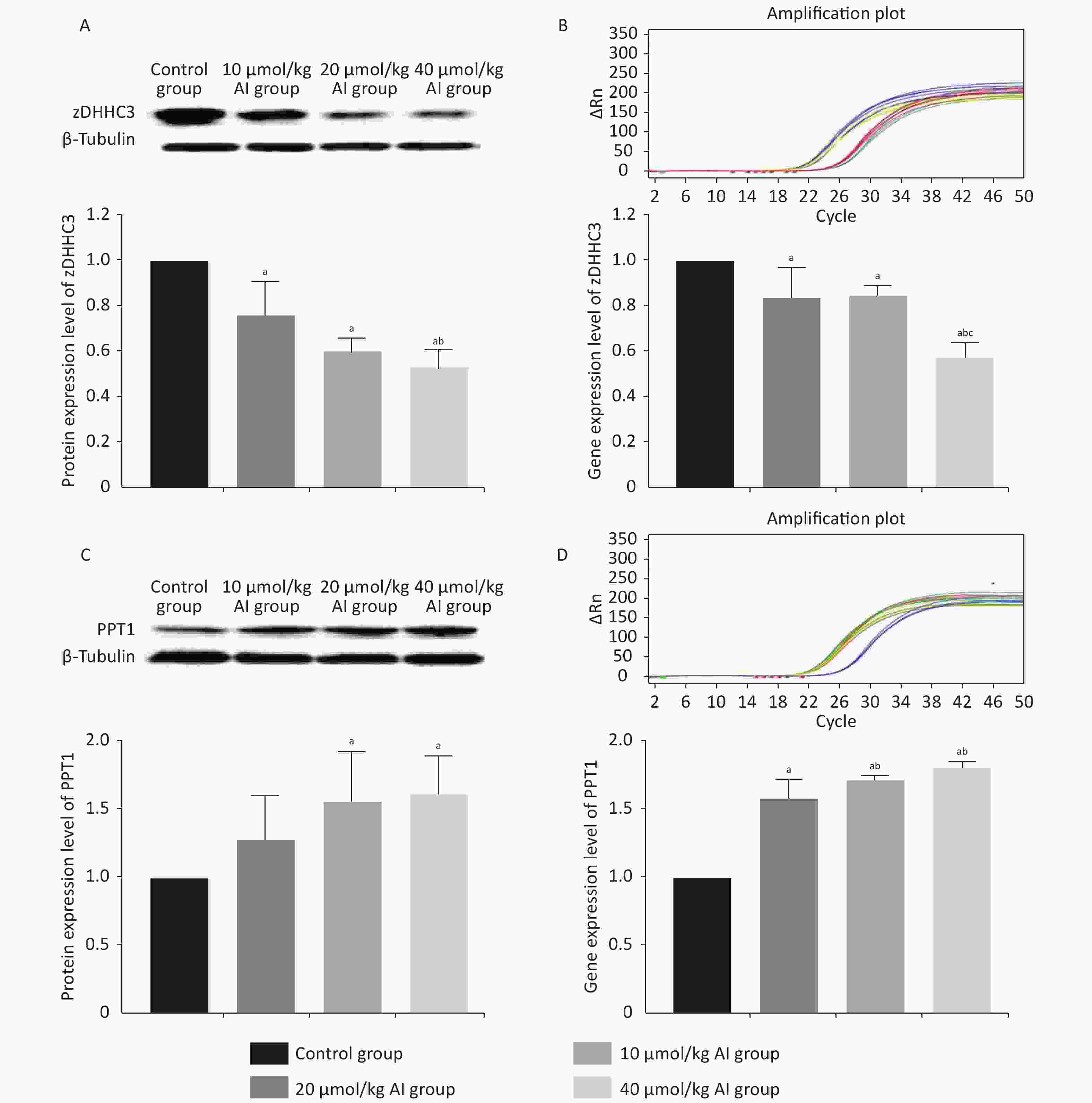
Figure 3. Expression of zDHHC3 and PPT1 in the hippocampus of rats subchronically exposed to Al(mal)3. (A) Protein expression of zDHHC3. (B) Gene expression of zDHHC3. (C) Protein expression of PPT1. (D) Gene expression of PPT1. Values are presented as the mean ± SD (n = 5); a: P < 0.05 vs. the control group, b: P < 0.05 vs. 10 μmol/kg group, c: P < 0.05 vs. 20 μmol/kg group.
The mechanism responsible for the effects of Al exposure on zDHHC3 and PPT1 is not fully elucidated. One possible aspect of this mechanism regarding zDHHC3 is the high affinity of Al for the metal binding sites in enzymes due to its small size and near maximal charge; this affinity causes Al to disrupt critical enzymatic reactions. The zDHHC3 catalytic motif is located within a cysteine-rich, zinc finger-like domain which relies on Zn2+ normal function[7]. Al3+ can compete with and substitute for essential metals in enzyme reactions because these reactions normally rely on metals having a rapid reversible dissociation; meanwhile, Al has very slow release rate from the ligands to which it binds. Al3+ dissociates from biological ligands 107 more slowly than Zn2+ [8]. Al3+ replaces Zn2+ in several Zn2+-containing enzyme proteins[9]. We speculate that Al3+ may inhibit the activity of zDHHC3 by competitively replacing Zn2+ in the zinc finger structure, thereby affecting the enzymatic reaction of palmitoylation modification. In addition, PPT1 is palmitoylated in vivo and is a substrate for two palmitoylating enzymes, zDHHC3 and DHHC7[10]. Therefore, we hypothesized that Al may enhance the activity of PPT1 by affecting zDHHC3.
In summary, we provide strong evidence that the decrease in AMPAR palmitoylation is involved in the impairment of hippocampal LTP. Additionally, zDHHC3 and PPT1 may be the Al targets that influence the AMPAR cycles of palmitoylation and depalmitoylation, respectively. This conclusion raises the possibility that the palmitoylation of AMPARs may be a new molecular event contributing to preventing the influence of Al on learning and memory to a certain degree and may lead to the exploration of valid targets to treat diseases correlated to Al.
Acknowledgments We sincerely thank our colleagues for their help and work on the research.
Conflicts of interest The authors declare no conflicts of interest.
doi: 10.3967/bes2020.133
Palmitoylation of AMPA Receptors is Involved in the Inhibition of Hippocampal Long-term Potentiation by Aluminum Exposure in Rats
-
-
Figure 1. Effects of subchronic Al(mal)3 exposure on LTP in rats. (A) Scatter plots of the standardized fEPSP amplitude in different dose groups. (B) Statistical analysis of the standardized fEPSP amplitude at four time points (basic, 1 min, 30 min, and 60 min). Each point represents the mean ± SD; n = 5; a: P < 0.05 vs. the control group, b: P < 0.05 vs. 10 μmol/kg group, c: P < 0.05 vs. 20 μmol/kg group.
Figure 2. Effects of subchronic Al(mal)3 on the palmitoylation of AMPAR Subunits (GluR1 and GluR2) in the rat hippocampus in vivo. The level of palmitoylation of each protein was quantified as the ratio obtained by normalizing the band in the pulldown blot representing the palmitoylated protein to the corresponding input band representing the total protein for each condition. (A, B): Palmitoylation level of GluR1 in the hippocampus. (C, D): Palmitoylation level of GluR2 in the hippocampus. Values are presented as the mean ± SD (n = 5); a: P < 0.05 vs. the control group, b: P < 0.05 vs. 10 μmol/kg group, c: P < 0.05 vs. 20 μmol/kg group.
S1. Scatter plots of the standardized fEPSP amplitude, the palmitoylation levels of AMPA receptors and the zDHHC3 and PPT1 protein expression in hippocampi exposed to Al(mal)3, (A) There was a positive correlation between the standardized fEPSP amplitude and the palmitoylation level of GluR1 (r = 0.660, *: P < 0.01). (B) There was a positive correlation between the standardized fEPSP amplitude and the palmitoylation level of GluR2 (r = 0.895, *: P < 0.01). (C, D) There was a positive correlation between the expression level of zDHHC3 and the palmitoylation levels of GluR1 (r = 0.785) and GluR2 (r = 0.785, *: P < 0.01). (E, F): There was a negative correlation between the expression level of PPT1 and the palmitoylation levels of GluR1 (r = −0.453, *: P < 0.01) and GluR2 (r = −0.578, *: P < 0.01).
Figure 3. Expression of zDHHC3 and PPT1 in the hippocampus of rats subchronically exposed to Al(mal)3. (A) Protein expression of zDHHC3. (B) Gene expression of zDHHC3. (C) Protein expression of PPT1. (D) Gene expression of PPT1. Values are presented as the mean ± SD (n = 5); a: P < 0.05 vs. the control group, b: P < 0.05 vs. 10 μmol/kg group, c: P < 0.05 vs. 20 μmol/kg group.
S1. Net increase in body weight and brain indices of rats after Al(mal)3 treatment for 12 weeks
Items Groups Control 10 μmol 20 μmol 40 μmol Net increase of body weight (g) 198 ± 16 190 ± 13 191 ± 14 177 ± 11a Brain indices (g/1,000g) 6.19 ± 0.23 6.11± 0.16 6.13 ± 0.12 5.95 ± 0.26 Note. Values are represented as mean ± SEM (n = 6). aP < 0.05 compared with the control group. -
[1] Frisardi V, Solfrizzi V, Capurso C, et al. Aluminum in the diet and Alzheimer‘s disease: from current epidemiology to possible disease-modifying treatment. J Alzheimer's Dis, 2010; 20, 17−30. doi: 10.3233/JAD-2010-1340 [2] Zhang LF, Jin CH, Liu QF, et al. Effects of subchronic aluminum exposure on spatial memory, ultrastructure and L-LTP of hippocampus in rats. J Toxicol Sci, 2013; 38, 255−68. doi: 10.2131/jts.38.255 [3] Chen ZC, Stockwell J, Cayabyab FS. Adenosine A1 receptor-mediated endocytosis of AMPA receptors contributes to impairments in long-term potentiation (LTP) in the middle-aged rat hippocampus. Neurochem Res, 2016; 41, 1085−97. doi: 10.1007/s11064-015-1799-3 [4] Han J, Wu PF, Wang F, et al. S-palmitoylation regulates AMPA receptors trafficking and function: a novel insight into synaptic regulation and therapeutics. Acta Pharm Sin B, 2015; 5, 1−7. doi: 10.1016/j.apsb.2014.12.002 [5] Song J, Liu Y, Zhang HF, et al. Effects of exposure to aluminum on long-term potentiation and AMPA receptor subunits in rats in vivo. Biomed Environ Sci, 2014; 27, 77−84. [6] Brigidi GS, Bamji SX. Detection of protein palmitoylation in cultured hippocampal neurons by immunoprecipitation and acyl-biotin exchange (ABE). J Vis Exp, 2013; 18, 50031. [7] Hanas JS, Gunn CG. Inhibition of transcription factor ⅢA-DNA interactions by xenobiotic metal ions. Nucleic Acids Res, 1996; 24, 924−30. doi: 10.1093/nar/24.5.924 [8] Martin RB. The chemistry of aluminium related to biology and medicine. Clin Chem, 1986; 32, 1797−806. doi: 10.1093/clinchem/32.10.1797 [9] Chakraborty A, Basak S. Interaction with Al and Zn induces structure formation and aggregation in natively unfolded caseins. J Photochem Photobiol B, 2008; 93, 36−43. doi: 10.1016/j.jphotobiol.2008.06.011 [10] Segal-Salto M, Sapir T, Reiner O. Reversible cysteine acylation regulates the activity of human palmitoyl-protein Thioesterase 1 (PPT1). PLoS One, 2016; 11, e0146466. doi: 10.1371/journal.pone.0146466 -




 下载:
下载:




 Quick Links
Quick Links