-
Infertility, defined as the inability to conceive within 1 year of unprotected intercourse, affects an estimated 80 million individuals worldwide, or 10%-15% of couples of reproductive age[1]. Approximately 40%-50% of infertility cases involve male factor infertility[2]. Obesity, which has been recognized as a major risk factor for male infertility, leads to decreased sperm number and reproductive dysfunction[3]. Male reproduction regulation is a complex process that involves the testes, epididymis, accessory sex glands, and associated hormones. To date, the mechanisms underlying obesity-associated infertility are not fully understood. However, many previous studies indicated that excessive cell apoptosis plays an important role in obesity-induced male spermatogenesis dysfunction.
Apoptosis is of great importance during male germ cell development[4]. Apoptosis is essential for cellular homeostasis and the normal male germ cell development process[5, 6]. Dysregulation of germ cell apoptosis, in turn, may cause suboptimal male reproductive function and even male infertility. A high-fat diet (HFD) has been demonstrated to induce apoptosis in rodents, which in turn promotes the progression of infertility. Conversely, many studies demonstrated that inhibiting excessive apoptosis attenuates HFD‑induced impairment of spermatogenesis[7-10]. Therefore, pharmacological agents that can inhibit testicular cell apoptosis are of great therapeutic interest.
Sodium 4-phenylbutyrate (4-PBA) is a low-molecular-weight fatty acid that has been used for treatment of urea cycle disorders in children, sickle cell disease, and thalassemia. 4-PBA has been demonstrated to have protective effects on increased cell apoptosis induced by various environmental stresses, such as toxicant exposure and oxidative stress. 4-PBA can protect against myocardial and liver ischemia-reperfusion injury, alleviate lipid-induced insulin resistance and beta-cell dysfunction in humans, and ameliorate lipopolysaccharide-induced lung inflammation[11-13]. Furthermore, a previous study indicated that 4-PBA protected against Cd-induced germ cell apoptosis in testes[14]. However, the effect of 4-PBA on obesity‑induced impaired spermatogenesis has not yet been reported. The aim of the present study was to investigate whether 4-PBA can protect against HFD‑induced defective spermatogenesis.
-
All experiments in the present study were performed in compliance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals and were approved by the Animal Care and Use Committees of Xinxiang Medical University. A total of 30 adult male Sprague-Dawley rats (90-120 g, 4 weeks) were obtained from Vital River Laboratory Animal Technology Co. Ltd (Beijing, China). All rats were quarantined for 7 days before the start of the study and then randomly divided into three groups: a control group (standard diet), a HFD group, and a HFD+4-PBA (Sigma-Aldrich) group. Rats in the control group were fed with a normal diet (control, 10 kcal% fat, D12450B, Research Diets, New Brunswick, NJ, USA), whereas the other rats were fed with a HFD (45 kcal% fat, D12451, Research Diets, New Brunswick, NJ, USA) for 13 weeks to induce obesity. Rats in the HFD+4-PBA group were orally administered 100 mg/kg 4-PBA once per day for 13 weeks, and the other two groups received the same volume of vehicle as a negative control. The 4-PBA dosage of 100 mg/kg was based on previous studies[15, 16] and the results of preliminary experiments. At the end of the experiment, the rats were sacrificed under sodium pentobarbital anesthesia (60 mg/kg, ip), and their testes, epididymis, and accessory glands (seminal vesicles and prostate) were carefully dissected out and weighed.
-
On the last day of the study, blood samples were collected and immediately placed in a refrigerator for storage at 4 ℃ until centrifugation at 3, 000 rpm for 10 min to separate the serum. Serum was collected and stored at -20 ℃ until analysis. Serum levels of follicle-stimulating hormone (FSH), luteotropic hormone (LH), and testosterone (T) were measured using hormone-specific kits purchased from Beijing North Institute of Biological Technology by the radioimmunoassay (RIA) method.
-
The head of the left epididymis was placed in clean 30-mm dishes containing 4 mL pre-warmed M199 and minced with fine scissors to allow spermatozoa to swim out for 10 to 15 min at 37 ℃. After incubation, the epididymal tissue-fluid mixture was filtered to separate the supernatant from tissue particles. The sperm suspensions were evaluated for sperm head counts, viability, motility, and morphology following the procedures described below. The sperm head counts were determined with a hemocytometer. A 0.5 mL sample of the sperm suspension was diluted with 9.5 mL of physiological saline. Approximately 10 µL of diluted sperm suspension was transferred to each counting chamber and then allowed to stand for 5 min and counted under a light microscope (LEICA DM 1000) at 400× magnification[17]. The total sperm counts (×106/mL) were calculated. The determination of sperm viability and motility was carried out according to the procedure described by Chen et al. (2013)[18]. Sperm morphological examination was performed as described by Terpsidis et al. (2009)[19] with slight modifications (a drop of the sperm suspension was spread onto a new glass slide for each sample and allowed to dry in air).
-
A complete gross visual pathology inspection was conducted on the testes of all animals during the necropsy by a senior pathologist assisted by a trained team. The testes were trimmed of extraneous fat and weighed immediately. The ratio of organ weight to terminal body weight was calculated according to the following formula: organ body weight ratio = organ weight/body weight × 100%. Tissue sections from testes were fixed with 4% formalin for 24 h, embedded in paraffin, sectioned to 5 µm and stained in hematoxylin and eosin for microscopic observation.
-
Testicular cell apoptosis was measured using a TUNEL assay kit (cat. no. G7360; Promega) according to the manufacturer's protocol. To assess testicular cell apoptosis, 200 different seminiferous tubules were selected randomly from each group (20 tubules/rat) and observed at a magnification of 400×. To determine the apoptotic germ cell index (AI), the percentage of labeled germ cells was calculated in 20 tubules for each rat. Finally, the apoptotic germ cell index‐1 (AI‐1) was determined as the number of tubules containing at least one TUNEL-positive cells/100 tubules, and the apoptotic index‐2 (AI‐2) was described as the number of TUNEL‐positive cells/100 tubules[20, 21].
-
Data were presented as means ± SD (standard deviation). Statistical analyses were performed by using one-way ANOVA to evaluate the homogeneity of the data followed by a least squared differences model or Dunnett's multiple comparison test if the homogeneity evaluation indicated significant deviation variances in SPSS for windows version 11.5 (SPSS Inc., Chicago, IL, USA). Differences were considered to be significant at P < 0.05
-
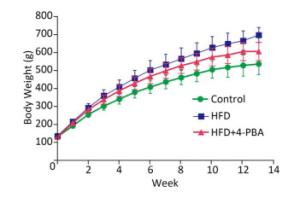
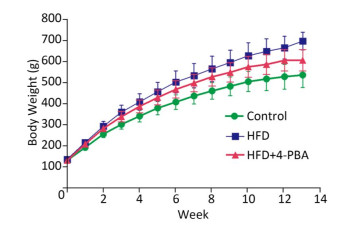
There were significant differences in body weight gain and reproductive organ/body weight loss in rats feed with a HFD compared with control rats (P < 0.05; Figure 1 and Table 1). 4-PBA treatment significantly attenuated HFD‑induced body weight increases and seminal vesicle weight loss (P < 0.05; Figure 1 and Table 1). 4-PBA treatment also showed a tendency to alleviate HFD‑induced organ weight loss of epididymis and testis, but there were no statistically significant differences between the two groups (Figure 1 and Table 1).
Table 1. Effect of HFD Exposure and Supplementation with 4-PBA on Final Body Weights and Reproductive Organ/Body Weight Ratios (n = 10)
Parameter Control HFD HFD+4-PBA Final body weight (g) 534.00 ± 59.03 695.10 ± 41.58* 604.20 ± 51.00*# Seminal vesicle weight/BW (%) 0.36 ± 0.12 0.25 ± 0.04* 0.32 ± 0.06# Epididymis weight/BW (%) 0.26 ± 0.06 0.20 ± 0.02* 0.23 ± 0.03 Testis weight/BW (%) 0.69 ± 0.12 0.53 ± 0.07* 0.59 ± 0.07* Note. *P < 0.05 vs. control and #P < 0.05 vs. HFD; BW: body weight. -
Serum T levels were decreased significantly in the HFD-fed group compared with the control group (P < 0.05; Table 2). However, 4-PBA treatment for 13 weeks significantly ameliorated the HFD‑induced abnormal serum T levels (P < 0.05; Table 2). There were no significant differences in the levels of FSH and LH between the HFD-fed group and the control group. However, a significant difference between serum LH levels of the 4-PBA treatment group and the control group was observed (P < 0.05; Table 2).
Table 2. Effect of HFD Exposure and Supplementation with 4-PBA on Serum Hormones and Sperm Parameters (n = 10)
Parameter Control HFD HFD+4-PBA FSH (mIU/mL) 3.68 ± 0.96 3.88 ± 0.80 3.39 ± 0.77 LH (mIU/mL) 10.24 ± 2.03 8.74 ± 2.37 8.48 ± 1.52* T (ng/mL) 3.65 ± 0.75 2.50 ± 0.51* 3.16 ± 0.68# Sperm Concentration (106/mL) 57.80 ± 6.89 47.67 ± 4.80* 53.30 ± 3.16# Sperm viability (%) 95.60 ± 2.76 91.89 ± 4.14* 97.50 ± 1.43# Sperm motility (%) 67.70 ± 3.37 53.44 ± 4.03* 68.20 ± 3.01# Sperm abnormalities (%) 13.00 ± 3.37 21.00 ± 2.91* 12.60 ± 2.99# Note.*P < 0.05 vs. control and #P < 0.05 vs. HFD. -
Compared with the control, sperm parameters such as concentration, sperm viability, and motility were decreased significantly in rats subjected to a HFD diet (P < 0.05; Table 2). 4-PBA treatment significantly attenuated HFD‑induced impairment of spermatogenesis (P < 0.05; Table 2).
-
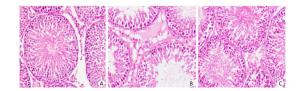
Following 13 weeks of HFD feeding, rats in the HFD-fed group had significantly decreased testis weight/body ratios compared with the control group (P < 0.05; Table 1), which was also confirmed by analysis of histological changes in the testes (Figure 2). In the control group, the testicular structure consisted of abundant seminiferous tubule cells arranged normally and filled with numerous aligned spermatogenetic cells. As shown in Figure 2, atrophic seminiferous tubules, more vacuoles and decreased numbers of spermatogenic cell were observed in the HFD-fed group. However, 4-PBA treatment attenuated extenuated the testicular tissue damage caused by the HFD (Figure 2).
-
TUNEL analysis was used to investigate the apoptosis rate of germ cells in the testis (Figure 3). The results revealed that rats in the HFD group had a significantly higher rate of apoptosis compared with control rats and that 4-PBA significantly inhibited HFD‑induced testicular cell apoptosis (P < 0.05; Figure 4).
-
Obesity refers to excessive accumulation of body fat, which has been proven to associate with many negative impacts on health. The number of overweight and obese people has increased dramatically in the past several decades, both in developing and developed countries, including China. The epidemic of overweight and obesity inevitably results in many obesity‑associated health problems, including infertility[22-24]. Consequently, a great demand to develop or explore pharmacological agents that provide a protective effect against obesity‑associated infertility has been created. The results of the present study revealed that 4-PBA treatment may attenuate HFD‑induced impaired spermatogenesis. 4-PBA treatment was also demonstrated to ameliorate declining serum T and HFD‑induced testicular cell apoptosis.
Spermatogenesis is a complex process that takes place in the seminiferous tubules of the mammalian testes to produce spermatozoa. Sex hormones such as FSH, LH, and T play important roles in this extremely complex process. Testosterone, the primary androgen synthesized in Leydig cells, plays key roles in the development of male reproductive tissues and spermatogenesis. Several recent studies indicated that metformin, fish oil, Asiatic acid, and curcumin ameliorate HFD-induced T level decline, testicular lesions, and germ cell apoptosis and could improve reproductive function in obese male rats[7-10]. Consistent with these studies, the present study demonstrated that 4-PBA upregulates the level of T, which suggests that restoration of T levels may contribute to the protective effects of 4-PBA. 4-PBA had no significant effect on FSH levels. As for LH levels, similar to the results of previous studies, HFD exposure decreased its serum concentration[7-10]. Although a significant difference (P < 0.05; Table 2) was observed in serum levels of LH between the 4-PBA-treated group and the control, we predict that 4-PBA will have little effect on LH, as no significant difference (P < 0.05; Table 2) was found between the HFD group and the 4-PBA group.
Spermatozoa production in the testes is a regulated balance between germ cell proliferation and apoptosis[25]. Apoptosis, also known as programmed cell death, is required for normal spermatogenesis in mammals[26]. However, improper activation of apoptosis in the testis can affect spermatogenesis and cause reproductive defects[27, 28]. Therefore, inhibiting excessive cell apoptosis and reconstructing the balance between cell proliferation and apoptosis may be an effective treatment for defective spermatogenesis. The low concentrations of spermatozoa, poor morphology, and high levels of TUNEL positivity in the HFD group appeared to be correlated with pathological damage of the testes and high levels of testicular cell apoptosis induced by HFD exposure. Our data simply revealed that 4-PBA significantly inhibits testicular cell apoptosis, which suggests that apoptosis may be one of the underlying mechanisms by which 4-PBA protects against HFD-induced defective spermatogenesis.
The precise mechanisms that mediate the anti-apoptotic effects of 4-PBA remain to be elucidated. Many studies indicated that 4-PBA can protect against injury to many tissues through the inhibition of endoplasmic reticulum stress (ERS)[29-33]. According to the above literature, the main mechanisms behind attenuation of ERS by 4-PBA were the following: inhibition of the expression of Chop/GADD153, CHOP, PERK protein, inhibition of ERS-mediated cytokine release, diminished JNK phosphorylation, and prohibition of X-BP1 splicing and caspase-3 and caspase-9 activation[30-33]. Recently, spermatogenic cell apoptosis induced by ERS was shown to play a key role in the induction of reproductive toxicology[34-36]. Further research is required to determine the precise mechanisms underlying the protective effects of 4-PBA.
In conclusion, the results of the present study reflect that 4-PBA can attenuate HFD‑induced spermatogenesis dysfunction via inhibition of excessive apoptosis. These findings provide a theoretical basis for the use of 4-PBA as a treatment for obesity‑associated infertility.
-
The authors declared that there was no conflict of interest.
doi: 10.3967/bes2018.118
-
Abstract:
Objective To determine the mitigating effects of sodium 4-phenylbutyrate (4-PBA) on high-fat diet (HFD)-induced spermatogenesis dysfunction. Methods Male rats (n=30) were randomly divided into three groups:control, HFD, and 4-PBA (HFD +4-PBA). After 13 weeks, rats were euthanized. Testes and epididymis were harvested for further analysis. Sex hormones were detected, and hematoxylin and eosin staining was performed to examine the histological changes in the testes. Semen samples were collected to evaluate sperm quality. Spermatogenic cell apoptosis was detected by TUNEL assay. Results Compared with the control group, the final body weight and body weight gain were significantly higher in HFD-fed rats, while the testicle/body weight ratios were lower (P < 0.05). In HFD-fed rats, obvious pathological changes in the testicular tissue were observed. Treatment with 4-PBA attenuated HFD-induced histological damage, ameliorated the HFD-induced decrease in serum testosterone (T), and reduced the rate of testicular cell apoptosis (P < 0.05) in obese male rats. Finally, 4-PBA significantly improved semen parameters in HFD rats (P < 0.05). Conclusion HFD exposure induced detrimental effects on spermatogenesis, semen quality, serum T level, and testicular cell apoptosis in rats. Treatment with 4-PBA ameliorated HFD induced impaired spermatogenesis via inhibition of apoptosis in rats. 4-PBA may have therapeutic value in the treatment of obesity related impairment of spermatogenesis. -
Key words:
- Male infertility /
- Sodium 4-phenylbutyrate /
- Obesity /
- Spermatogenesis /
- Semen /
- Testis
-
Table 1. Effect of HFD Exposure and Supplementation with 4-PBA on Final Body Weights and Reproductive Organ/Body Weight Ratios (n = 10)
Parameter Control HFD HFD+4-PBA Final body weight (g) 534.00 ± 59.03 695.10 ± 41.58* 604.20 ± 51.00*# Seminal vesicle weight/BW (%) 0.36 ± 0.12 0.25 ± 0.04* 0.32 ± 0.06# Epididymis weight/BW (%) 0.26 ± 0.06 0.20 ± 0.02* 0.23 ± 0.03 Testis weight/BW (%) 0.69 ± 0.12 0.53 ± 0.07* 0.59 ± 0.07* Note. *P < 0.05 vs. control and #P < 0.05 vs. HFD; BW: body weight. Table 2. Effect of HFD Exposure and Supplementation with 4-PBA on Serum Hormones and Sperm Parameters (n = 10)
Parameter Control HFD HFD+4-PBA FSH (mIU/mL) 3.68 ± 0.96 3.88 ± 0.80 3.39 ± 0.77 LH (mIU/mL) 10.24 ± 2.03 8.74 ± 2.37 8.48 ± 1.52* T (ng/mL) 3.65 ± 0.75 2.50 ± 0.51* 3.16 ± 0.68# Sperm Concentration (106/mL) 57.80 ± 6.89 47.67 ± 4.80* 53.30 ± 3.16# Sperm viability (%) 95.60 ± 2.76 91.89 ± 4.14* 97.50 ± 1.43# Sperm motility (%) 67.70 ± 3.37 53.44 ± 4.03* 68.20 ± 3.01# Sperm abnormalities (%) 13.00 ± 3.37 21.00 ± 2.91* 12.60 ± 2.99# Note.*P < 0.05 vs. control and #P < 0.05 vs. HFD. -
[1] Luke B. Pregnancy and birth outcomes in couples with infertility with and without assisted reproductive technology: with an emphasis on US population-based studies. Am J Obstet Gynecol, 2017; 217, 270-81. doi: 10.1016/j.ajog.2017.03.012 [2] Naina K, Kant SA. Trends of male factor infertility, an important cause of infertility: A review of literature. J Hum Reprod Sci, 2015; 8, 191-6. doi: 10.4103/0974-1208.170370 [3] Du PS, Cabler S, Mcalister DA, et al. The effect of obesity on sperm disorders and male infertility. Nat Rev Urol, 2010; 7, 153-61. doi: 10.1038/nrurol.2010.6 [4] Shaha C, Tripathi R, Mishra DP. Male germ cell apoptosis: regulation and biology. Philos Trans R Soc Lond B Biol Sci, 2010; 365, 1501-15. doi: 10.1098/rstb.2009.0124 [5] Russell LD, Chiarini-Garcia H, Korsmeyer, SJ, et al. Bax-dependent spermatogonia apoptosis is required for testicular development and spermatogenesis. Biol Reprod, 2002; 66, 950-8. doi: 10.1095/biolreprod66.4.950 [6] Schaller CE, Wang CL, Beck-Engeser G, et al. Expression of Aire and the early wave of apoptosis in spermatogenesis. J Immunol, 2008; 180, 1338-43. doi: 10.4049/jimmunol.180.3.1338 [7] Yan W, Mu Y, Yu N, et al. Protective effects of metformin on reproductive function in obese male rats induced by high-fat diet. J Assist Reprod Gen, 2015; 32, 1097-104. doi: 10.1007/s10815-015-0506-2 [8] Yang M, Yan WJ, Yin TL, et al. Curcumin ameliorates high-fat diet-induced spermatogenesis dysfunction. Mol Med Rep, 2016; 14, 3588-94. doi: 10.3892/mmr.2016.5712 [9] Wang H, Cai Y, Shao Y, et al. Fish Oil Ameliorates High-Fat Diet Induced Male Mouse Reproductive Dysfunction via Modifying the Rhythmic Expression of Testosterone Synthesis Related Genes. Int J Mol Sci, 2018; 19, E1325. doi: 10.3390/ijms19051325 [10] Miao XL, Gao GM, Jiang L, et al. Asiatic acid attenuates high-fat diet-induced impaired spermatogenesis. Exp Ther Med, 2018; 15, 2397-403. http://www.ncbi.nlm.nih.gov/pubmed/29467846 [11] Okajima M, Takamura M, Usui S, et al. Sodium 4-phenylbutylate protects against myocardial ischemia- reperfusion injury by reducing unfolded protein response- mediated apoptosis in mice. Crit Care, 2010; 14, 2. http://d.old.wanfangdata.com.cn/OAPaper/oai_pubmedcentral.nih.gov_2934080 [12] Vilatoba M, Eckstein C, Bilbao G, et al. Sodium 4-phenylbutyrate protects against liver ischemia reperfusion injury by inhibition of endoplasmic reticulum-stress mediated apoptosis. Surgery, 2005; 138, 342-51, doi: 10.1016/j.surg.2005.04.019 [13] Takatori O, Usui S, Okajima M, et al. Sodium 4-Phenylbutyrate Attenuates Myocardial Reperfusion Injury by Reducing the Unfolded Protein Response. J Cardiovasc Pharmacol Ther, 2016; 22, 283-92. http://www.ncbi.nlm.nih.gov/pubmed/27909014 [14] Ji YL, Wang H, Zhao XF, et al. Crosstalk between endoplasmic reticulum stress and mitochondrial pathway mediates cadmium-induced germ cell apoptosis in testes. Toxicol Sci, 2011; 124, 446-59. doi: 10.1093/toxsci/kfr232 [15] Park CS, Cha H, Kwon EJ, et al. The chemical chaperone 4-phenylbutyric acid attenuates pressure-overload cardiac hypertrophy by alleviating endoplasmic reticulum stress. Biochem Biophys Res Commun, 2012; 421, 578-84. doi: 10.1016/j.bbrc.2012.04.048 [16] Lupachyk S, Watcho P, Stavniichuk R, et al. Endoplasmic reticulum stress plays a key role in the pathogenesis of diabetic peripheral neuropathy. Diabetes, 2013; 62, 944-52. doi: 10.2337/db12-0716 [17] Suresh S, Prithiviraj E, Prakash S. Effect of Mucuna pruriens on oxidative stress mediated damage in aged rat sperm. Int J Androl, 2010; 33, 22-32. doi: 10.1111/j.1365-2605.2008.00949.x [18] Chen XL, Gong LZ, Xu JX. Antioxidative activity and protective effect of probiotics against high-fat diet-induced sperm damage in rats. Animal, 2013; 7, 287-92. doi: 10.1017/S1751731112001528 [19] Terpsidis KI, Papazahariadou MG, Taitzoglou IA, et al. Toxoplasma gondii: reproductive parameters in experimentally infected male rats. Exp Parasitol, 2009; 121, 238-41. doi: 10.1016/j.exppara.2008.11.006 [20] Shokri S, Aitken RJ, Abdolvahhabi M, et al. Exercise and supraphysiological dose of nandrolone decanoate increase apoptosis in spermatogenic cells. Basic Clin Pharmacol Toxicol, 2010; 106, 324-30. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=6a051e29f62ac46e5abf6fdc39f37768 [21] Keyhanmanesh R, Hamidian G, Alipour MR, et al. Protective effects of sodium nitrate against testicular apoptosis and spermatogenesis impairments in streptozotocin-induced diabetic male rats. Life Sci, 2018; 211, 63-73. doi: 10.1016/j.lfs.2018.09.019 [22] Wyatt SB, Winters KP, Dubbert PM. Overweight and Obesity: Prevalence, Consequences, and Causes of a Growing Public Health Problem. Am J Med Sci, 2006; 331, 166-74. doi: 10.1097/00000441-200604000-00002 [23] Hruby A, Hu FB. The Epidemiology of Obesity: A Big Picture. Pharmacoeconomics, 2015; 33, 673-89. doi: 10.1007/s40273-014-0243-x [24] Kahn BE, Brannigan RE. Obesity and male infertility. Curr Opin Urol, 2017; 27, 441-5. doi: 10.1097/MOU.0000000000000417 [25] Jeng HA, Bocca SM. Influence of Exposure to Benzo[a]pyrene on Mice Testicular Germ Cells during Spermatogenesis. J Toxicol, 2013; 2013, 387850. http://europepmc.org/articles/PMC3885195 [26] Shukla KK, Mahdi AA, Rajender S. Apoptosis, spermatogenesis and male infertility. Front Biosci (Elite Ed), 2012; 4, 746-45. http://d.old.wanfangdata.com.cn/NSTLQK/NSTL_QKJJ0215059273/ [27] Hou S, Xian L, Shi P, et al. The Magea gene cluster regulates male germ cell apoptosis without affecting the fertility in mice. Sci Rep, 2016; 6, 26735. doi: 10.1038/srep26735 [28] Sukhotnik I, Nativ O, Roitburt A, et al. Methotrexate induces germ cell apoptosis and impairs spermatogenesis in a rat. Pediatr Surg Int, 2013; 29, 179-84. doi: 10.1007/s00383-012-3197-0 [29] Takada A, Miki T, Kuno AP, et al. Role of ER Stress in Ventricular Contractile Dysfunction in Type 2 Diabetes. Plos One, 2012; 7, e39893. doi: 10.1371/journal.pone.0039893 [30] Liu L, Wu H, Zang J, et al. 4-Phenylbutyric Acid Reveals Good Beneficial Effects on Vital Organ Function via Anti-Endoplasmic Reticulum Stress in Septic Rats. Crit Care Med, 2016; 44, e689-701. doi: 10.1097/CCM.0000000000001662 [31] Carlisle RE, Werner KE, Yum V, et al. Endoplasmic reticulum stress inhibition reduces hypertension through the preservation of resistance blood vessel structure and function. J Hypertens, 2016; 34, 1556-69. doi: 10.1097/HJH.0000000000000943 [32] Malo A, Krüger B, Göke B, et al. 4-Phenylbutyric acid reduces endoplasmic reticulum stress, trypsin activation, and acinar cell apoptosis while increasing secretion in rat pancreatic acini. Pancreas, 2013; 42, 92-101. doi: 10.1097/MPA.0b013e318259f6ca [33] Park CS, Cha H, Kwon EJ, et al. The chemical chaperone 4-phenylbutyric acid attenuates pressure-overload cardiac hypertrophy by alleviating endoplasmic reticulum stress. Biochem Biophys Res Commun, 2012; 421, 578-84. doi: 10.1016/j.bbrc.2012.04.048 [34] Cui Y, Ren L, Li B, et al. Melatonin Relieves Busulfan-Induced Spermatogonial Stem Cell Apoptosis of Mouse Testis by Inhibiting Endoplasmic Reticulum Stress. Cell Physiol Biochem, 2017; 44, 2407-21. doi: 10.1159/000486165 [35] Liu X, Jin X, Su R, et al. The reproductive toxicology of male SD rats after PM2.5 exposure mediated by the stimulation of endoplasmic reticulum stress. Chemosphere, 2017; 189, 547-55. doi: 10.1016/j.chemosphere.2017.09.082 [36] Chow CY, Avila FW, Clark AG, et al. Induction of Excessive Endoplasmic Reticulum Stress in the Drosophila Male Accessory Gland Results in Infertility. Plos One, 2015; 10, e0119386. doi: 10.1371/journal.pone.0119386 -




 下载:
下载:





 Quick Links
Quick Links