-
Infertility, defined as the inability to achieve a clinical pregnancy after 1 year of unprotected and regular sexual intercourse, affects approximately 8%–12% of couples worldwide during their childbearing years. Assisted reproductive technology (ART) offers interventions to facilitate conception in couples with infertility. Since the inception of traditional in vitro fertilization (IVF) in 1978, ART has facilitated the birth of millions of infants. Intracytoplasmic sperm injection (ICSI) is an effective technique for couples ineligible for traditional IVF. Individual female factors, such as age and endometrial thickness, have been shown to affect embryo development during the IVF process, resulting in unfavorable pregnancy outcomes[1-3]. However, the influence of environmental and male factors on early reproductive outcomes cannot be ignored. As a novel form of environmental pollution, light at night (LAN) has intensified with the rapid pace of urbanization, potentially leading to reproductive health problems in both women and men. However, to our knowledge, no study has explored the effects of LAN exposure on the early reproductive outcomes of IVF. Furthermore, the normal development of embryos relies on the contributions of both partners, and the influence of male factors on the early reproductive outcomes of IVF should not be overlooked. Therefore, this study aimed to evaluate the association between outdoor LAN exposure and early reproductive outcomes of IVF while simultaneously considering factors from both partners, using data from a multicenter prospective cohort study conducted in Anhui, China. We hypothesized that high LAN levels are associated with an elevated risk of unfavorable early reproductive outcomes in IVF.
Study participants were drawn from the Infertility Cohort, which is a sub-cohort of the Reproductive Health of Childbearing Couples–Anhui Cohort. This prospective cohort study included couples who underwent infertility examinations at the Reproductive Centers of the Maternal and Child Health Hospital of Ma’anshan City and 901st Hospital of the Joint Logistics Support Force of the People’s Liberation Army between December 2020 and August 2023. Women aged 20–49 years and men aged 22–49 years were eligible, provided they had a time to pregnancy ≥ 12 months or specific identified causes of infertility.
Overall, 2,437 couples were recruited from the Infertility Cohort. Couples were excluded if they had missing electronic medical records (n = 796), underwent intrauterine insemination or rescue-ICSI treatment (n = 253), had karyotype abnormalities (n = 72), or had no transferable embryos (n = 41). Additionally, couples whose detailed residential addresses could not be accurately obtained were excluded (n = 144). In total, 1,131 couples with 1,445 cycles were analyzed (Supplementary Figure S1). This study was approved by the Institutional Review Board of Anhui Medical University (No. 20189999). Written informed consent was obtained from all participants prior to data collection.
The evaluation of LAN utilized global LAN imagery provided by the Earth Observation Group (https://eogdata.mines.edu/products/vnl/). These cloud-free night images were derived from the Visible Infrared Imaging Radiometer Suite Day/Night Band using the Joint Polar Satellite System. We utilized Visible Infrared Imaging Radiometer Suite Day/Night Band nighttime light data with a native resolution of 750 m at the nadir, which underwent additional processing to produce monthly composite products at a 15 arc-second (approximately 500 m) resolution. LAN exposure was quantified as the average radiance measured in nanowatts per square centimeter per steradian (nW/cm2/sr). The LAN exposure level for each participant was calculated based on the average monthly LAN exposure during the 12 months prior to completing the baseline questionnaire. ArcGIS software (ESRI, Redlands, California, USA) was used to calculate LAN exposure levels for each individual.
Early reproductive outcomes of IVF included the total number of oocytes, metaphase II (MII) oocyte numbers, normally fertilized oocyte numbers, blastocyst numbers, best-quality embryo numbers, normal fertilization rates, blastocyst rates, best-quality embryo rates, and fertilization rates. The total numbers of oocytes, MII oocytes, normally fertilized oocytes, blastocysts, and best-quality embryos were evaluated at the hospital and extracted from electronic medical records. The fertilization rate was defined as the number of normally fertilized oocytes divided by the number of MII oocytes. Similarly, the best-quality embryo rate was calculated as the number of best-quality embryos divided by the number of fertilized oocytes. The normal fertilization rate was defined as the number of normally fertilized oocytes divided by the number of fertilized oocytes. Additionally, the blastocyst rate was calculated by dividing the number of blastocysts by the number of fertilized oocytes[4].
Demographic characteristics, including lifestyle factors (use time of electronic devices and sleep chronotypes) and socioeconomic status (education level and annual personal income), were obtained from each participant using a baseline questionnaire. Data on age, height, weight, parity, causes of infertility, ovulation stimulation protocol, and sperm concentration were collected from electronic medical records. Body mass index (BMI) was calculated by dividing the weight (kg) by the square of the height (m). Passive smoking was defined as self-reported exposure to environmental tobacco smoke for over 15 min/d, whether at home or in the workplace. We categorized the frequency of passive smoking exposure over the past year as almost none (0–2 d/week), frequent (3–5 d/week), or nearly every day (≥ 6 d/week). Participants self-reported their sleep chronotypes by responding to the question: “Sleep chronotypes are categorized as 'morning type' and 'evening type.' Which category do you believe best describes your sleep habits?” The options included definite morning (typically sleeping from 9–10 p.m. to 5–6 a.m.), moderate morning (approximating morning chronotypes), moderate evening (approximating evening chronotypes), and definite evening (typically sleeping after 11 p.m. to 7–8 a.m.). Use time for electronic devices was defined as the average daily time spent on smartphones, computers, televisions, and other electronic devices in the past month. Physicians evaluated the causes of infertility using the preoperative health examination results and classified them as female, male, mixed, or unexplained.
Multivariate generalized linear mixed models (GLMM) with random intercepts, using both continuous LAN based on a 10 nW/cm2/sr increase and tertiles of LAN, were fitted to explore the association between LAN levels and early reproductive outcomes of IVF. A binomial distribution and logit link function were used to examine associations with normally fertilized oocyte, blastocyst, best-quality embryo, and fertilization rates. Poisson distribution and log link function were used to analyze the associations between the total number of oocytes, MII oocyte numbers, normally fertilized oocyte numbers, blastocyst numbers, and best-quality embryo numbers. Additionally, we tested whether the associations between LAN and early reproductive outcomes of IVF were modified by treatment protocols (gonadotropin-releasing hormone (GnRH) antagonist vs. non-GnRH antagonist), fertilization protocols (IVF vs. ICSI), female age (≤ 35 vs. > 35), and BMI for women (< 25 kg/m2 vs. ≥ 25 kg/m2). Additionally, we conducted several sensitivity analyses: (1) further adjusted for PM2.5; (2) additionally adjusted for PM2.5, residence (Central Anhui, Northern Anhui, Southern Anhui), season, and baseline enrollment year (2020–2023); and (3) categorized LAN by quartiles rather than tertiles. To improve result interpretation, we back-transformed the regression coefficients for count data using 100 × [exp(β) – 1] to calculate the percent changes.
We utilized generalized additive mixed models to explore the exposure-response relation between LAN and early reproductive outcomes of IVF. When nonlinear correlations were observed, a multi-piecewise GLMM was used to determine the threshold effect of LAN on the early reproductive outcomes of IVF. When the fitted exposure-response curve exhibited an inverted U-shape, the highest point of the plot was taken as the threshold, and when the curve formed a U-shape, the lowest point of the plot was taken as the threshold. The threshold was calculated using a recursive method employing a maximum likelihood model. The plot was divided into multiple parts based on these thresholds, and a GLMM was fitted to each part separately.
We first conducted partner-specific analyses to evaluate the relations between LAN and the total and MII oocyte numbers, as these two outcomes are only associated with women. This analysis was adjusted for female covariates. Given the potential effects of characteristics and LAN exposure from both partners on couple fecundity, when the outcomes involve both partners (normally fertilized oocyte numbers, blastocyst numbers, best-quality embryo numbers, normal fertilization rate, blastocyst rate, best-quality embryo rate, and fertilization rate), we further performed couple-based analyses adjusted for covariates from women. Data analyses were performed using R version 4.2.1. All statistical tests were two-sided, with P < 0.05 considered to be significant.
The participants were divided into tertiles based on LAN exposure levels: T1 (≤ 1.18 nW/cm2/sr), T2 (1.19–17.04 nW/cm2/sr), and T3 (≥ 17.05 nW/cm2/sr) (Table 1). The mean (standard deviation) age of women was 32.85 (5.32) years and of men was 33.94 (6.02) years, respectively. While most female participants reported no alcohol consumption or active smoking in the 6 months preceding baseline (80.3% and 94.4%, respectively), the majority of their male partners reported these behaviors (62.1% and 56.7%, respectively). Over half of the IVF cycles used the GnRH antagonist protocol (58.2%) or IVF insemination (81.7%) (Table 2).
Table 1. Demographic characteristics among 1,131 couples undergoing 1,445 IVF cyclesa
Characteristics Overall (n = 1,131) 0.30 ≤ LAN
≤ 1.18 (n = 427)1.18 < LAN
≤ 17.04 (n = 349)17.04 < LAN
≤ 65.20 (n = 355)P-Valueb Female Age (years) 32.85 (5.32) 31.90 (5.33) 33.05 (5.25) 32.85 (5.32) < 0.001 BMI (kg/m2) 0.470 < 18.5 57 (5.0) 23 (5.4) 16 (4.6) 18 (5.1) 18.5– 675 (59.7) 260 (60.9) 195 (55.9) 220 (62.0) 25.0– 328 (29.0) 121 (28.3) 115 (33.0) 92 (25.9) ≥ 30.0 71 (6.3) 23 (5.4) 23 (7.6) 25 (7.0) Education < 0.001 Less than High school 556 (49.2) 263 (61.6) 204 (58.4) 89 (25.0) High school 186 (16.4) 72 (16.9) 68 (19.5) 46 (13.0) College and above 389 (34.4) 92 (21.5) 77 (22.1) 220 (62.0) Personal income (yuan/year) < 0.001 < 60,000 884 (78.2) 368 (86.1) 293 (84.0) 223 (62.8) ≥ 60,000 247 (21.8) 59 (13.9) 56 (16.0) 132 (37.2) Active smoking 0.110 Never 1068 (94.4) 408 (95.6) 330 (94.6) 330 (93.0) Former or Current 63 (5.6) 19 (4.4) 19 (5.4) 25 (7.0) Passive smoking Never 910 (80.5) 320 (80.8) 278 (76.7) 287 (80.8) 0.840 Frequent 40 (3.5) 15 (3.5) 14 (4.0) 11 (3.1) Nearly every day 181 (16.0) 67 (15.7) 57 (16.3) 57 (16.1) Alcohol use 0.420 Never 908 (80.3) 351 (82.2) 270 (77.4) 287 (80.8) Frequent 213 (18.8) 74 (17.3) 75 (21.5) 64 (18.0) Nearly every day 10 (0.9) 2 (0.5) 4 (1.1) 4 (1.1) Sleep chronotype Definite morning 243 (21.5) 107 (25.1) 79 (22.6) 57 (16.1) 0.010 Moderate morning 244 (21.6) 86 (20.1) 88 (25.2) 70 (19.7) Moderate evening 410 (36.3) 148 (34.7) 111 (31.8) 151 (42.5) Definite evening 234 (20.7) 86 (20.1) 71 (20.3) 77 (21.7) Usage time of electronic devices 5.73 (3.23) 5.66 (3.39) 5.47 (3.14) 6.07 (3.11) 0.030 Male Age (years) 33.94 (6.02) 32.73 (5.58) 34.39 (6.06) 34.94 (6.27) < 0.001 BMI (kg/m2) 0.050 < 18.5 38 (3.4) 16 (3.7) 16 (4.6) 6 (1.7) 18.5–24.9 534 (47.2) 216 (50.6) 167 (47.9) 151 (42.5) 25–29.9 432 (38.2) 148 (34.7) 132 (37.8) 152 (42.8) ≥ 30 127 (11.2) 47 (11.0) 34 (9.7) 46 (13.0) Education < 0.001 Less than high school 471 (41.6) 215 (50.4) 184 (52.7) 72 (20.3) High school 237 (21.0) 95 (22.2) 73 (20.9) 69 (19.4) College and above 423 (37.4) 117 (27.4) 92 (26.3) 214 (60.3) Personal income (yuan/year) < 0.001 < 60,000 528 (46.7) 230 (53.9) 184 (52.7) 233 (65.6) ≥ 60,000 603 (53.3) 197 (46.1) 165 (47.3) 122 (34.4) Active smoking 0.010 Never 490 (43.3) 181 (42.4) 131 (37.5) 178 (50.1) Former or Current 641 (56.7) 246 (57.6) 218 (2.5) 177 (49.9) Passive smoking 0.300 Never 745 (65.9) 302 (70.8) 216 (61.9) 227 (64.0) Frequent 93 (8.2) 33 (7.7) 29 (8.3) 31 (8.7) Nearly every day 293 (25.9) 92 (21.5) 104 (29.8) 97 (27.3) Alcohol use 0.400 Never 428 (37.8) 160 (37.5) 123 (35.2) 145 (40.8) Frequent 549 (48.5) 213 (49.9) 170 (48.7) 166 (46.8) Nearly every day 154 (13.6) 54 (12.6) 56 (16.0) 44 (12.4) Sleep chronotype 0.003 Definite morning 215 (19.0) 82 (19.2) 83 (23.8) 50 (14.1) Moderate morning 221 (19.5) 89 (20.8) 69 (19.8) 63 (17.7) Moderate evening 433 (38.3) 172 (40.3) 109 (31.2) 152 (42.8) Definite evening 262 (23.2) 84 (19.7) 88 (25.2) 90 (25.4) Usage time of electronic devices 6.52 (3.74) 6.47 (3.63) 6.45 (3.80) 6.64 (3.84) 0.800 Infertility causes < 0.001 Female factor 713 (63.0) 304 (71.2) 231 (66.2) 178 (50.1) Male factor 68 (6.0) 26 (6.1) 20 (5.7) 22 (6.2) Mixed factors 311 (27.5) 82 (19.2) 91 (26.1) 138 (38.9) Unexplained factors 39 (3.4) 15 (3.5) 7 (2.0) 17 (4.8) Note. IVF, in vitro fertilization; SD, standard deviation; LAN, light at night. aValues are presented as mean (SD) for continuous variables or as n (%) for categorical variables. bP-values were calculated using chi-square tests for categorical variables and Kruskal–Wallis tests for non-normally distributed continuous variables. Table 2. Cycle-specific clinical characteristics from 1,445 ART cycles (n = 1,131) in this study
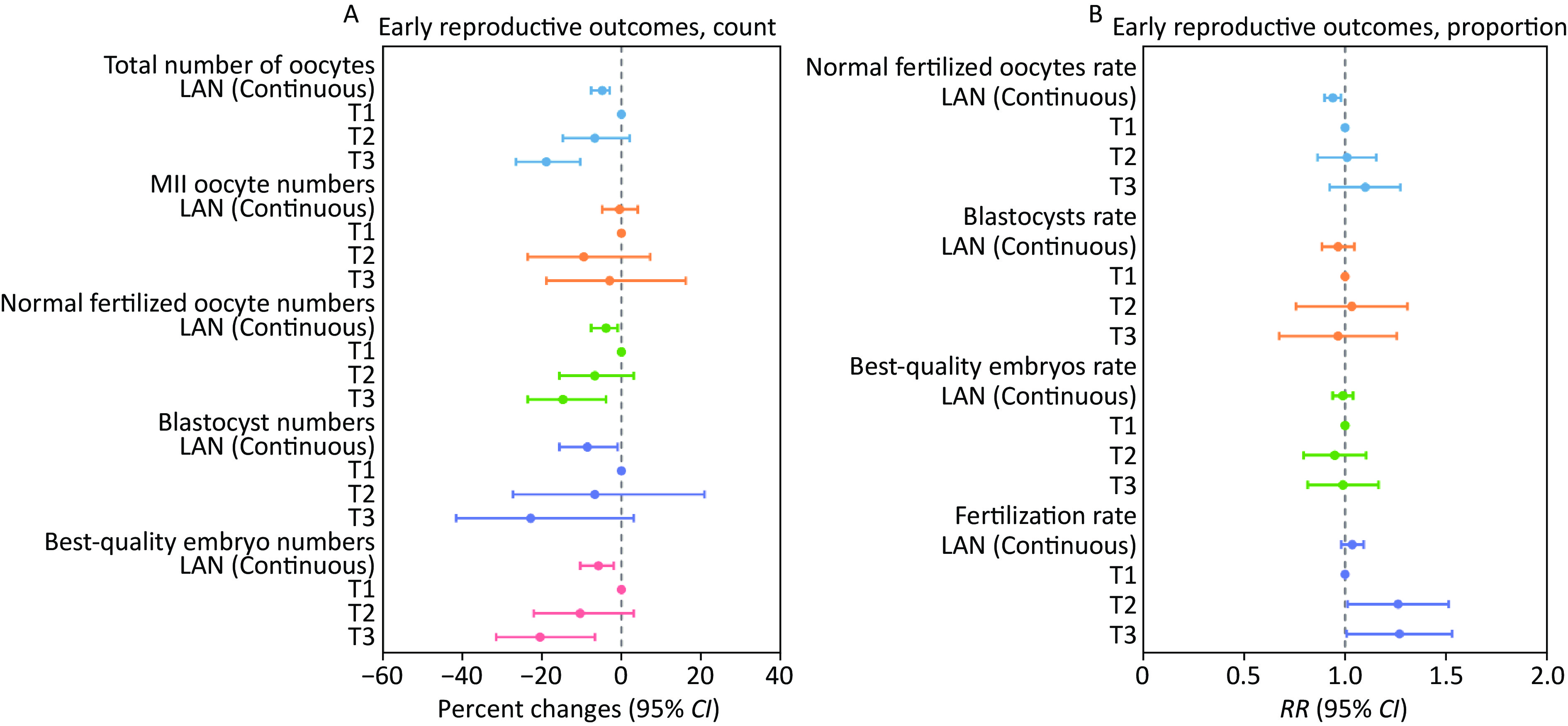
Characteristics Mean (SD) or n (%) Fertilization protocol IVF 1,180 (81.7) ICSI 265 (18.3) Treatment protocol GnRH antagonist 841 (58.2) Non-GnRH antagonist 604 (41.8) Type of embryos transferred No embryos transferred 482 (33.4) Fresh embryos transferred 623 (43.1) Frozen-thawed embryos transferred 340 (23.5) Controlled ovarian hyperstimulation outcomes Total number of oocytes 12.56 (9.26) MII oocyte numbers 7.38 (6.18) Normal fertilized oocyte numbers 6.49 (5.24) Blastocyst numbers 2.26 (3.69) Best-quality embryo numbers 2.85 (3.11) ART cycles 1 908 (80.3) 2 157 (13.9) 3 49 (4.3) 4 12 (1.1) 5 2 (0.2) 6 3 (0.3) Note. IVF, in vitro fertilization; ICSI, intracytoplasmic sperm injection; GnRH, gonadotropin-releasing hormone; MII, metaphase II; SD, standard deviation; ART, assisted reproductive technology. After adjusting for female age, BMI, education level, drinking status, passive smoking status, annual personal income, parity, ovulation stimulation protocol, use time of electronic devices, and sleep chronotypes, for every 10-unit (nW/cm2/sr) increase in LAN, there was a significant decrease in the total number of oocytes by 4.88% [95% confidence interval (CI): -7.69%, -2.96%], but no significant change in MII oocyte numbers (Figure 1 and Supplementary Table S1). We also considered the association between couple-level factors and early reproductive outcomes. Specifically, after additionally adjusting for male partner factors, including sperm concentration, smoking status, drinking status, education level, age difference between partners, annual personal income, use time of electronic devices, and sleep chronotypes, we found a significant negative association between LAN and early reproductive outcomes, which demonstrated that for every 10-unit (nW/cm2/sr) increase in LAN, the numbers of normally fertilized oocytes, blastocysts, and best-quality embryos decreased by 3.92% (95% CI: –7.69%, –1.00%), 8.61% (95% CI: –15.63%, –1.00%), and 5.82% (95% CI: –10.42%, –1.98%), respectively (Figure 1 and Supplementary Table S1). LAN also exhibited a significant negative association with the normal fertilization rate (RR = 0.94, 95% CI: 0.90, 0.98) (Figure 1 and Supplementary Table S1). For LAN levels modeled as categorical variables, these negative associations generally persisted (Figure 1). Compared with those in the first tertile, participants in the third tertile of LAN had 18.94% (95% CI: –26.66%, –10.42%) lower total oocyte numbers, 14.79% (95% CI: –23.66%, –3.92%) lower normally fertilized oocyte numbers, and 20.55% (95% CI: –31.61%, –6.76%) lower best-quality embryo numbers (Figure 1 and Supplementary Table S2). As shown in Supplementary Figure S2, the results of the stratified analysis were generally consistent with those of the main analysis. For example, in the IVF group, for every 10-unit (nW/cm2/sr) increase in LAN, the total number of oocytes, normally fertilized oocytes, blastocysts, and best-quality embryos decreased by 5.40% (95% CI: –8.19%, –2.52%), 4.78% (95% CI: –8.22%, –1.22%), 9.88% (95% CI: –17.57%, –1.47%), and 6.85% (95% CI: –11.22%, –2.27%), respectively (Supplementary Figure S2 and Supplementary Tables S3–S6). The results of the sensitivity analyses were generally consistent with those of the primary analyses (Supplementary Tables S7, S9, and S10). To date, no epidemiological studies have explored the association between outdoor LAN and the early reproductive outcomes of IVF in couples undergoing ART. While no prior epidemiological studies have directly examined outdoor LAN and the early reproductive outcomes of IVF, our results align with broader evidence linking circadian disruption to reproductive dysfunction. Animal studies have demonstrated that LAN reduces fecundity. Drosophila melanogaster exposed to chronic LAN exhibit diminished egg production[5], and rodents subjected to altered light cycles show impaired pregnancy rates and litter size[6,7]. In humans, shift work (a proxy for circadian disruption) is associated with decreased fecundability[8,9] and fewer mature oocytes in women[10], which is consistent with our findings that outdoor LAN is associated with reduced total oocyte numbers and normal fertilization rates. Our study extends this evidence by quantifying dose-dependent thresholds for LAN’s effects on specific IVF parameters, providing novel insights into environmental influences on ART success. Practical mitigation strategies for couples undergoing ART include using blackout curtains in bedrooms and minimizing LAN exposure during ART. These easily implementable measures may help reduce potential LAN-related risks while awaiting further validation. From a public health perspective, our results suggest that light pollution is a modifiable risk factor in preconception care guidelines, particularly in populations undergoing ART.
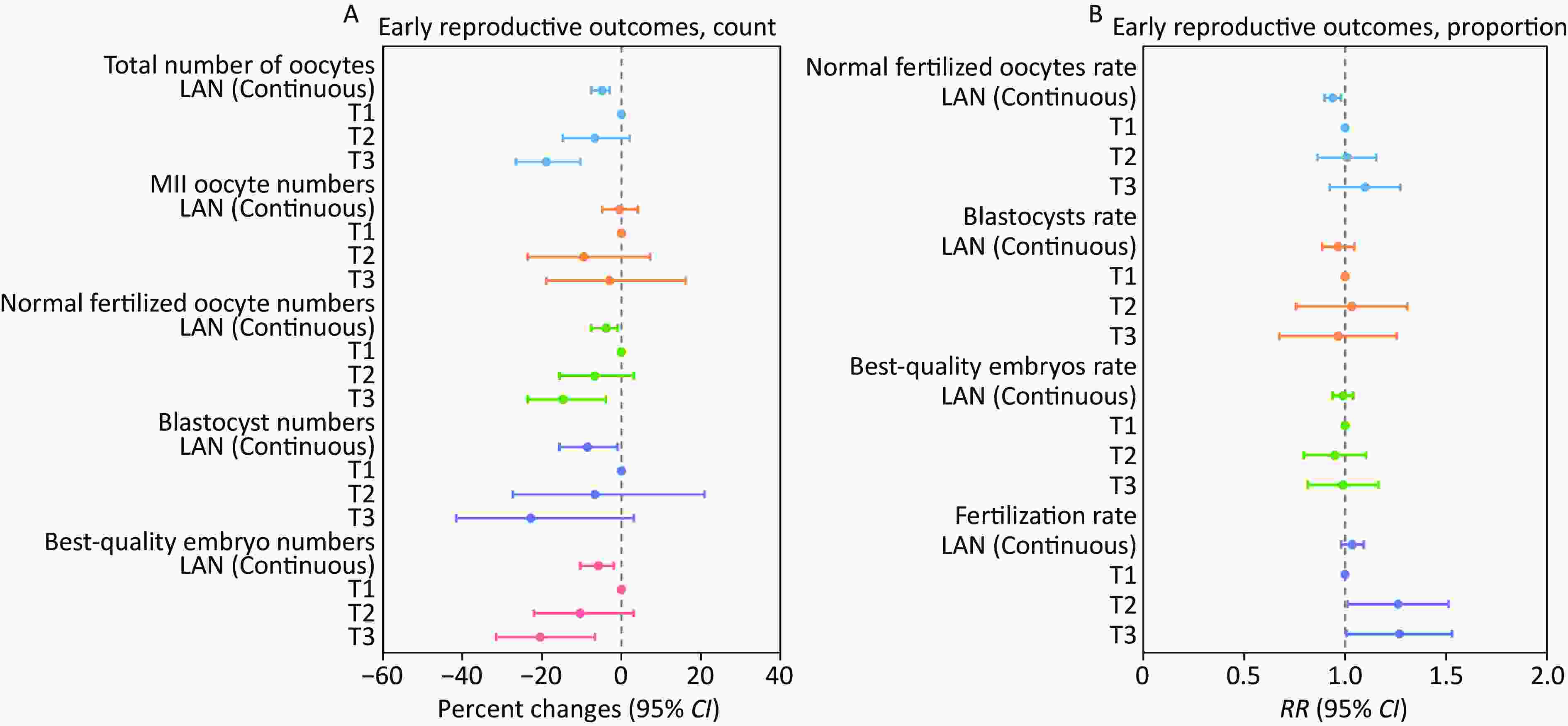
Figure 1. Associations between LAN (continuous and categorical) and early reproductive outcomes of IVF among 1,131 couples undergoing 1,445 IVF cycles. The analyses were conducted using generalized linear mixed models with random intercepts. For (A), a Poisson distribution and log link function were applied, with data presented as percent changes (95% CI). For (B), a binomial distribution and logit link function were utilized, with data presented as RR (95% CI). The cut point of LAN: T1 (≤ 1.18 nW/cm2/sr), T2 (1.19–17.04 nW/cm2/sr), T3 (≥ 17.05 nW/cm2/sr). LAN, light at night; MII, metaphase II; CI, confidence interval; RR, relative risk.
Overall, the relation between LAN and most early reproductive IVF outcomes showed a negative trend (Supplementary Figure S3). GLMM conducted separately on both sides of the threshold for the normal fertilization rate and fertilization rate showed that for each 10-unit (nW/cm2/sr) increase in LAN below the first threshold, the normal fertilization rate increased by 4.0% (1.0%, 7.0%), while when LAN exceeded the first threshold but remained below the second, the normal fertilization rate and fertilization rate decreased by 7.0% (–14.0%, –1.0%) and 6.0% (–11.0%, –1.0%), respectively (Supplementary Table S8). Above the second LAN threshold, neither the normal fertilization rate nor the fertilization rate was significantly associated with LAN exposure. These findings suggest that LAN may impair early IVF outcomes, particularly at moderate exposure levels, with potential saturation or adaptive mechanisms occurring at high exposure levels.
This study had several limitations. First, while this study provides important insights into LAN's effects on IVF outcomes, the recruitment of participants from infertility clinics may limit its generalizability to the general population. Second, individual data on indoor LAN exposure were unavailable. While we collected data on the participants’ electronic device use time for non-work/study purposes, we were unable to accurately assess the duration of nighttime device use. The potential use of blackout curtains by participants may have led to exposure misclassification. This may have led to an overestimation of the association between LAN exposure and the early reproductive outcomes of IVF. The lack of data on participants' occupational schedules, including potential night or shift work, represents an important limitation of our study. For example, work chronotypes may independently influence reproductive outcomes. In addition, our study lacked information on participants' use of blackout curtains, which may have led to exposure misclassification, as well as data on dietary chronotypes that could potentially confound the observed associations. Future studies should consider collecting these important covariates. Finally, we were unable to address all potential unmeasured or residual confounding factors, such as changes in residence. Further studies are needed to obtain more representative and robust conclusions.
doi: 10.3967/bes2025.101
Influence of Outdoor Light at Night on Early Reproductive Outcomes of In Vitro Fertilization and Its Threshold Effect: Evidence from a Couple-Based Preconception Cohort Study
-
Investigation and writing of the original draft preparation: Wenbin Fang, Ying Tang; Data management and formal analysis: Yaning Sun, Yanlan Tang, Yinyin Chen, Yawen Cao; Supervision and methodology: Jiqi Fang, Kunjing He, Yushan Li; Methodology and interpretation of results: Yaning Dai, Shuangshuang Bao, Peng Zhu; Data management and investigation: Shanshan Shao; Supervision and funding acquisition: Fangbiao Tao, Ying Tang, Guixia Pan. All the authors have read and approved the manuscript.
All authors declare no conflicts of interest.
&These authors contributed equally to this work.
注释:1) Authors’ Contributions: 2) Competing Interests: -
Figure 1. Associations between LAN (continuous and categorical) and early reproductive outcomes of IVF among 1,131 couples undergoing 1,445 IVF cycles. The analyses were conducted using generalized linear mixed models with random intercepts. For (A), a Poisson distribution and log link function were applied, with data presented as percent changes (95% CI). For (B), a binomial distribution and logit link function were utilized, with data presented as RR (95% CI). The cut point of LAN: T1 (≤ 1.18 nW/cm2/sr), T2 (1.19–17.04 nW/cm2/sr), T3 (≥ 17.05 nW/cm2/sr). LAN, light at night; MII, metaphase II; CI, confidence interval; RR, relative risk.
Table 1. Demographic characteristics among 1,131 couples undergoing 1,445 IVF cyclesa
Characteristics Overall (n = 1,131) 0.30 ≤ LAN
≤ 1.18 (n = 427)1.18 < LAN
≤ 17.04 (n = 349)17.04 < LAN
≤ 65.20 (n = 355)P-Valueb Female Age (years) 32.85 (5.32) 31.90 (5.33) 33.05 (5.25) 32.85 (5.32) < 0.001 BMI (kg/m2) 0.470 < 18.5 57 (5.0) 23 (5.4) 16 (4.6) 18 (5.1) 18.5– 675 (59.7) 260 (60.9) 195 (55.9) 220 (62.0) 25.0– 328 (29.0) 121 (28.3) 115 (33.0) 92 (25.9) ≥ 30.0 71 (6.3) 23 (5.4) 23 (7.6) 25 (7.0) Education < 0.001 Less than High school 556 (49.2) 263 (61.6) 204 (58.4) 89 (25.0) High school 186 (16.4) 72 (16.9) 68 (19.5) 46 (13.0) College and above 389 (34.4) 92 (21.5) 77 (22.1) 220 (62.0) Personal income (yuan/year) < 0.001 < 60,000 884 (78.2) 368 (86.1) 293 (84.0) 223 (62.8) ≥ 60,000 247 (21.8) 59 (13.9) 56 (16.0) 132 (37.2) Active smoking 0.110 Never 1068 (94.4) 408 (95.6) 330 (94.6) 330 (93.0) Former or Current 63 (5.6) 19 (4.4) 19 (5.4) 25 (7.0) Passive smoking Never 910 (80.5) 320 (80.8) 278 (76.7) 287 (80.8) 0.840 Frequent 40 (3.5) 15 (3.5) 14 (4.0) 11 (3.1) Nearly every day 181 (16.0) 67 (15.7) 57 (16.3) 57 (16.1) Alcohol use 0.420 Never 908 (80.3) 351 (82.2) 270 (77.4) 287 (80.8) Frequent 213 (18.8) 74 (17.3) 75 (21.5) 64 (18.0) Nearly every day 10 (0.9) 2 (0.5) 4 (1.1) 4 (1.1) Sleep chronotype Definite morning 243 (21.5) 107 (25.1) 79 (22.6) 57 (16.1) 0.010 Moderate morning 244 (21.6) 86 (20.1) 88 (25.2) 70 (19.7) Moderate evening 410 (36.3) 148 (34.7) 111 (31.8) 151 (42.5) Definite evening 234 (20.7) 86 (20.1) 71 (20.3) 77 (21.7) Usage time of electronic devices 5.73 (3.23) 5.66 (3.39) 5.47 (3.14) 6.07 (3.11) 0.030 Male Age (years) 33.94 (6.02) 32.73 (5.58) 34.39 (6.06) 34.94 (6.27) < 0.001 BMI (kg/m2) 0.050 < 18.5 38 (3.4) 16 (3.7) 16 (4.6) 6 (1.7) 18.5–24.9 534 (47.2) 216 (50.6) 167 (47.9) 151 (42.5) 25–29.9 432 (38.2) 148 (34.7) 132 (37.8) 152 (42.8) ≥ 30 127 (11.2) 47 (11.0) 34 (9.7) 46 (13.0) Education < 0.001 Less than high school 471 (41.6) 215 (50.4) 184 (52.7) 72 (20.3) High school 237 (21.0) 95 (22.2) 73 (20.9) 69 (19.4) College and above 423 (37.4) 117 (27.4) 92 (26.3) 214 (60.3) Personal income (yuan/year) < 0.001 < 60,000 528 (46.7) 230 (53.9) 184 (52.7) 233 (65.6) ≥ 60,000 603 (53.3) 197 (46.1) 165 (47.3) 122 (34.4) Active smoking 0.010 Never 490 (43.3) 181 (42.4) 131 (37.5) 178 (50.1) Former or Current 641 (56.7) 246 (57.6) 218 (2.5) 177 (49.9) Passive smoking 0.300 Never 745 (65.9) 302 (70.8) 216 (61.9) 227 (64.0) Frequent 93 (8.2) 33 (7.7) 29 (8.3) 31 (8.7) Nearly every day 293 (25.9) 92 (21.5) 104 (29.8) 97 (27.3) Alcohol use 0.400 Never 428 (37.8) 160 (37.5) 123 (35.2) 145 (40.8) Frequent 549 (48.5) 213 (49.9) 170 (48.7) 166 (46.8) Nearly every day 154 (13.6) 54 (12.6) 56 (16.0) 44 (12.4) Sleep chronotype 0.003 Definite morning 215 (19.0) 82 (19.2) 83 (23.8) 50 (14.1) Moderate morning 221 (19.5) 89 (20.8) 69 (19.8) 63 (17.7) Moderate evening 433 (38.3) 172 (40.3) 109 (31.2) 152 (42.8) Definite evening 262 (23.2) 84 (19.7) 88 (25.2) 90 (25.4) Usage time of electronic devices 6.52 (3.74) 6.47 (3.63) 6.45 (3.80) 6.64 (3.84) 0.800 Infertility causes < 0.001 Female factor 713 (63.0) 304 (71.2) 231 (66.2) 178 (50.1) Male factor 68 (6.0) 26 (6.1) 20 (5.7) 22 (6.2) Mixed factors 311 (27.5) 82 (19.2) 91 (26.1) 138 (38.9) Unexplained factors 39 (3.4) 15 (3.5) 7 (2.0) 17 (4.8) Note. IVF, in vitro fertilization; SD, standard deviation; LAN, light at night. aValues are presented as mean (SD) for continuous variables or as n (%) for categorical variables. bP-values were calculated using chi-square tests for categorical variables and Kruskal–Wallis tests for non-normally distributed continuous variables. Table 2. Cycle-specific clinical characteristics from 1,445 ART cycles (n = 1,131) in this study
Characteristics Mean (SD) or n (%) Fertilization protocol IVF 1,180 (81.7) ICSI 265 (18.3) Treatment protocol GnRH antagonist 841 (58.2) Non-GnRH antagonist 604 (41.8) Type of embryos transferred No embryos transferred 482 (33.4) Fresh embryos transferred 623 (43.1) Frozen-thawed embryos transferred 340 (23.5) Controlled ovarian hyperstimulation outcomes Total number of oocytes 12.56 (9.26) MII oocyte numbers 7.38 (6.18) Normal fertilized oocyte numbers 6.49 (5.24) Blastocyst numbers 2.26 (3.69) Best-quality embryo numbers 2.85 (3.11) ART cycles 1 908 (80.3) 2 157 (13.9) 3 49 (4.3) 4 12 (1.1) 5 2 (0.2) 6 3 (0.3) Note. IVF, in vitro fertilization; ICSI, intracytoplasmic sperm injection; GnRH, gonadotropin-releasing hormone; MII, metaphase II; SD, standard deviation; ART, assisted reproductive technology. -
[1] Broekmans FJ, Kwee J, Hendriks DJ, et al. A systematic review of tests predicting ovarian reserve and IVF outcome. Hum Reprod Update, 2006; 12, 685−718. doi: 10.1093/humupd/dml034 [2] Eldar-Geva T, Ben-Chetrit A, Spitz IM, et al. Dynamic assays of inhibin B, anti-Mullerian hormone and estradiol following FSH stimulation and ovarian ultrasonography as predictors of IVF outcome. Hum Reprod, 2005; 20, 3178−83. doi: 10.1093/humrep/dei203 [3] Pathare ADS, Loid M, Saare M, et al. Endometrial receptivity in women of advanced age: an underrated factor in infertility. Hum Reprod Update, 2023; 29, 773−93. doi: 10.1093/humupd/dmad019 [4] Li DY, Liang CM, Cao Y, et al. The associations of serum metals concentrations with the intermediate and pregnancy outcomes in women undergoing in vitro fertilization (IVF). Ecotoxicol Environ Saf, 2022; 233, 113309. doi: 10.1016/j.ecoenv.2022.113309 [5] McLay LK, Green MP, Jones TM. Chronic exposure to dim artificial light at night decreases fecundity and adult survival in Drosophila melanogaster. J Insect Physiol, 2017; 100, 15−20. doi: 10.1016/j.jinsphys.2017.04.009 [6] Bahougne T, Kretz M, Angelopoulou E, et al. Impact of circadian disruption on female mice reproductive function. Endocrinology, 2020; 161, bqaa028. doi: 10.1210/endocr/bqaa028 [7] Summa KC, Vitaterna MH, Turek FW. Environmental perturbation of the circadian clock disrupts pregnancy in the mouse. PLoS One, 2012; 7, e37668. doi: 10.1371/journal.pone.0037668 [8] Lin YC, Chen MH, Hsieh CJ, et al. Effect of rotating shift work on childbearing and birth weight: a study of women working in a semiconductor manufacturing factory. World J Pediatr, 2011; 7, 129−35. doi: 10.1007/s12519-011-0265-9 [9] Sponholtz TR, Bethea TN, Ruiz-Narváez EA, et al. Night shift work and fecundability in late reproductive-aged African American women. J Womens Health (Larchmt), 2021; 30, 137−44. doi: 10.1089/jwh.2019.8166 [10] Mínguez-Alarcón L, Souter I, Williams PL, et al. Occupational factors and markers of ovarian reserve and response among women at a fertility centre. Occup Environ Med, 2017; 74, 426−31. doi: 10.1136/oemed-2016-103953 -




 下载:
下载:



 Quick Links
Quick Links